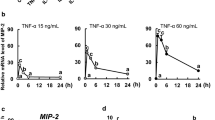
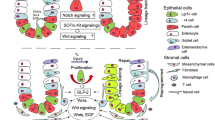
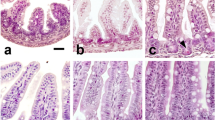
Abstract
Interleukin (IL)-4 is known as a cytokine mainly involved in allergy and inflammation, but recent studies have suggested that IL-4 plays a part in the differentiation process of various cells. Since the effect of IL-4 on intestinal epithelial cells, particularly cryptic cells including stem cells, is poorly understood, we investigated IL-4-induced changes in intestinal epithelial cells using mouse jejunal organoids called enteroids. IL-4 treatment decreased cell proliferation, the expression of the stem cell markers leucine-rich repeat-containing G protein-coupled receptor 5 (Lgr5) and olfactomedin 4 (Olfm4), and Lgr5-positive cells in enteroids. Among the differentiation markers, IL-4 significantly decreased the gene expression levels of the Paneth cell markers lysozyme 1 (Lyz1) and regenerating islet-derived protein 3 gamma (Reg3γ). A fluorescent immunostaining showed that IL-4 attenuated the emission and fluorescence intensity derived from lysozyme, which is enriched in Paneth cells. These results suggest that functional changes in Paneth cells caused by IL-4 may contribute to the reduction in Lgr5-positive cells and proliferative activity. IL-4 may affects gut function by altering the proliferation and the gene expression in enteroids.



Similar content being viewed by others
References
Andrews C, McLean MH, Durum SK (2018) Cytokine tuning of intestinal epithelial function. Front Immunol 9:1–15. https://doi.org/10.3389/fimmu.2018.01270
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, Clevers H (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449:1003–1007. https://doi.org/10.1038/nature06196
Bartuzi Z, Zbikowska-Gotz M, Romański B, Sinkiewicz W (2000) Evaluating the profile of selected cytokines in patients with food allergy and chronic gastritis. Med Sci Monit 6:1128–1135
Demitrack ES, Samuelson LC (2016) Notch regulation of gastrointestinal stem cells. J Physiol 594:4791–4803. https://doi.org/10.1113/JP271667
Fujii M, Matano M, Toshimitsu K, Takano A, Mikami Y, Nishikori S, Sugimoto S, Sato T (2018) Human intestinal organoids maintain self-renewal capacity and cellular diversity in niche-inspired culture condition. Cell Stem Cell 23:787–793. https://doi.org/10.1016/j.stem.2018.11.016
Gerbe F, Sidot E, Smyth DJ, Ohmoto M, Matsumoto I, Dardalhon V, Cesses P, Garnier L, Pouzolles M, Brulin B, Bruschi M, Harcus Y, Zimmermann VS, Taylor N, Maizels RM, Jay P (2016) Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 529:226–230. https://doi.org/10.1038/nature16527
Haber AL, Biton M, Rogel N, Herbst RH, Shekhar K, Smillie C, Burgin G, Delorey TM, Howitt MR, Katz Y, Tirosh I, Beyaz S, Dionne D, Zhang M, Raychowdhury R, Garrett WS, Ronzenblatt-Rosen O, Shi HN, Yilmaz O, Xavier RJ, Regev A (2017) A single-cell survey of the small intestinal epithelium. Nature 551:333–339. https://doi.org/10.1038/nature24489
Han Z, Gu YY, Cong N, Ma R, Chi FL (2018) Celastrol enhances Atoh1 expression in inner ear stem cells and promotes their differentiation into functional auditory neuronal-like cells. Organogenesis 14:82–93. https://doi.org/10.1080/15476278.2018.1462433
Howitt MR, Lavoie S, Michaud M, Blum AM, Tran SV, Weinstock JV, Gallini CA, Redding K, Margolskee RF, Osborne LC, Artis D, Garrett WS (2016) Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351:1329–1333. https://doi.org/10.1126/science.aaf1648
Junttila IS (2018) Tuning the cytokine responses: an update on interleukin (IL)-4 and IL-13 receptor complexes. Front Immunol 9:1–6. https://doi.org/10.3389/fimmu.2018.00888
Kay SK, Harrington HA, Shepherd S, Brennan K, Dale T, Osborne JM, Gavaghan DJ, Byrne HM (2017) The role of the Hes1 crosstalk hub in Notch-Wnt interactions of the intestinal crypt. PLoS Comput Biol 13:1–28. https://doi.org/10.1371/journal.pcbi.1005400
Ke MT, Fujimoto S, Imai T (2013) SeeDB: a simple and morphology-preserving optical clearing agent for neuronal circuit reconstruction. Nat Neurosci 16:1154–1161. https://doi.org/10.1038/nn.3447
Kordulewska NK, Cieślińska A, Fiedorowicz E, Jarmolowska B, Piskorz-Ogórek K, Kostyra E (2018) Cytokines concentrations in serum samples from allergic children-multiple analysis to define biomarkers for better diagnosis of allergic inflammatory process. Immunobiology 223:648–657. https://doi.org/10.1016/j.imbio.2018.07.010
Lechuga S, Ivanov AI (2017) Disruption of the epithelial barrier during intestinal inflammation: quest for new molecules and mechanisms. Biochim Biophys Acta Mol Cell Res 1864:1183–1194. https://doi.org/10.1016/j.bbamcr.2017.03.007
Mochizuki T, Satsu H, Totsuka M, Shimizu M (2009) Transepithelial transport of macromolecular substances in IL-4 treated human intestinal T84 cell monolayers. Biosci Biotechnol Biochem 73:2422–2426. https://doi.org/10.1271/bbb.90383
Nakamura T, Tsuchiya K, Watanabe M (2007) Crosstalk between Wnt and Notch signaling in intestinal epithelial cell fate decision. J Gastroenterol 42:705–710. https://doi.org/10.1007/s00535-007-2087-z
Possidonio ACB, Senna ML, Portilho DM, Soares CP, Sampaio LS, Eincker-Lamas M, Branco MTLC, Costa ML, Mermelstein C (2011) α-cyclodextrin enhances myoblast fusion and muscle differentiation by the release of IL-4. Cytokine 55:280–287. https://doi.org/10.1016/j.cyto.2011.04.018
Rothenberg ME, Mishra A, Brandt EB, Hogan SP (2001) Gastrointestinal eosinophils. Immunol Rev 179:139–155. https://doi.org/10.1034/j.1600-065X.2001.790114.x
Saggini A, Maccauro G, Tripodi D, De Lutils MA, Conti F, Felaco P, Fulcheri M, Galzio R, Caraffa A, Antinolfi P, Felaco M, Pandolfi F, Sabatino G, Neri G, Shaik-Dasthagirisaheb YB (2011) Allergic inflammation: role of cytokines with special emphasis on IL-4. Int J Immunopathol Pharmacol 24:305–311. https://doi.org/10.1177/039463201102400204
Saito Y, Iwatsuki K, Hanyu H, Maruyama N, Aihara E, Tadaishi M, Shimizu M, Kobayashi-Hattori K (2017) Effect of essential amino acids on enteroids: methionine deprivation suppresses proliferation and affects differentiation in enteroid stem cells. Biochem Biophys Res Commun 488:171–176. https://doi.org/10.1016/j.bbrc.2017.05.029
Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ, Clevers H (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265. https://doi.org/10.1038/nature07935
Sato T, van Es JH, Snippert HJ, Stange DE, Vries RG, van den Born M, Barker N, Shroyer NF, van de Wetering M, Clevers H (2011) Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469:415–418. https://doi.org/10.1038/nature09637
Schmidt-Weber CB (2012) Anti-IL-4 as a new strategy in allergy. Chem Immunol Allergy 96:120–125. https://doi.org/10.1159/000332235
Shimizu M (2010) Interaction between food substances and the intestinal epithelium. Biosci Biotechnol Biochem 74:232–241. https://doi.org/10.1271/bbb.90730
van Es JH, Sato T, van de Wetering M, Lyubimova A, Gregorieff A, Zeinstra L, van den Born M, Korving J, Martens ACM, van den Oudenaarden A, Clevers H (2012) Dll1 marks early secretory progenitors in gut crypts that can revert to stem cells upon tissue damage. Nat Cell Biol 14:1099–1104. https://doi.org/10.1038/ncb2581
van Neerven SM, Vermeulen L (2019) The interplay between intrinsic and extrinsic Wnt signaling in controlling intestinal transformation. Differentiation 108:17–23. https://doi.org/10.1016/j.diff.2019.02.002
Vila MF, Trudeau MP, Hung YT, Zeng Z, Urriola PE, Shurson GC, Saqui-Salces M (2018) Dietary fiber sources and non-starch polysaccharide-degrading enzymes modify mucin expression and the immune profile of the swine ileum. PLoS ONE 13:1–16. https://doi.org/10.1371/journal.pone.0207196
von Moltke J, Ji M, Liang HE, Locksley RM (2016) Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 529:221–225. https://doi.org/10.1038/nature16161
Yu LCH, Glynn AN, Turner JR, Buret AG (2005) SGLT-1-mediated glucose uptake protects intestinal epithelial cells against LPS-induced apoptosis and barrier defects: a novel cellular rescue mechanism? FASEB J 19:1822–1835. https://doi.org/10.1096/fj.05-4226com
Zhang Q, Zhu W, Xu F, Dai X, Shi L, Cai W, Mu H, Hitchens TK, Foley LM, Liu X, Yu F, Chen J, Shi Y, Leak RK, Gao Y, Chen J, Hu X (2019) The interleukin-4/PPARγ signaling axis promotes oligodendrocyte differentiation and remyelination after brain injury. PLoS Biol 17:1–33. https://doi.org/10.1371/journal.pbio.3000330
Acknowledgements
We express our gratitude to Dr. Hans Clevers (Hubrecht Institute) for the kind gift of the Noggin-secreting cell line, Dr. Eitaro Aihara (Cincinnati University) for kindly donating the R-spondin-secreting cell line, and Dr. Tokiyoshi Ayabe (Hokkaido University) for kind gift of anti-lysozyme antibody.
Funding
This work was supported by Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for JSPS fellows Grant Number JP18J22466.
Author information
Authors and Affiliations
Contributions
YS, KI, MT, Makoto Shimizu, and KK designed the experiments. YS, Mika Sato, and AI performed the experiments. YS, KI, Makoto Shimizu, and KK wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saito, Y., Iwatsuki, K., Inaba, A. et al. Interleukin-4 suppresses the proliferation and alters the gene expression in enteroids. Cytotechnology 72, 479–488 (2020). https://doi.org/10.1007/s10616-020-00395-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-020-00395-7