Abstract
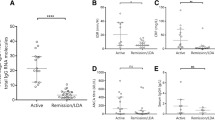
Background: Antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis is an autoimmune disorder of unknown etiology with dysregulated cytokines levels. Objectives: The main aim of this study was to assess the clinical correlation between antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, granulomatosis with polyangiitis (GPA) serum levels of the microscopic polyangiitis (MPA), serum levels of the proinflammatory cytokines, interleukin (IL)-32 and interleukin-6. Methods: Study included 71 patients, 47 with GPA and 24 with MPA. Serum IL-32 and IL-6 concentrations were analyzed in all patients, and compared with levels observed in 10 controls. IL-32 and IL-6 were evaluated using DuoSet and Quantikine HS ELISA, respectively. IL-32 and IL-6 concentrations were correlated with disease-related clinical and laboratory findings. Results: IL-32 and IL-6 levels were significantly higher in GPA and MPA than in controls, especially IL-32 levels in GPA were elevated. IL-32 concentrations correlated positively with anti-proteinase 3 - ANCA (PR3-ANCA) levels in GPA (P < 0.0001), and with anti-myeloperoxidase ANCA (MPO-ANCA) in MPA(P = 0.049). IL-32 levels correlated positively with disease activity inGPA and MPA(P < 0.0001). GPA patients with pulmonary, cutaneous, and musculoskeletal involvement presented the highest IL-6 serum levels. Cutaneous manifestations correlated positively with IL-6 levels in MPA patients (P = 0.05). ANCA-positive patients with GPA expressed significantly high IL-6 levels (P = 0.036). No significant difference in IL-32 values was observed between ANCA-positiveandANCA-negativepatients. Conclusions: Patientswith GPAandMPApresenthigher serumIL-32 and IL-6 levels than controls. IL-32 levels correlate positively with disease activity.
Similar content being viewed by others
References
Gomez-Puerta JA, Bosch X. Anti-neutrophil cytoplasmic antibody pathogenesis in small-vessel vasculitis: an update. Am J Pathol 2009; 2009: 1790–8.
Kallenberg CG. Key advances in the clinical approach to ANCA-associated vasculitis. Nat Rev Rheumatol 2014; 2014: 484–93.
Cornec D, Cornec-Le Gall E, Fervenza FC, Specks U. ANCA-associated vasculitis - clinical utility of using ANCA specificity to classify patients. Nat Rev Rheumatol 2016; 2016: 570–9.
Tadema H, Heeringa P, Kallenberg CG. Bacterial infections in Wegener’s granulomatosis: mechanisms potentially involved in autoimmune pathogenesis. Curr Opin Rheumatol 2011; 2011: 366–71.
Wilde B, Thewissen M, Damoiseaux J, van Paassen P, Witzke O, Tervaert JW. T cells in ANCA-associated vasculitis: what can we learn from lesional versus circulating T cells? Arthritis Res Ther 2010; 121: 204.
Chen M, Kallenberg CG. ANCA-associated vasculitides - advances in pathogenesis and treatment. Nat Rev Rheumatol 2010; 2010: 653–64.
Al-Hussain T, Hussein MH, Conca W, Al Mana H, Akhtar M. Pathophysiology of ANCA-associated Vasculitis. Adv Anat Pathol 2017; 2017: 226–34.
Abdulahad WH, Stegeman CA, Kallenberg CG. Review article: The role of CD4(+) T cells in ANCA-associated systemic vasculitis. Nephrology (Carlton) 2009; 2009: 26–32.
Kallenberg CG. Advances in pathogenesis and treatment of ANCA-associated vasculitis. Discov Med 2014; 2014: 195–201.
Bae S, Kim YG, Choi J, et al. Elevated interleukin-32 expression in granulomatosis with polyangiitis. Rheumatology (Oxford) 2012; 2012: 1979–88.
Novick D, Rubinstein M, Azam T, Rabinkov A, Dinarello CA, Kim SH. Proteinase 3 is an IL-32 binding protein. Proc Natl Acad Sci USA 2006; 2006: 3316–21.
Csernok E, Holle JU, Gross WL. Proteinase 3, protease-activated receptor-2 and interleukin-32: linking innate and autoimmunity in Wegener’s granulomatosis. Clin Exp Rheumatol 2008; 261: S112–7.
Khawar B, Abbasi MH, Sheikh N. A panoramic spectrum of complex interplay between the immune system and IL-32 during pathogenesis of various systemic infections and inflammation. Eur J Med Res 2015; 201: 7.
Dinarello CA, Kim SH. IL-32, a novel cytokine with a possible role in disease. Ann Rheum Dis 2006; 65(Suppl. 31): iii61–4.
Kim SH, Han SY, Azam T, Yoon DY, Dinarello CA. Interleukin-32: a cytokine and inducer of TNFalpha. Immunity 2005; 2005: 131–42.
Netea MG, Azam T, Ferwerda G, et al. IL-32 synergizes with nucleotide oligomerization domain (NOD) 1 and NOD2 ligands for IL-1beta and IL-6 production through a caspase 1-dependent mechanism. Proc Natl Acad Sci USA 2005; 2005: 16309–14.
Netea MG, Lewis EC, Azam T, et al. Interleukin-32 induces the differentiation of monocytes into macrophage-like cells. Proc Natl Acad Sci USA 2008; 2008: 3515–20.
Shimizu M, Sekiguchi T, Kishi N, et al. A case of a 6-year-old girl with anti-neutrophil cytoplasmic autoantibody-negative pauci-immune crescentic glomerulonephritis. Clin Exp Nephrol 2011; 2011: 596–601.
Ohlsson S, Wieslander J, Segelmark M. Circulating cytokine profile in anti-neutrophilic cytoplasmatic autoantibody-associ-ated vasculitis: prediction of outcome? Mediators Inflamm 2004; 2004: 275–83.
Berti A, Cavalli G, Campochiaro C, et al. Interleukin-6 in ANCA-associated vasculitis: Rationale for successful treatment with tocilizumab. Semin Arthritis Rheum 2015; 2015: 48–54.
Ohta N, Fukase S, Aoyagi M. Serum levels of soluble adhesion molecules ICAM-1, VCAM-1 and E-selectin in patients with Wegener’s granulomatosis. Auris Nasus Larynx 2001; 2001: 311–4.
Muller Kobold AC, Kallenberg CG, Tervaert JW. Monocyte activation in patients with Wegener’s granulomatosis. Ann Rheum Dis 1999; 1999: 237–45.
Booth AD, Wallace S, McEniery CM, et al. Inflammation and arterial stiffness in systemic vasculitis: a model of vascular inflammation. Arthritis Rheum 2004; 2004: 581–8.
Berti A, Warner R, Johnson K, et al. Brief report: Circulating cytokine profiles and antineutrophil cytoplasmic antibody specificity in patients with antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheumatol 2018; 2018: 1114–21.
Leavitt RY, Fauci AS, Bloch DA, et al. The American College of Rheumatology 1990 criteria for the classification of Wegener’s granulomatosis. Arthritis Rheum 1990; 1990: 1101–7.
Khan I, Watts RA. Classification of ANCA-associated vasculitis. Curr Rheumatol Rep 2013; 151: 383.
Mukhtyar C, Lee R, Brown D, et al. Modification and validation of the Birmingham Vasculitis Activity Score (version 3). Ann Rheum Dis 2009; 2009: 1827–32.
Hellmich B, Flossmann O, Gross WL, et al. EULAR recommendations for conducting clinical studies and/or clinical trials in systemic vasculitis: focus on anti-neutrophil cytoplasm antibody-associated vasculitis. Ann Rheum Dis 2007; 2007: 605–17.
Su H, Lei CT, Zhang C. Interleukin-6 signaling pathway and its role in kidney disease: an update. Front Immunol 2017; 81: 405.
Acknowledgements
The study was supported by grant from Medical University in Wrocław, grant number: STM. C250.16.025.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure. The authors declare they have no conflicts of interests
About this article
Cite this article
Krajewska (Wojciechowska), J., Koṡcielska-Kasprzak, K., Krajewski, W. et al. Serum levels of interleukin-32 and interleukin-6 in granulomatosis with polyangiitis and microscopic polyangiitis: association with clinical and biochemical findings. Eur Cytokine Netw 30, 151–159 (2019). https://doi.org/10.1684/ecn.2019.0439
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ecn.2019.0439