Abstract
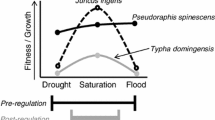
Wet meadows are a common focus of wetland restoration efforts, and the species within them often exist within a restricted range of water levels. Unfortunately, many restored wetlands have higher water levels and more open water than naturally occurring reference wetlands, and many are invaded and dominated by species of Typha. Most studies evaluating the optimal water level for plant species use observational methods, yet experimental methods are required to understand the breadth of a species’ niche. We used experimental transplants of Carex pellita, a common wet meadow sedge used in restoration in the interior of the USA, and Typha latifolia, a species of cattail which invades many restored wetlands, to test whether higher water levels in a restored wetland were prohibitive to the target sedge species. Physiological and growth measurements were collected on both species. We found that C. pellita grew as well or better when transplanted into the ponded water levels, while T. latifolia had reduced growth when transplanted into the relatively drier meadow conditions. Interestingly, C. pellita was able to adjust its Turgor Loss Point in response to changing water levels. Only recently the assumption of a constant Turgor Loss Point for each species has been questioned. Our results provide evidence that wet meadow species have a broader hydrologic niche than previously thought, and their ability to make physiological adjustments in response to changing water levels may allow them to thrive in areas with widely varying water levels.





Similar content being viewed by others
References
Armstrong W (1980) Aeration in Higher Plants. Adv Bot Res 7:225–332. https://doi.org/10.1016/S0065-2296(08)60089-0
Asamoah SA, Bork EW (2010) Drought tolerance thresholds in cattail (Typha latifolia): A test using controlled hydrologic treatments. Wetlands 30:99–110. https://doi.org/10.1007/s13157-009-0005-2
Bartlett MK, Scoffoni C, Sack L (2012) The determinants of leaf turgor loss point and prediction of drought tolerance of species and biomes: A global meta-analysis. Ecol Lett 15:393–405. https://doi.org/10.1111/j.1461-0248.2012.01751.x
Bartlett MK, Zhang Y, Kreidler N, Sun S, Ardy R, Cao K, Sack L (2014) Global analysis of plasticity in turgor loss point, a key drought tolerance trait. Ecol Lett 17:1580–1590. https://doi.org/10.1111/ele.12374
Bertness MD, Ellison AM (1987) Determinants of pattern in a New England salt marsh plant community. Ecol Monogr 57:129–147. https://doi.org/10.2307/1942621
Blom CWPM, Voesenek LACJ (1996) Flooding: the survival strategies of plants. Trends Ecol Evol 11:290–295. https://doi.org/10.1016/0169-5347(96)10034-3
Brinson MM, Malvárez AI (2002) Temperate freshwater wetlands: types, status, and threats. Environ Conserv 29:115–133. https://doi.org/10.1017/S0376892902000085
Duru M, Khaled RAH, Ducourtieux C, Theau JP, de Quadros FL, Cruz P (2009) Do plant functional types based on leaf dry matter content allow characterizing native grass species and grasslands for herbage growth pattern? Plant Ecol 201:421–433. https://doi.org/10.1007/s11258-008-9516-9
Dwire KA, Kauffman JB, Baham JE (2006) Plant species distribution in relation to water-table depth and soil redox potential in montane riparian meadows. Wetlands 26:131–146. https://doi.org/10.1672/0277-5212(2006)26[131:PSDIRT]2.0.CO;2
Eliceiri K, Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ : 25 years of image analysis HISTORICAL commentary NIH Image to ImageJ : 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Farrer ECEEC, Goldberg DE (2009) Litter drives ecosystem and plant community changes in cattail invasion. Ecol Appl 19:398–412. https://doi.org/10.1890/08-0485.1
Fennessy S, Mack J, Rokosch A, Knapp M, Micacchion M (2004) Integrated wetland assessment program: part 5: biogeochemical and hydrological investigations of natural and mitigation wetlands. Ohio EPA Tech. Rep. WET/2004–5. Ohio Environ. Prot. Agency, Wetl. Ecol. Group, Div. Surf. Water, Columbus Ohio
Fennessy SM, Cronk JK, Mitsch WJ (1994) Macrophyte productivity and community development in created freshwater wetlands under experimental hydrological conditions. Ecol Eng 3:469–484. https://doi.org/10.1016/0925-8574(94)00013-1
Findlay CS, Houlahan J (1997) Anthropogenic correlates of species richness in southeastern ontario wetlands. Correlativos Antropogenicos de la Riqueza de Especies en Humedales del Sureste de Ontario. Conserv Biol 11:1000–1009. https://doi.org/10.1046/j.1523-1739.1997.96144.x
Flora of North America Editorial Committee E (Ed.) (1993) Flora of North America North of Mexico. New York and Oxford
García-Baquero G, Silvertown J, Gowing DJ, Valle CJ (2016) Dissecting the hydrological niche: Soil moisture, space and lifespan. J Veg Sci 27:219–226. https://doi.org/10.1111/jvs.12353
Grace JB, Wetzel RG (1981) Habitat partitioning and competitive displacement in cattails: experimental field studies. Am Nat 118:463–474
Griffin-Nolan RJ, Ocheltree TW, Mueller KE, Blumenthal DM, Kray JA, Knapp AK (2019) Extending the osmometer method for assessing drought tolerance in herbaceous species. Oecologia 189:353–363. https://doi.org/10.1007/s00442-019-04336-w
Hirzel AH, Hausser J, Chessel D, Perrin N (2002) Ecological-niche factor analysis: how to compute habitat-suitability maps without absence data? Ecology 83:2027–2036. https://doi.org/10.1890/0012-9658(2002)083[2027:ENFAHT]2.0.CO;2
Hubbell S (2005) Neutral theory in community ecology and the hypothesis of functional equivalence. Funct Ecol 19:166–172. https://doi.org/10.1111/j.0269-8463.2005.00965.x
Hudson H, Wright E, Atkinson RB, Perry JE (2014) Assessment of woody vegetation for replacement of ecological functions in created forested wetlands of the piedmont province of Virginia. Piedmont Wetl. Res, Progr
Jung V, Hoffmann L, Muller S (2009) Ecophysiological responses of nine floodplain meadow species to changing hydrological conditions. Plant Ecol 201:589–598. https://doi.org/10.1007/s11258-008-9508-9
Kercher SM, Zedler JB (2004) Flood tolerance in wetland angiosperms: A comparison of invasive and noninvasive species. Aquat Bot 80:89–102. https://doi.org/10.1016/j.aquabot.2004.08.003
Konar M, Jason Todd M, Muneepeerakul R, Rinaldo A, Rodriguez-Iturbe I (2013) Hydrology as a driver of biodiversity: Controls on carrying capacity, niche formation, and dispersal. Adv Water Resour 51:317–325. https://doi.org/10.1016/j.advwatres.2012.02.009
Kotowski W, Thorig W, Van Diggelen R, Wassen MJ (2006) Competition as a factor structuring species zonation in riparian fens - a transplantation experiment. Appl Veg Sci 9:231–240. https://doi.org/10.1111/j.1654-109X.2006.tb00672.x
Lea H, Ülo N, Ian JW (2009) Are species shade and drought tolerance reflected in leaf-level structural and functional differentiation in Northern Hemisphere temperate woody flora? New Phytol 184:257–274
Lhotsky B, Kovács B, Ónodi G, Csecserits A, Rédei T, Lengyel A, Kertész M, Botta-Dukát Z (2016) Changes in assembly rules along a stress gradient from open dry grasslands to wetlands. J Ecol 104:507–517. https://doi.org/10.1111/1365-2745.12532
Lovell JT, Menges E (2013) Dominant species responses to drought in seasonal wetlands: evidence from reciprocal transplants across a moisture gradient 1. J Torrey Bot Soc 140:157–169. https://doi.org/10.3159/TORREY-D-12-00055.1
Meinzer FC, Woodruff DR, Marias DE, Mcculloh KA, Sevanto S (2014) Dynamics of leaf water relations components in co-occurring iso- and anisohydric conifer species. Plant Cell Environ 37:2577–2586. https://doi.org/10.1111/pce.12327
Pavlik BM (1984) Seasonal changes of osmotic pressure, symplasmic water content and tissue elasticity in the blades of dune grasses growing in situ along the coast of Oregon. Plant Cell Environ 7:531–539. https://doi.org/10.1111/1365-3040.ep11616220
Pezeshki SR (2001) Wetland plant responses to soil flooding. Environ Exp Bot 46:299–312. https://doi.org/10.1016/S0098-8472(01)00107-1
R Core Team (2016) R: A language and environment for statistical computing
Sala OE, Chapin FS, Armesto JJ, Berlow EL, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774. https://doi.org/10.1126/science.287.5459.1770
Sanderson JS, Kotliar NB, Steingraeber DA (2008) Opposing environmental gradients govern vegetation zonation in an intermountain playa. Wetlands 28:1060–1070. https://doi.org/10.1672/07-111.1
Silvertown J, Araya Y, Gowing D (2015) Hydrological niches in terrestrial plant communities: a review. J Ecol 103:93–108. https://doi.org/10.1111/1365-2745.12332
Snow AA, Vince SW (1984) Plant zonation in an alaskan salt marsh: ii. an experimental study of the role of edaphic conditions. J Ecol 72:669–684. https://doi.org/10.2307/2260075
Toogood SE, Joyce CB, Waite S (2008) Response of floodplain grassland plant communities to altered water regimes. Plant Ecol 197:285–298. https://doi.org/10.1007/s11258-007-9378-6
Touchette BW, Iannacone LR, Turner GE, Frank AR (2007) Drought tolerance versus drought avoidance: A comparison of plant-water relations in herbaceous wetland plants subjected to water withdrawal and repletion. Wetlands 27:656–667. https://doi.org/10.1672/0277-5212(2007)27[656:DTVDAA]2.0.CO;2
Tuchman NNC, Larkin DJD, Geddes P, Wildova R, Jankowski K, Goldberg DE (2009) Patterns of environmental change associated withTypha xglauca invasion in a Great Lakes coastal wetland. Wetlands 29:964–975. https://doi.org/10.1672/08-71.1
Tyree MT, Hammel HT (1972) The measurement of the turgor pressure and the water relations of plants by the pressure-bomb technique. J Exp Bot 23:267–282. https://doi.org/10.1093/jxb/23.1.267
Wetzel PR, Kitchens WM, Brush JM, Dusek ML (2004) Use of a reciprocal transplant study to measure the rate of plant community change in a tidal marsh along a salinity gradient. Wetlands 24:879–890. https://doi.org/10.1672/0277-5212(2004)024[0879:UOARTS]2.0.CO;2
Zedler J (2000) Progress in wetland restoration ecology. Trends Ecol Evol 15:402–407
Acknowledgements
The authors would like to thank the City of Fort Collins Natural Areas for allowing this research to take place on their property. Many thanks to Lydia Baldwin for her help establishing the experimental transplant plots. Additional thanks to Mark Paschke, John Sanderson, and Monique Rocca for comments on earlier versions of this manuscript. The authors have no conflict of interest to report.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Neal J. Enright.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sueltenfuss, J.P., Ocheltree, T.W. & Cooper, D.J. Evaluating the realized niche and plant–water relations of wetland species using experimental transplants. Plant Ecol 221, 333–345 (2020). https://doi.org/10.1007/s11258-020-01015-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-020-01015-2