Abstract
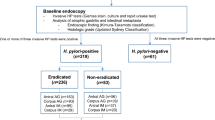
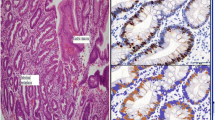
Pyloric metaplasia (PM) and pseudopyloric metaplasia (PPM) are metaplastic changes resulting in pyloric-type glands in the gastric oxyntic mucosa that mainly occur in chronic gastritis caused by Helicobacter pylori (H. pylori) infection. Focusing on PM and PPM, we classified the histological changes in gastric mucosa according to the Updated Sydney System, using 314 biopsy specimens of gastric greater curvature of the middle body before H. pylori eradication (HPE). Next, the numbers of PM and PPM glands were counted in 47 specimens, and subjects were followed up over 10 years after HPE. PPM was recognized jointly with inflammation, activity, atrophy, and intestinal metaplasia, but PM was recognized more frequently than PPM as atrophy and intestinal metaplasia progressed. Both PM and PPM regressed significantly within 6 years after HPE. Additionally, we demonstrated that PM and PPM are not always coincident with spasmolytic polypeptide-expressing metaplasia (SPEM). In conclusion, PM and PPM are considered different modulations of the same line of differentiation, which are both reversible, with PM potentially emerging from PPM upon progression.




Similar content being viewed by others
References
Graham DY (2014) History of Helicobacter pylori, duodenal ulcer, gastric ulcer, and gastric cancer. World J Gastroenterol 20:5191–5204
Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ (2001) Helicobacter pylori infection and the development of gastric cancer. N Engl J Med 345(11):784–789
Graham DY (2000) Helicobacter pylori infection is the primary cause of gastric cancer. J Gastroenterol 35(12):90–97
Satoh K, Kimura K, Taniguchi Y, Yoshida Y, Kihira K, Takimoto T, Kawata H, Saifuku K, Ido K, Takemoto T, Ota Y, Tada M, Karita M, Sakaki N, Hoshihara Y (1996) Distribution of inflammation and atrophy in the stomach of helicobacter pylori-positive and -negative patients with chronic gastritis. Am J Gastroenterol 91(5):963–969
Xia HHX, Yang Y, Lam SK, Wong WM, Leung SY, Yuen ST, Elia G, Wright NA, Wong BC (2004) Aberrant epithelial expression of trefoil family factor 2 and mucin 6 in Helicobacter pylori infected gastric antrum, incisura, and body and its association with antralisation. J Clin Pathol 57(8):861–866
El-Zimaity HM, Ota H, Graham DY, Akamatsu T, Katsuyama T (2002) Patterns of gastric atrophy in intestinal type gastric carcinoma. Cancer 94(5):1428–1436
Xia HH, Zhang GS, Talley NJ, Wong BC, Yang Y, Henwood C, Wyatt JM, Adams S, Cheung K, Xia B, Zhu YQ, Lam SK (2002) Topographic association of gastric epithelial expression of Ki-67, Bax, and Bcl-2 with antralization in the gastric incisura, body, and fundus. Am J Gastroenterol 97(12):3023–3031
Xia HH, Lam SK, Wong WM, Hu WH, Lai KC, Wong SH, Leung SY, Yuen ST, Wright NA, Wong BC (2003 Jun) Antralization at the edge of proximal gastric ulcers: does helicobacter pylori infection play a role? World J Gastroenterol 9(6):1265–1269
Hattori T, Helpap B, Gedigk P (1982) The morphology and cell kinetics of pseudopyloric glands. Cell Pathol 39:31–40
Helpap B, Hattori T, Gedigk P (1981) Repair of gastric ulcer. A cell kinetic study. Pathol Anat 392:159–170
Whitehead R, Truelove SC, Gear MWL (1972) The histological diagnosis of chronic gastritis in fibreoptic gastroscope biopsy specimens. J Clon Pathol 25:1–11
Solcia E, Capella C, Fiocca R, Cornaggia M, Rindi G, Villani L, Bosi F, Ambrosiani L (1990) Exocrine and endocrine epithelial changes in types A and B chronic gastritis. Helicobacter Pylori, Gastritis Peptic Ulcer:245–258
Fiocca R, Villani L, Tenti P, Solcia E, Cornaggia M, Frigerio B, Capella C (1987) Characterization of four main cell types in gastric cancer: foveolar, mucopeptic, intestinal columnar and goblet cells. An histopathological, histochemical and ultrastructural study of early and advanced tumors. Pathol Res Pract 182:308–325
Cornaggia M, Capella C, Riva C, Finzi G, Solcia E (1986) Electron immunocytochemical localization of pepsinogen I (PgI) in chief cells, mucous-neck cells and transitional mucous-neck/chief cells of the human oxyntic mucosa. Histochemistry. 85(1):5–11
Kodama M, Murakami K, Okimoto T, Sato R, Uchida M, Abe T, Shiota S, Nakagawa Y, Mizukami K, Fujioka T (2012) Ten-year prospective follow-up of histological changes at five points on the gastric mucosa as recommended by the updated Sydney system after Helicobacter pylori eradication. J Gastroenterol 47(4):394–403
Kong YJ, Yi HG, Dai JC, Wei MX (2014) Histological changes of gastric mucosa after Helicobacter pylori eradication: a systematic review and meta-analysis. World J Gastroenterol 20(19):5903–5911
Schmidt PH, Lee JR, Joshi V, Playford RJ, Poulsom R, Wright NA, Goldenring JR (1999) Identification of a metaplastic cell lineage associated with human gastric adenocarcinoma. Lab Investig 79(6):639–646
Goldenring JR (2018) Pyloric metaplasia, pseudopyloric metaplasia, ulcer-associated cell lineage and spasmolytic polypeptide-expressing metaplasia: reparative lineages in the gastrointestinal mucosa. J Pathol 245(2):132–137
Lennerz JKM, Kim S, Oates EL, Huh WJ, Doherty JM, Tian X, Bredemeyer AJ, Goldenring JR, Lauwers GY, Shin YK, Mills JC (2010) The transcription factor MIST1 is a novel human gastric chief cell marker whose expression is lost in metaplasia, dysplasia and carcinoma. Amer J Pathol 177:1514–1533
Jin RU, Mills JC (2018) Are gastric and esophageal metaplasia relatives? The case for Barrett’s stemming from SPEM. Dig Dis Sci 63(8):2028–2041
Goldenring JR, Nam KT (2010) Oxyntic atrophy, metaplasia, and gastric cancer. Prog Mol Biol Transl Sci 96:117–131
Fox JG, Rogers AB, Whary MT, Ge Z, Ohtani M, Jones EK, Wang TC (2007) Accelerated progression of gastritis to dysplasia in the pyloric antrum of TFF2 -/- C57BL6 x Sv 129 Helicobacter pylori-infected mice. Am J Pathol 171:1520–1528
Dixon MF, Genta RM, Yardley JH, Correa P (1996) Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol 20(10):1161–1181
Nam KT, O'Neal RL, Coffey RJ, Finke PE, Barker N, Goldenring JR (2012) Spasmolytic polypeptide-expressing metaplasia (SPEM) in the gastric oxyntic mucosa does not arise from Lgr5-expressing cells. Gut. 61(12):1678–1685
Yoshizawa N, Takenaka Y, Yamaguchi H, Tetsuya T, Tanaka H, Tatematsu M, Nomura S, Goldenring JR, Kaminishi M (2007) Emergence of spasmolytic polypeptide expressing metaplasia in Mongolian gerbils infected with Helicobacter pylori. Lab Investig 87(12):1265–1276
Hu GY, Yu BP, Dong WG, Li MQ, Yu JP, Luo HS, Rang ZX (2003) Expression of TFF2 and Helicobacter pylori infection in carcinogenesis of gastric mucosa. World J Gastroenterol 9(5):910–914
Halldórsdóttir AM, Sigurdardóttrir M, Jónasson JG, Oddsdóttir M, Magnússon J, Lee JR, Goldenring JR (2003) Spasmolytic polypeptide-expressing metaplasia (SPEM) associated with gastric cancer in Iceland. Dig Dis Sci 48(3):431–441
Graham DY, Zou WY (2018) Guilt by association: intestinal metaplasia does not progress to gastric cancer. Curr Opin Gastroenterol 34(6):458–464
Kuo HY, Chang WL, Yeh YC, Cheng HC, Tsai YC, Wu CT, Lin SH, Yang HB, Lu CC, Sheu BS (2019) Spasmolytic polypeptide-expressing metaplasia associated with higher expressions of miR-21, 155, and 223 can be regressed by Helicobacter pylori eradication in the gastric cancer familial relatives. Helicobacter. 24(3):e12578
Acknowledgments
Sincere thanks are due to Dr. Ken-ichi Mukaisho of Department of Pathology, Faculty of Medicine, Shiga University of Medical Science, who revised the work. We also thank Mrs. Yoko Kudo and Mrs. Kanako Ito for help with immunohistochemical staining.
Author information
Authors and Affiliations
Contributions
Yasuhiro Wada contributed to design work, data analysis, and manuscript writing; Ryoji Kushima contributed to the conception of the work, revised the work and approved the final version manuscript; Masaaki Kodama contributed to design work, data analysis, and manuscript writing; Masahide Fukuda contributed to acquisition of data; Kensuke Fukuda contributed to acquisition of data; Kazuhisa Okamoto contributed to acquisition of data; Ryo Ogawa contributed to acquisition of data; Kazuhiro Mizukami contributed to acquisition of data; Tadayoshi Okimoto contributed to acquisition of data; Kazunari Murakami contributed to the conception of the work and critically revised it.
Corresponding author
Ethics declarations
The appropriate Ethical Committee approved the collection of the specimens included in this study, and informed consent was obtained from all participants.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Quality in Pathology
Rights and permissions
About this article
Cite this article
Wada, Y., Kushima, R., Kodama, M. et al. Histological changes associated with pyloric and pseudopyloric metaplasia after Helicobacter pylori eradication. Virchows Arch 477, 489–496 (2020). https://doi.org/10.1007/s00428-020-02805-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-020-02805-9