Abstract
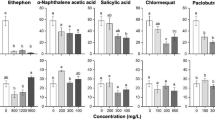
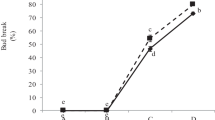
Reactivation of dormant meristem in banjhi (dormant) shoots is important to enhance the quality and quantity of tea production. The field grown tea bushes were subjected to treatment with dormancy breaking agents such as potassium nitrate (KNO3), thiourea, sodium nitro prusside (SNP), the phytohormones kinetin (Kn) and gibberellins (GA). The efficacy of Kn and GA were comparatively lesser than KNO3 while the combination of Kn and GA (50 and100 ppm respectively) resulted in better dormancy reduction in tea buds. This observation was supported by our results from gene expression study where accumulation patterns of mRNAs corresponding to histones (H2A, H2B, H3 and H4), cyclins (B2, D1 and D3), cyclin-dependent kinase (CDKA), ubiquitination enzymes (FUS, EXT CE2), cyclophilin, E2F, and tubulin were analyzed during growth-dormancy cycles in tea apical buds under the influence of Kn, GA and their combinations. The level of these mRNAs was low in dormant buds, which was significantly increased by foliar application of GA and Kn combination. The present study indicated that the foliar application of GA in combination with Kn will help to improve quality and quantity of tea production by breaking dormancy and stimulating the bud growth.


Similar content being viewed by others
References
Bajji M, MʼHamdi M, Gastiny F et al (2007) Catalase inhibition accelerates dormancy release and sprouting in potato (Solanum tuberosum L.) tubers. Biotechnol Agron Soc Environ 11:121–131
Balasaravanan T, Pius PK, Raj Kumar R et al (2003) Genetic diversity among south Indian tea germplasm (Camellia sinensis, C. assamica and C. assamica spp. lasiocalyx) using AFLP markers. Plant Sci 165:365–372. https://doi.org/10.1016/S0168-9452(03)00196-1
Barua DN (1969) Seasonal Dormancy in Tea (Camellia sinensis L.). Nature 224:514. https://doi.org/10.1038/224514a0
Barua DN, Das SC (1979) Mechanism of growth periodicity in tea [India]. Two A Bud 26:36–40
Bond TET (1942) Studies in the vegetative growth and anatomy of the tea plant (Camellia thea Link.) with special reference to the phloem. Ann Bot 6:607–630. https://doi.org/10.1093/oxfordjournals.aob.a088424
Brault M, Maldiney R (1999) Mechanisms of cytokinin action. Plant Physiol Biochem 37:403–412. https://doi.org/10.1016/S0981-9428(99)80046-1
Campbell MA, Suttle JC, Sell TW (1996) Changes in cell cycle status and expression of p34cdc2 kinase during potato tuber meristem dormancy. Physiol Plant 98:743–752. https://doi.org/10.1111/j.1399-3054.1996.tb06680.x
Chen Z, Hagler J, Palombella VJ et al (1995) Signal-induced site-specific phosphorylation targets I kappa B alpha to the ubiquitin-proteasome pathway. Genes Dev 9:1586–1597
Cleland RE (1999) Nature, occurrence, and functioning of plant hormones. In: Hooykaas PJJ, Hall MA, Libbenga KR (eds) Biochemistry and molecularbiology of plant hormones, 1st edn. Elsevier Science, Amsterdam, pp 3–22
Davies WJ, Zhang J (1991) Root signals and the regulation of growth and development of plants in drying soil. Annu Rev Plant Physiol Plant Mol Biol 42:55–76. https://doi.org/10.1146/annurev.pp.42.060191.000415
Devitt ML, Stafstrom JP (1995) Cell cycle regulation during growth-dormancy cycles in pea axillary buds. Plant Mol Biol 29:255–265. https://doi.org/10.1007/BF00043650
Dunlap JR, Morgan PW (1977) Reversal of induced dormancy in lettuce by ethylene, kinetin, and gibberellic acid. Plant Physiol 60:222–224
Erez A, Viémont JD, Crabbé J (2000) Bud dormancy: a suggestion for the control mechanism and its evolution, dormancy in plants: from whole plant behaviour to cellular control. UKCAB International, Wallingford
Fobert PR, Coen ES, Murphy GJ, Doonan JH (1994) Patterns of cell division revealed by transcriptional regulation of genes during the cell cycle in plants. EMBO J 13:616–624
Fontaine O, Huault C, Pavis N, Billard JP (1994) Dormancy breakage of Hordeum vulgare seeds: effects of hydrogen peroxide and scarification on glutathione level and glutathione reductase activity. Plant Physiol Biochem 32:677–683
Freeman D, Riou-Khamlichi C, Oakenfull EA, Murray JAH (2003) Isolation, characterization and expression of cyclin and cyclin-dependent kinase genes in Jerusalem artichoke (Helianthus tuberosus L.). J Exp Bot 54:303–308
Hao X, Yang Y, Yue C et al (2017) Comprehensive transcriptome analyses reveal differential gene expression profiles of Camellia sinensis axillary buds at para-, endo-, ecodormancy, and bud flush stages. Front Plant Sci. https://doi.org/10.3389/fpls.2017.00553
Hartmann A, Senning M, Hedden P et al (2011) Reactivation of meristem activity and sprout growth in potato tubers require both cytokinin and gibberellin. Plant Physiol 155:776–796. https://doi.org/10.1104/pp.110.168252
Horvath DP, Chao WS, Anderson JV (2002) Molecular analysis of signals controlling dormancy and growth in underground adventitious buds of leafy spurge. Plant Physiol 128:1439–1446. https://doi.org/10.1104/pp.010885
Hu Y, Bao F, Li J (2000) Promotive effect of brassinosteroids on cell division involves a distinct CycD3-induction pathway in Arabidopsis. Plant J 24:693–701
Jeleńska J, Deckert J, Kondorosi E, Legocki AB (2000) Mitotic B-type cyclins are differentially regulated by phytohormones and during yellow lupine nodule development. Plant Sci 150:29–39. https://doi.org/10.1016/S0168-9452(99)00158-2
Kabar K (1998) Comparative effects of kinetin, benzyladenine, and gibberellic acid on abscisic acid inhibited seed germination and seedling growth of red pine and arbor vitae. Turk J Bot 22:1–6
Khattab HI, Emam MM, Shehata MM (2000) The correlative changes associated with bud dormancy and rooting of cane cuttings in grapevine. Egypt J Biotechnol 7:255–274
Koller D, Mayer AM, Poljakoff-Mayber A, Klein S (1962) Seed germination. Annu Rev Plant Physiol 13:437–464. https://doi.org/10.1146/annurev.pp.13.060162.002253
Krishnaraj T, Gajjeraman P, Palanisamy S et al (2011) Identification of differentially expressed genes in dormant (banjhi) bud of tea (Camellia sinensis (L.) O. Kuntze) using subtractive hybridization approach. Plant Physiol Biochem 49:565–571. https://doi.org/10.1016/j.plaphy.2011.03.011
Kulasegarum S (1969) Studies on the dormancy of tea shoots. I. Hormonal stimulation of the growth of dormant buds. Tea Q 40:31–46
Lang GA (1987) Dormancy: a new universal terminology. HortScience 22:817–820
Lang G, Early J, Martin G, Darnell R (1987) Endo-, para-, and ecodormancy: physiological terminology and classification for dormancy research. HortScience 22:371–377
Mazzitelli L, Hancock RD, Haupt S et al (2007) Co-ordinated gene expression during phases of dormancy release in raspberry (Rubus idaeus L.) buds. J Exp Bot 58:1035–1045. https://doi.org/10.1093/jxb/erl266
Mok MC (1994) Cytokinins and plant development—an overview. In: Mok DWS, Mok MC (eds) Cytokinins chemistry, activity and function. CRC Press, Boca Raton, pp 155–166
Nir G, Shulman Y, Fanberstein L, Lavee S (1986) changes in the activity of catalase (EC 1.11.1.6) in relation to the dormancy of grapevine (Vitis vinifera L.) buds. Plant Physiol 81:1140–1142
Noppakoonwong U, Sripinta P, Pasopa P et al (2005) A trial of rest-breaking chemicals on low-chill peach and nectarine. ACIAR Tech Reports Ser 73–80
Pérez FJ, Lira W (2005) Possible role of catalase in post-dormancy bud break in grapevines. J Plant Physiol 162:301–308. https://doi.org/10.1016/j.jplph.2004.07.011
Rebers M, Romeijn G, Knegt E, Plas LHW (1994) Effects of exogenous gibberellins and paclobutrazol on floral stalk growth of tulip sprouts isolated from cooled and non-cooled tulip bulbs. Physiol Plant 92:661–667. https://doi.org/10.1111/j.1399-3054.1994.tb03037.x
Riou-Khamlichi C, Huntley R, Jacqmard A, Murray JA (1999) Cytokinin activation of Arabidopsis cell division through a D-type cyclin. Science 283:1541–1544
Rosin FM, Hart JK, Van Onckelen H, Hannapel DJ (2003) Suppression of a vegetative MADS box gene of potato activates axillary meristem development. Plant Physiol 131:1613–1622. https://doi.org/10.1104/pp.102.012500
Rossouw JA (2008) Effect of cytokinin and gibberellin on potato tuber dormancy. University of Pretoria, Pretoria
Shimizu S, Mori H (1998) Analysis of cycles of dormancy and growth in pea axillary buds based on mRNA accumulation patterns of cell cycle-related genes. Plant Cell Physiol 39:255–262
Soni R, Carmichael JP, Shah ZH, Murray JA (1995) A family of cyclin D homologs from plants differentially controlled by growth regulators and containing the conserved retinoblastoma protein interaction motif. Plant Cell 7:85–103
Stephens W, Carr MKV (1990) Seasonal and clonal differences in shoot extension rates and numbers in tea (Camellia sinensis). Exp Agric 26:83. https://doi.org/10.1017/S001447970001543X
Suttle JC (2004) Involvement of endogenous gibberellins in potato tuber dormancy and early sprout growth: a critical assessment. J Plant Physiol 161:157–164. https://doi.org/10.1078/0176-1617-01222
Tanton TW (1981) The Banjhi (Dormancy) Cycle in Tea (Camellia sinensis). Exp Agric 17:149–156. https://doi.org/10.1017/S001447970001139X
Thirugnanasambantham K, Prabu G, Palanisamy S et al (2013) Analysis of dormant bud (Banjhi) specific transcriptome of tea (Camellia sinensis (L.) O. Kuntze) from cDNA library revealed dormancy-related genes. Appl Biochem Biotechnol 169:1405–1417. https://doi.org/10.1007/s12010-012-0070-5
Thirugnanasambantham K, Prabu GR, Mandal AKA (2014) Isolation and characterization of cDNA encoding cyclophilin gene from dormant bud of Camellia sinensis (L.) O. Kuntze. 42:256–261
Vantard M, Cowling R, Delichère C (2000) Cell cycle regulation of the microtubular cytoskeleton. Plant Mol Biol 43:691–703
Warnes GR, Bolker B, Bonebakker L, Gentleman R, Andy Liaw WH, Lumley T, Maechler M, Magnusson A, Schwartz MV (2015) gplots: Various R programming tools for plotting data. R Package Version 3.0.1. http://cran.r-project.org/package=gplots. Accessed 21 Nov 2019
Wei C, Yang H, Wang S et al (2018) Draft genome sequence of Camellia sinensis var. sinensis provides insights into the evolution of the tea genome and tea quality. Proc Natl Acad Sci 115:E4151–E4158. https://doi.org/10.1073/pnas.1719622115
Ye J, Coulouris G, Zaretskaya I et al (2012) Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinform 13:134. https://doi.org/10.1186/1471-2105-13-134
Zhang K, Letham DS, John PC (1996) Cytokinin controls the cell cycle at mitosis by stimulating the tyrosine dephosphorylation and activation of p34cdc2-like H1 histone kinase. Planta 200:2–12
Acknowledgements
The authors are thankful to the Director, UPASI Tea Research Foundation for his encouragement and support during the course of study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thirugnanasambantham, K., Prabu, G. & Mandal, A.K.A. Synergistic effect of cytokinin and gibberellins stimulates release of dormancy in tea (Camellia sinensis (L.) O. Kuntze) bud. Physiol Mol Biol Plants 26, 1035–1045 (2020). https://doi.org/10.1007/s12298-020-00786-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-020-00786-2