Abstract
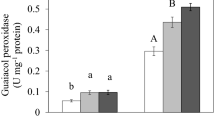
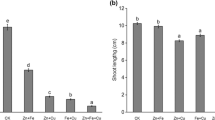
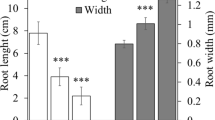
Copper (Cu) interferes with numerous biological functions in plants, including plant growth, which is partly governed by plant hormones. In the present study, Cu stress effect on the roots of pre-emerging maize seedlings in terms of growth, nutrient composition, protein modifications, and root hormone homeostasis was investigated, focusing on possible metabolic differences between the root apex and the rest of the root tissues. Significant decreases in root length and root biomass after 72 h of Cu exposure (50 and 100 μM CuCl2), accompanied by reductions in Ca, Mg, and P root contents, were found. Cu also generated cell redox imbalance in both root tissues and revealed by altered enzymatic and non-enzymatic antioxidant defenses. Oxidative stress was evidenced by an increased protein carbonylation level in both tissues. Copper also induced protein ubiquitylation and SUMOylation and affected 20S proteasome peptidase activities in both tissues. Drastic reductions in ABA, IAA, JA (both free and conjugated), GA3, and GA4 levels in the root apex were detected under Cu stress. Our results show that Cu exposure generated oxidative damage and altered root hormonal homeostasis, mainly at the root apex, leading to a strong root growth inhibition. Severe protein post-translational modifications upon Cu exposure occurred in both tissues, suggesting that even when hormonal adjustments to cope with Cu stress occurred mainly at the root apex, the entire root is compromised in the protein turnover that seems to be necessary to trigger and/or to sustain defense mechanisms against Cu toxicity.







Similar content being viewed by others
References
AbdElgawad H, Zinta G, Hamed BA, Selim S, Beemster G, Hozzein WN, Wadaan MAM, Asard H, Abuelsoud W (2020) Maize roots and shoots show distinct profiles of oxidative stress and antioxidant defense under heavy metal toxicity. Environ Pollut 258:113705. https://doi.org/10.1016/j.envpol.2019.113705
Abendroth LJ, Elmore RW, Boyer MJ, Marlay SK (2011) Corn growth and development. PMR 1009. Iowa State University Extension, Ames, Iowa
Achard P, Gong F, Cheminant S, Alioua M, Hedden P, Genschik P (2008) The cold-inducible CBF1 factor-dependent signaling pathway modulates the accumulation of the growth-repressing DELLA proteins via its effect on gibberellin metabolism. Plant Cell 20:2117–2129. https://doi.org/10.1105/tpc.108.058941
Aebi H (1984) Catalase in vitro. Oxygen radicals in biological systems. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahmad P, Rasool S, Gul A, Sheikh SA, Akram NA, Ashraf M, Kazi AM, Gucel S (2016) Jasmonates: multifunctional roles in stress tolerance. Front Plant Sci 7:813. https://doi.org/10.3389/fpls.2016.00813
Antoniadis V, Golia EE, Liu YT, Wang SL, Shaheen SM, Rinkleb J (2019) Soil and maize contamination by trace elements and associated health risk assessment in the industrial area of Volos, Greece. Environ Int 124:79–88. https://doi.org/10.1016/j.envint.2018.12.053
Augustine RC, York SL, Rytz TC, Vierstra RD (2016) Defining the SUMO system in maize: SUMOylation is up-regulated during endosperm development and rapidly induced by stress. Plant Physiol 171(3):2191–2210. https://doi.org/10.1104/pp.16.00353
Bartoli CG, Casalongué CA, Simontacchi M, Márquez-García B, Foyer CH (2013) Interactions between hormone and redox signalling pathways in the control of growth and cross tolerance to stress. Environ Exp Bot 94:73–88. https://doi.org/10.1016/j.envexpbot.2012.05.003
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Ben Massoud M, Sakouhi L, Karmous I, Zhu Y, El Ferjani E, Sheehan D, Chaoui A (2018) Protective role of exogenous phytohormones on redox status in pea seedlings under copper stress. J Plant Physiol 221:51–61. https://doi.org/10.1016/j.jplph.2017.11.014
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bulteau AN, Lundberg KC, Humphries KM, Sadek HA, Szweda PA, Friguet B, Szweda LI (2001) Oxidative modification and inactivation of the proteasome during coronary occlusion/reperfusion. J Biol Chem 276:30057–30063. https://doi.org/10.1074/jbc.M100142200
Burkhead JL, Reynolds KA, Abdel-Ghany SE, Cohu CM, Pilon M (2009) Copper homeostasis. New Phytol 182(4):799–816. https://doi.org/10.1111/j.1469-8137.2009.02846.x
Carrió-Seguí A, Romero P, Sanz A, Peñarrubia L (2016) Interaction between ABA signaling and copper homeostasis in Arabidopsis thaliana. Plant Cell Physiol 57(7):1568–1582. https://doi.org/10.1093/pcp/pcw087
Častorálová M, Ruml T, Knejzlík Z (2012) Using dot blot with immunochemical detection to evaluate global changes in SUMO-2/3 conjugation. Biotechniques 53(3). https://doi.org/10.2144/000113925
Catala R, Ouyang J, Abreu IA, Hu Y, Seo H, Zhang X, Chua NH (2007) The Arabidopsis E3 SUMO ligase SIZ1 regulates plant growth and drought responses. Plant Cell 19:2952–2966. https://doi.org/10.1105/tpc.106.049981
Černý M, Habánová H, Berka M, Luklová M, Brzobohatý B (2018) Hydrogen peroxide: its role in plant biology and crosstalk with signalling networks. Int J Mol Sci 19:2812. https://doi.org/10.3390/ijms19092812
Chen CC, Chen YY, Tang IC, Liang HM, Lai CC, Chiou JM, Yeh KC (2011) Arabidopsis SUMO E3 ligase SIZ1 is involved in excess copper tolerance. Plant Physiol 156(4):2225–2234. https://doi.org/10.1104/pp.111.178996
Conti L, Price G, O’Donnell E, Schwessinger B, Dominy P, Sadanandom A (2008) Small ubiquitin-like modifier proteases OVERLY TOLERANT TO SALT1 and −2 regulate SALT stress responses in Arabidopsis. Plant Cell 20:2894–2908. https://doi.org/10.1105/tpc.108.058669
De Tullio MC, Jiang K, Feldman LJ (2010) Redox regulation of root apical meristem organization: connecting root development to its environment. Plant Physiol Biochem 48:328–336. https://doi.org/10.1016/j.plaphy.2009.11.005
Djebali W, Gallusci P, Polge C, Boulila L, Galtier N, Raymond P, Chaibi W, Brouquisse R (2008) Modifications in endopeptidase and 20S proteasome expression and activities in cadmium treated tomato (Solanum lycopersicum L.) plants. Planta 227:625–639. https://doi.org/10.1007/s00425-007-0644-6
Durgbanshi A, Arbona V, Pozo O, Miersch O, Sancho JV, Gómez-Cadenas A (2005) Simultaneous determination of multiple phytohormones in plant extracts by liquid chromatography-electrospray tandem mass spectrometry. J Agric Food Chem 53:8437–8442. https://doi.org/10.1021/jf050884b
Farahat EA, Galal TM, Elawa OE, Hassan LM (2017) Health risk assessment and growth characteristics of wheat and maize crops irrigated with contaminated wastewater. Environ Monit Assess 189:535. https://doi.org/10.1007/s10661-017-6259-x
Florijn PJ, Van Beusichem ML (1993) Uptake and distribution of cadmium in maize inbred lines. Plant Soil 150:25–32. https://doi.org/10.1007/BF00779172
Fonseca S, Chini A, Hamberg M, Adie B, Porzel A, Kramell R, Miersch O, Wasternack C, Solano R (2009) (+)-7-iso-jasmonoyl-L-isoleucine is the endogenous bioactive jasmonate. Nat Chem Biol 5:344–350. https://doi.org/10.1038/nchembio.161
Foyer CH, Noctor G (2011) Ascorbate and glutathione: the heart of the redox hub. Plant Physiol 155:2–18. https://doi.org/10.1104/pp.110.167569
Gu Q, Yu T, Yang Z, Ji J, Hou Q, Wang L, Wei X, Zhang Q (2019) Prediction and risk assessment of five heavy metals in maize and peanut: a case study of Guangxi, China. Environ Toxicol Pharmacol 70:103199. https://doi.org/10.1016/j.etap.2019.103199
Guzel S, Terzi R (2013) Exogenous hydrogen peroxide increases dry matter production, mineral content and level of osmotic solutes in young maize leaves and alleviates deleterious effects of copper stress. Bot Stud 54:26. https://doi.org/10.1186/1999-3110-54-26
Hayat Q, Hayat S, Irfan M, Ahmad A (2010) Effect of exogenous salicylic acid under changing environment: a review. Environ Exp Bot 68:14–25. https://doi.org/10.1016/j.envexpbot.2009.08.005
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. California Agricultural Experiments Station, Circular 347
Hochholdinger F (2009) The maize root system: morphology, anatomy, and genetics. In: Bennetzen JL, Hake SC (eds) Handbook of maize: its biology. Springer, New York. https://doi.org/10.1007/978-0-387-79418-1_8
Hossain MA, Hoque MA, Burritt DJ, Fujita M (2014) Chapter 16 - proline protects plants against abiotic oxidative stress: biochemical and molecular mechanisms. In: Oxidative damage to plants antioxidant networks and signaling. Academic Press, Cambridge, pp 477–522. https://doi.org/10.1016/B978-0-12-799963-0.00016-2
Ingvardsen C, Veierskov B (2001) Ubiquitin- and proteasome-dependent proteolysis in plants. Physiol Plant 112:451–459. https://doi.org/10.1034/j.1399-3054.2001.1120401.x
Jung T, Höhn A, Grune T (2014) The proteasome and the degradation of oxidized proteins: part II – protein oxidation and proteasomal degradation. Redox Biol 2:99–104. https://doi.org/10.1016/j.redox.2013.12.008
Kelley DR, Estelle M (2012) Ubiquitin-mediated control of plant hormone signaling. Plant Physiol 160:47–55. https://doi.org/10.1104/pp.112.200527
Kumar P, Tewari RK, Sharma PN (2008) Modulation of copper toxicity-induced oxidative damage by excess supply of iron in maize plants. Plant Cell Rep 27:399–409. https://doi.org/10.1007/s00299-007-0453-1
Kurepa J, Walker JM, Smalle J, Gosink MM, Davis SJ, Durham TL, Sung DY, Vierstra RD (2003) The small ubiquitin-like modifier (SUMO) protein modification system in Arabidopsis. Accumulation of SUMO1 and −2 conjugates is increased by stress. J Biol Chem 278(9):6862–6872. https://doi.org/10.1074/jbc.M209694200
Law MY, Charles SA, Halliwell B (1983) Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplast. The effect of hydrogen peroxide and paraquat. Biochem J 210:899–903. https://doi.org/10.1042/bj2100899
Li Q, Chen HH, Qi YP, Ye X, Yang LT, Huang ZR, Chen LS (2019) Excess copper effects on growth, uptake of water and nutrients, carbohydrates, and PSII photochemistry revealed by OJIP transients in Citrus seedlings. Environ Sci Pollut Res 26:30188–30205. https://doi.org/10.1007/s11356-019-06170-2
Liu T, Zhu S, Tang Q, Tang S (2015) Genome-wide transcriptomic profiling of ramie (Boehmeria nivea L. Gaud) in response to cadmium stress. Gene 558:131–137. https://doi.org/10.1016/j.gene.2014.12.057
Liu J, Wang J, Lee S, Wen R (2018) Copper-caused oxidative stress triggers the activation of antioxidant enzymes via ZmMPK3 in maize leaves. PLoS One 13(9):e0203612. https://doi.org/10.1371/journal.pone.0203612
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Madejón P, Ramírez-Benítez JE, Corrales I, Barceló J, Poschenrieder C (2009) Copper-induced oxidative damage and enhanced antioxidant defenses in the root apex of maize cultivars differing in Cu tolerance. Environ Exp Bot 67:415–420. https://doi.org/10.1016/j.envexpbot.2009.08.006
Magome H, Yamaguchi S, Hanada A, Kamiya Y, Oda K (2008) The DDF1 transcriptional activator upregulates expression of a gibberellin-deactivating gene, GA2ox7, under high-salinity stress in Arabidopsis. Plant J 56:613–626. https://doi.org/10.1111/j.1365-313X.2008.03627.x
Marastoni L, Tauber P, Pii Y, Valentinuzzi F, Astolfi S, Simoni A, Brunetto G, Cesco S, Mimmo T (2019) The potential of two different Avena sativa L. cultivars to alleviate cu toxicity. Ecotoxicol Environ Saf 182:109430. https://doi.org/10.1016/j.ecoenv.2019.109430
Miura K, Lee J, Gong Q, Ma S, Jin JB, Yoo CY, Miura T, Sato A, Bohnert HJ, Hasegawa PM (2011) SIZ1 regulation of phosphate starvation-induced root architecture remodeling involves the control of auxin accumulation. Plant Physiol 155:1000–1012. https://doi.org/10.1105/tpc.114.133090
Moravcová Š, Tůma J, Dučaiová ZK, Waligórski P, Kula M, Saja D, Słomka A, Bąba W, Libik-Konieczny M (2018) Influence of salicylic acid pretreatment on seeds germination and some defence mechanisms of Zea mays plants under copper stress. Plant Physiol Biochem 122:19–30. https://doi.org/10.1016/j.plaphy.2017.11.007
Mostofa MG, Fujita M (2013) Salicylic acid alleviates copper toxicity in rice (Oryza sativa L.) seedlings by up-regulating antioxidative and glyoxalase systems. Ecotoxicol 22:959–973. https://doi.org/10.1007/s10646-013-1073-x
Mostofa MG, Hossain MA, Fujita M, Tran LS (2015) Physiological and biochemical mechanisms associated with trehalose-induced copper-stress tolerance in rice. Sci Rep 5:11433. https://doi.org/10.1038/srep11433
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplast. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nannoni F, Rossi S, Protano G (2016) Potentially toxic element contamination in soil and accumulation in maize plants in a smelter area in Kosovo. Environ Sci Pollut Res 23:11937–11946. https://doi.org/10.1007/s11356-016-6411-0
Ninh Pham A, Xing G, Miller CJ, David Waite T (2013) Fenton-like copper redox chemistry revisited: hydrogen peroxide and superoxide mediation of copper-catalyzed oxidant production. J Catal 301:54–64. https://doi.org/10.1016/j.jcat.2013.01.025
Orlowski M, Wilk S (2000) Catalytic activities of the 20S proteasome, a multicatalytic proteinase complex. Arch Biochem Biophys 383:1–16. https://doi.org/10.1006/abbi.2000.2036
Ouzounidou G, Čiamporová M, Moustakas M, Karataglis S (1995) Responses of maize (Zea mays L.) plants to copper stress-I. growth, mineral content and ultrastructure of roots. Environ Exp Bot 35:167–176. https://doi.org/10.1016/0098-8472(94)00049-B
Pena LB, Pasquini LA, Tomaro ML, Gallego SM (2007) 20S proteasome and accumulation of oxidized and ubiquitinated proteins in maize leaves subjected to cadmium stress. Phytochemistry 68:1139–1146. https://doi.org/10.1016/j.phytochem.2007.02.022
Pena LB, Azpilicueta CE, Gallego SM (2011) Sunflower cotyledons cope with copper stress by inducing catalase subunits less sensitive to oxidation. J Trace Elem Med Biol 25(3):125–129. https://doi.org/10.1016/j.jtemb.2011.05.001
Pilet PE, Saugy M (1987) Effect on root growth of endogenous and applied IAA and ABA: a critical re-examination. Plant Physiol 83:33–38. https://doi.org/10.1104/pp.83.1.33
Piñeros MA, Shaff JE, Kochian V (1998) Development, characterization, and application of a cadmium-selective microelectrode for the measurement of cadmium fluxes in roots of Thlaspi species and wheat. Plant Physiol 116:1393–1401. https://doi.org/10.1104/pp.116.4.1393
Polge C, Jaquinod M, Holzer F, Bourguignon J, Walling L, Brouquisse R (2009) Evidence for the existence in Arabidopsis thaliana of the proteasome proteolytic pathway: activation in response to cadmium. J Biol Chem 284(51):35412–35424. https://doi.org/10.1074/jbc.M109.035394
Popova LP, Maslenkova LT, Yordanova RY, Ivanova AP, Kranteva AP, Szalai G, Janda T (2009) Exogenous treatment with salicylic acid attenuates cadmium toxicity in pea seedlings. Plant Physiol Biochem 47(3):224–231. https://doi.org/10.1016/j.plaphy.2008.11.007
Raynes R, Pomatto LCD, Davies KJA (2016) Degradation of oxidized proteins by the proteasome: distinguishing between the 20S, 26S, and immunoproteasome proteolytic pathways. Mol Asp Med 50:41–55. https://doi.org/10.1016/j.mam.2016.05.001
Robyt JF, White BJ (1990) Biochemical techniques: theory and practice. Waveland Press, Inc., Prospect Heights
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signalling. Nature 459:1071–1078. https://doi.org/10.1038/nature08122
Shakirova FM, Allagulova CR, Maslennikova DR, Klyuchnikova EO, Avalbaev AM, Bezrukova MV (2016) Salicylic acid-induced protection against cadmium toxicity in wheat plants. Environ Exp Bot 122:19–28. https://doi.org/10.1016/j.envexpbot.2015.08.002
Sharma S, Uttam KN (2018) Early stage detection of stress due to copper on maize (Zea mays L.) by laser-induced fluorescence and infrared spectroscopy. J Appl Spectrosc 85:771. https://doi.org/10.1007/s10812-018-0717-2
Shiyab S (2018) Phytoaccumulation of copper from irrigation water and its effect on the internal structure of lettuce. Agriculture 8:29. https://doi.org/10.3390/agriculture8020029
Singh S, Singh A, Bashri G, Prasad SM (2016) Impact of cd stress on cellular functioning and its amelioration by phytohormones: an overview on regulatory network. Plant Growth Regul 80:253–263. https://doi.org/10.1007/s10725-016-0170-2
Skelly MJ, Frungillo L, Spoel SH (2016) Transcriptional regulation by complex interplay between post-translational modifications. Curr Opin Plant Biol 33:126–132. https://doi.org/10.1016/j.pbi.2016.07.004
Soares AM, Souza TF, Jacinto T, Machado OLT (2010) Effect of methyl jasmonate on antioxidative enzyme activities and on the contents of ROS and H2O2 in Ricinus communis leaves. Braz J Plant Physiol 22:151–158. https://doi.org/10.1590/S1677-04202010000300001
Tai H, Lu X, Opitz N, Marcon C, Paschold A, Lithio A, Nettleton D, Hochholdinger F (2016) Transcriptomic and anatomical complexity of primary, seminal, and crown roots highlight root type-specific functional diversity in maize (Zea mays L.). J Exp Bot 67(4):1123–1135. https://doi.org/10.1093/jxb/erv513
Tang RJ, Luan S (2017) Regulation of calcium and magnesium homeostasis in plants: from transporters to signaling network. Curr Opin Plant Biol 39:97–105. https://doi.org/10.1016/j.pbi.2017.06.009
Thounaojam TC, Panda P, Mazumdar P, Kumar D, Sharma GD, Sahoo L, Sanjib P (2012) Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol Biochem 53:33–39. https://doi.org/10.1016/j.plaphy.2012.01.006
Todgham A, Hoaglund E, Hofmann G (2007) Is cold the new hot? Elevated ubiquitin-conjugated protein levels in tissues of Antarctic fish as evidence for cold-denaturation of proteins in vivo. J Comp Physiol B 177(8):857–866. https://doi.org/10.1007/s00360-007-0183-2
Tóth G, Hermann T, Da Silva MR, Montanarella L (2016) Heavy metals in agricultural soils of the European Union with implications for food safety. Environ Int 88:299–309. https://doi.org/10.1016/j.envint.2015.12.017
Vatehová Z, Malovíková A, Kollárová K, Kučerová D, Lišková D (2016) Impact of cadmium stress on two maize hybrids. Plant Physiol Biochem 108:90–98. https://doi.org/10.1016/j.plaphy.2016.06.035
Vavoulidou E, Avramides EJ, Papadopoulos P, Dimirkou A, Charoulis A, Konstantinidou-Doltsinis S (2005) Copper content in agricultural soils related to cropping systems in different regions of Greece. Commun Soil Sci Plant Anal 36:759–773. https://doi.org/10.1081/CSS-2000433677/s10812-018-0717-2
Wasternack C, Hause B (2013) Jasmonates: biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann Bot 111:1021–1058. https://doi.org/10.1093/aob/mct067
Wasternack C, Song S (2017) Jasmonates: biosynthesis, metabolism, and signaling by proteins activating and repressing transcription. J Exp Bot 68:1303–1321. https://doi.org/10.1093/jxb/erw443
Wehr NB, Levine RL (2012) Quantitation of protein carbonylation by dot blot. Anal Biochem 423:241–245. https://doi.org/10.1016/j.ab.2012.01.031
Ye N, Li H, Zhu G, Liu Y, Liu R, Xu W, Jing Y, Peng X, Zhang J (2014) Copper suppresses abscisic acid catabolism and catalase activity, and inhibits seed germination of rice. Plant Cell Physiol 55:2008–2016. https://doi.org/10.1093/pcp/pcu136
Yoo CY, Miura K, Jin JB, Lee J, Park HC, Salt DE, Yun DJ, Bressan RA, Hasegawa PM (2006) SIZ1 small ubiquitin-like modifier E3 ligase facilitates basal thermotolerance in Arabidopsis independent of salicylic acid. Plant Physiol 142:1548–1558. https://doi.org/10.1104/pp.106.088831
Yuan HM, Xu HH, Liu WC, Lu YT (2013) Copper regulates primary root elongation through PIN1-mediated auxin redistribution. Plant Cell Physiol 54(5):766–778. https://doi.org/10.1093/pcp/pct030
Zeng Q, Ling Q, Wu J, Yang Z, Liu R, Qi Y (2019) Excess copper-induced changes in antioxidative enzyme activity, mineral nutrient uptake and translocation in sugarcane seedlings. Bull Environ Contam Toxicol 103:834–840. https://doi.org/10.1007/s00128-019-02735-6
Acknowledgments
We thank Dr. Myriam S. Zawoznik for her helpful criticism and for improving the English. CLM is a Research Fellow at the UBA (Argentina). LBP and SMG are Career Investigators from CONICET (Argentina). Hormone measurements were performed at Servei Central d’Instrumentació Científica (SCIC) of Universitat Jaume I (Spain).
Funding
This work was supported by the Universidad de Buenos Aires (20020170100331BA UBACYT), and Consejo Nacional de Investigaciones Científicas y Técnicas (PIP 0266).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Néstor Carrillo
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Fig. 1
Hydroponic culture and maize seedlings. The left panel is a top view of the hydroponic system. The right panels show representative images of control maize seedlings and Cu-treated (100 μM of CuCl2) maize seedlings at 72 h of metal treatment (BMP 1349 kb).
Supplemental Fig. 2
Representative image of the dot blots obtained to determine carbonylated proteins. Root protein extracts obtained from the root apex (Ap) and the remaining root tissue (Rt) of control (C) and Cu-treated seedlings (50 and 100 μM of CuCl2) were derivatized with 2,4-DNPH. Equal amounts of total protein (7.5 μg) were blotted onto a PVDF, blocked in 5% (w/v) non-fat dried milk in PBS, and incubated at room temperature for 1 h with anti-DNP (Sigma, St Luis) as the primary antibody. Dots were subsequently visualized using a secondary rabbit antibody conjugated with horseradish peroxidase (HRP) and stained using 3,3′-diaminobenzidine (DAB) as substrate. The dot blots shown are representative of four replicates prepared in five independent experiments (PPT 136 kb).
Rights and permissions
About this article
Cite this article
Matayoshi, C.L., Pena, L.B., Arbona, V. et al. Early responses of maize seedlings to Cu stress include sharp decreases in gibberellins and jasmonates in the root apex. Protoplasma 257, 1243–1256 (2020). https://doi.org/10.1007/s00709-020-01504-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-020-01504-1