Abstract
Optimization of tissue culture systems able to complete male germ cell maturation to post-meiotic stages is considered as an important matter in reproductive biology. Considering that hypoxia is one of the factors limiting the efficiency of organ culture, the aim of this study was to use isolated seminiferous tubules (STs), having more surface and less thickness, in an organotypic culture system in order to improve oxygen diffusion and reduce hypoxia. The mechanically separated STs embedded in agarose or alginate and 1–3-mm3 testicular tissue fragments of 3 adult mice were separately placed on the flat surface of agarose gel that was half-soaked in the medium. Survival and differentiation of germ cells using PLZF and SCP3 markers, identity of Sertoli cell using GATA4, cell proliferation with the Ki67 marker, and ST integrity using a ST scoring were evaluated up to 36 d at different culture times, each corresponding to the duration of one spermatogenic cycle. We observed a significantly reduced ST integrity in STs embedded in agarose or alginate on day 9 (versus tissue fragments p ≤ 0.05). There was no difference in the number of PLZF-positive cells between groups, but the number of SCP3 (in all-time points) and GATA4-positive cells was significantly higher in the culture of embedded STs. Although embedding STs can be useful for the progress of in vitro spermatogenesis, it makes them sensitive to degeneration. Further improvements are required to modify the air-liquid interface method to maintain ST integrity.






Similar content being viewed by others
References
Aliakbari F, Gilani MAS, Amidi F, Baazm M, Korouji M, Izadyar F, Yazdekhasti H, Abbasi M (2016) Improving the efficacy of cryopreservation of spermatogonia stem cells by antioxidant supplements. Cell Rep 18(2):87–95
Beachey W. Respiratory care anatomy and physiology: foundations for clinical practice: Elsevier health sciences; 2018
Davis J (1967) The structure of guinea-pig lung maintained in organ culture. Br J Exp Pathol 48(4):371–378
De Michele F, Poels J, Weerens L, Petit C, Evrard Z, Ambroise J et al (2016) Preserved seminiferous tubule integrity with spermatogonial survival and induction of Sertoli and Leydig cell maturation after long-term organotypic culture of prepubertal human testicular tissue. Hum Reprod 32(1):32–45
Del Vento F, Vermeulen M, de Michele F, Giudice M, Poels J, des Rieux A et al (2018) Tissue engineering to improve immature testicular tissue and cell transplantation outcomes: one step closer to fertility restoration for prepubertal boys exposed to gonadotoxic treatments. Int J Mol Sci 19(1):286
Galdon G, Atala A, Sadri-Ardekani H (2016) In vitro spermatogenesis: how far from clinical application? Curr Urol Rep 17(7):49
Gholami K, Pourmand G, Koruji M, Ashouri S, Abbasi M (2018b) Organ culture of seminiferous tubules using a modified soft agar culture system. Stem Cell Res Ther 9(1):249
Gholami K, Pourmand G, Koruji M, Sadighigilani M, Navid S, Izadyar F, Abbasi M (2018a) Efficiency of colony formation and differentiation of human spermatogenic cells in two different culture systems. Reprod Biol 18(4):397–403
Giudice MG, De Michele F, Poels J, Vermeulen M, Wyns C (2017) Update on fertility restoration from prepubertal spermatogonial stem cells: how far are we from clinical practice? Stem Cell Res 21:171–177
Howlader N, Noone A, Krapcho M, Garshell J, Neyman N, Altekruse S, et al. SEER Cancer Statistics Review. National Cancer Institute; Bethesda, MD: 1975-2010
Klöckner W, Büchs J (2012) Advances in shaking technologies. Trends Biotechnol 30(6):307–314
Komeya M, Kimura H, Nakamura H, Yokonishi T, Sato T, Kojima K et al (2016) Long-term ex vivo maintenance of testis tissues producing fertile sperm in a microfluidic device. Sci Rep 6:21472
Kraemer WJ, Fleck SJ, Deschenes MR. Exercise physiology: integrating theory and application: Lippincott Williams & Wilkins; 2011
Lee K, Silva EA, Mooney D (2011) Growth factor delivery-based tissue engineering: general approaches and a review of recent developments. J R Soc Interface 8(55):153–170
McMurtrey RJ (2016) Analytic models of oxygen and nutrient diffusion, metabolism dynamics, and architecture optimization in three-dimensional tissue constructs with applications and insights in cerebral organoids. Tissue Engineering Part C: Methods 22(3):221–249
Mitchell AC, Briquez PS, Hubbell JA, Cochran JR (2016) Engineering growth factors for regenerative medicine applications. Acta Biomater 30:1–12
Nakamura N, Merry GE, Inselman AL, Sloper DT, Del Valle PL, Sato T et al (2017) Evaluation of culture time and media in an in vitro testis organ culture system. Birth Defects Res 109(7):465–474
Navid S, Abbasi M, Hoshino Y (2017a) The effects of melatonin on colonization of neonate spermatogonial mouse stem cells in a three-dimensional soft agar culture system. Stem Cell Res Ther 8(1):233
Navid S, Rastegar T, Baazm M, Alizadeh R, Talebi A, Gholami K, Khosravi-Farsani S, Koruji M, Abbasi M (2017b) In vitro effects of melatonin on colonization of neonate mouse spermatogonial. Syst Biol Reprod Med 63(6):370–381
Poels J, Abou-Ghannam G, Decamps A, Leyman M, des Rieux A, Wyns C (2016a) Transplantation of testicular tissue in alginate hydrogel loaded with VEGF nanoparticles improves spermatogonial recovery. J Control Release 234:79–89
Poels J, Abou-Ghannam G, Decamps A, Leyman M, des Rieux A, Wyns CJ (2016b) Transplantation of testicular tissue in alginate hydrogel loaded with VEGF nanoparticles improves spermatogonial recovery. J Control Release 234:79–89
Poels J, Abou-Ghannam G, Herman S, Van Langendonckt A, Wese F-X, Wyns C (2014) In search of better spermatogonial preservation by supplementation of cryopreserved human immature testicular tissue xenografts with N-acetylcysteine and testosterone. Front Surg 1:47
Reakasame S, Boccaccini AR (2018) Oxidized alginate-based hydrogels for tissue engineering applications: a review. Biomacromolecules 19(1):3–21
Sato T, Katagiri K, Gohbara A, Inoue K, Ogonuki N, Ogura A et al (2011a) In vitro production of functional sperm in cultured neonatal mouse testes. Nature 471(7339):504
Sato T, Katagiri K, Gohbara A, Inoue K, Ogonuki N, Ogura A, Kubota Y, Ogawa T (2011b) In vitro production of functional sperm in cultured neonatal mouse testes. Nature. 471(7339):504–507
Sato T, Katagiri K, Kojima K, Komeya M, Yao M, Ogawa T (2015) In vitro spermatogenesis in explanted adult mouse testis tissues. PLoS One 10(6):e0130171
Schmidt JA, de Avila JM, McLean DJ (2006) Effect of vascular endothelial growth factor and testis tissue culture on spermatogenesis in bovine ectopic testis tissue xenografts. Biol Reprod 75(2):167–175
Summerlin WT, Charlton E, Karasek M (1970) Transplantation of organ cultures of adult human skin. J Invest Dermatol 55(5):310–316
Trowell O (1959) The culture of mature organs in a synthetic medium. Exp Cell Res 16(1):118–147
Turkmen S, Mentese A, Karaguzel E, Karaca Y, Kucuk A, Uzun A et al (2012) A comparison of the effects of N-acetylcysteine and ethyl pyruvate on experimental testicular ischemia-reperfusion injury. Fertil Steril 98(3):626–631
Van Eyck A-S, Jordan BF, Gallez B, Heilier J-F, Van Langendonckt A, Donnez J (2009) Electron paramagnetic resonance as a tool to evaluate human ovarian tissue reoxygenation after xenografting. Fertil Steril 92(1):374–381
Van Saen D, Goossens E, Haentjens P, Baert Y, Tournaye H (2013) Exogenous administration of recombinant human FSH does not improve germ cell survival in human prepubertal xenografts. Reprod BioMed Online 26(3):286–298
Wyns C, Collienne C, Shenfield F, Robert A, Laurent P, Roegiers L, Brichard B (2015) Fertility preservation in the male pediatric population: factors influencing the decision of parents and children. Hum Reprod 30(9):2022–2030
Wyns C, Curaba M, Vanabelle B, Van Langendonckt A, Donnez J (2010) Options for fertility preservation in prepubertal boys. Hum Reprod Update 16(3):312–328
Yokonishi T, Sato T, Katagiri K, Ogawa T (2013) In vitro spermatogenesis using an organ culture technique. Methods Mol Biol 927:479–488
Acknowledgments
The authors thank all the staff members of Institut de Recherche Expérimentale et Clinique who were involved in this project.
Funding
This study was supported by the Université Catholique de Louvain and funds from the Salus Sanguinis foundation and Saint-Luc foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors alone are responsible for the content and writing of this article.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Editor: Tetsuji Okamoto
Electronic supplementary material
FIG S1
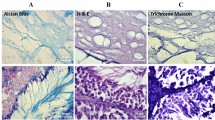
Histological staining at different magnifications showing seminiferous tubules integrity of STs embedded in alginate or agarose and within tissue fragments after 27 days of culture. The central and marginal areas are magnified in the right and left insets respectively showing intraepithelial vacuoles (orange arrow) of varying sizes in STs in marginal areas and a relatively good integrity in central areas. STs with poor score (score 3) are located more in marginal areas and peripheral degeneration was observed in all groups. (PNG 9782 kb)
FIG S2
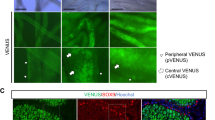
A) the pattern of PLZF expression in central (right) and marginal areas (left) is shown. The more number of PLZF-positive cells was observed in marginal areas. A) Quantification of tubules positive for PLZF staining in well-preserved tubules located in central areas. Scale bars: 500 μm (Alginate and tissue in the middle), 1 mm (Agarose in the middle), 50 μm (all groups, left and right). (PNG 5678 kb)
FIG S3
A) Expression of GATA4 is more limited to marginal and submarginal areas in tissue culture but it was observed in all areas in STs embedded in alginate or agarose. B) Number of GATA4 - positive cells in centrally-located tubules at different culture times. Scale bars: 500 μm (Alginate and tissue in the middle), 1 mm (Agarose in the middle), 50 μm (all groups, left and right), ⁎: p ≤ 0.0181, ⁎ ⁎: p ≤ 0.0050. (PNG 5009 kb)
FIG S4
A) SCP3 expression is more seen in sub marginal/marginal areas of tissue fragments but distribution of SCP3 is almost observed in all parts in the mechanically-separated STs embedded in alginate or agarose. The expression of SCP3 partly disappeared in the central areas of tissue fragment. B) Quantification of SCP3 expression by counting the number of SCP3-positive cells per well-preserved ST only located in central areas in all groups. Scale bars: 500 μm (all groups in the middle), 50 μm (all groups, left and right), ⁎: p ≤ 0.0325, ⁎ ⁎: p ≤ 0.0019. (PNG 5359 kb)
FIG S5
A) Cell proliferation was observed in the different groups. The expression pattern of Ki67 in groups is shown in marginal / sub marginal (on the left) and central areas (on the right). Quantification of Ki67 expression by counting the number of Ki67-positive cells per well-preserved ST only located in central areas in all groups. Scale bars: 500 μm (all groups in the middle), 50 μm (all groups, left and right). (PNG 5032 kb)
Rights and permissions
About this article
Cite this article
Gholami, K., Vermeulen, M., Del Vento, F. et al. The air-liquid interface culture of the mechanically isolated seminiferous tubules embedded in agarose or alginate improves in vitro spermatogenesis at the expense of attenuating their integrity. In Vitro Cell.Dev.Biol.-Animal 56, 261–270 (2020). https://doi.org/10.1007/s11626-020-00437-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-020-00437-6