Abstract
Purpose
Fibrocalcific aortic valve disease (CAVD) is caused by the deposition of calcific nodules in the aortic valve leaflets, resulting in progressive loss of function that ultimately requires surgical intervention. This process is actively mediated by the resident valvular interstitial cells (VICs), which, in response to oxidized lipids, transition from a quiescent to an osteoblast-like state. The purpose of this study was to examine if the ryanodine receptor, an intracellular calcium channel, could be therapeutically targeted to prevent this phenotypic conversion.
Methods
The expression of the ryanodine receptor in porcine aortic VICs was characterized by qRT-PCR and immunofluorescence. Next, the VICs were exposed to lysophosphatidylcholine, an oxidized lipid commonly found in low-density lipoprotein, while the activity of the ryanodine receptor was modulated with ryanodine. The cultures were analyzed for markers of cellular mineralization, alkaline phosphatase activity, proliferation, and apoptosis.
Results
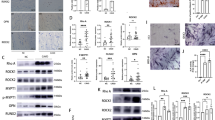
Porcine aortic VICs predominantly express isoform 3 of the ryanodine receptors, and this protein mediates the cellular response to LPC. Exposure to LPC caused elevated intracellular calcium concentration in VICs, raised levels of alkaline phosphatase activity, and increased calcific nodule formation, but these changes were reversed when the activity of the ryanodine receptor was blocked.
Conclusions
Our findings suggest blocking the activity of the ryanodine receptor can attenuate the valvular mineralization caused by LPC. We conclude that oxidized lipids, such as LPC, play an important role in the development and progression of CAVD and that the ryanodine receptor is a promising target for pharmacological intervention.






Similar content being viewed by others
References
Anderson, H. C. Matrix vesicles and calcification. Curr. Rheumatol. Rep. 5:222–226, 2003. https://doi.org/10.1007/s11926-003-0071-z.
Balaoing, L. R., A. D. Post, A. Y. Lin, H. Tseng, J. L. Moake, and K. J. Grande-Allen. Laminin peptide-immobilized hydrogels modulate valve endothelial cell hemostatic regulation. PLoS ONE. 10:e0130749, 2015. https://doi.org/10.1371/journal.pone.0130749.
Benjamin, E. J., M. J. Blaha, S. E. Chiuve, M. Cushman, S. R. Das, R. Deo, S. D. De Ferranti, J. Floyd, M. Fornage, C. Gillespie, C. R. Isasi, M. C. Jimenez, L. C. Jordan, S. E. Judd, D. Lackland, J. H. Lichtman, L. Lisabeth, S. Liu, C. T. Longenecker, R. H. MacKey, K. Matsushita, D. Mozaffarian, M. E. Mussolino, K. Nasir, R. W. Neumar, L. Palaniappan, D. K. Pandey, R. R. Thiagarajan, M. J. Reeves, M. Ritchey, C. J. Rodriguez, G. A. Roth, W. D. Rosamond, C. Sasson, A. Towfghi, C. W. Tsao, M. B. Turner, S. S. Virani, J. H. Voeks, J. Z. Willey, J. T. Wilkins, J. H. Y. Wu, H. M. Alger, S. S. Wong, and P. Muntner, Heart disease and stroke statistics—2017 update: a report from the American Heart Association. Circulation. 135:e146–e603, 2017. https://doi.org/10.1161/CIR.0000000000000485.
Bowler, M. A., and W. D. Merryman. In vitro models of aortic valve calcification: solidifying a system. Cardiovasc. Pathol. 24:1–10, 2014. https://doi.org/10.1016/j.carpath.2014.08.003.
Chen, J. H., and C. A. Simmons. Cell-matrix interactions in the pathobiology of calcific aortic valve disease: critical roles for matricellular, matricrine, and matrix mechanics cues. Circ. Res. 108:1510–1524, 2011. https://doi.org/10.1161/CIRCRESAHA.110.234237.
Cheng, H., Q. Yao, R. Song, Y. Zhai, W. Wang, D. A. Fullerton, and X. Meng. Lysophosphatidylcholine activates the Akt pathway to upregulate extracellular matrix protein production in human aortic valve cells. J. Surg. Res. 213:243–250, 2017. https://doi.org/10.1016/j.jss.2017.02.028.
Clapham, D. E. Calcium signaling. Cell. 131:1047–1058, 2007. https://doi.org/10.1016/j.cell.2007.11.028.
Drzazga, A., A. Sowinska, A. Krzeminska, P. Rytczak, M. Koziolkiewicz, and E. Gendaszewska-Darmach. Lysophosphatidylcholine elicits intracellular calcium signaling in a GPR55-dependent manner. Biochem. Biophys. Res. Commun. 489:242–247, 2017. https://doi.org/10.1016/j.bbrc.2017.05.145.
Fill, M., and J. A. Copello. Ryanodine receptor calcium release channels. Physiol. Rev. 82:893–922, 2002. https://doi.org/10.1152/physrev.00013.2002.
Garg, A., S. V. Rao, G. Visveswaran, S. Agrawal, A. Sharma, L. Garg, I. Mahata, J. Garg, D. Singal, M. Cohen, and J. B. Kostis. Transcatheter aortic valve replacement versus surgical valve replacement in low-intermediate surgical risk patients: a systematic review and meta-analysis. J. Invasive Cardiol. 29:209–216, 2017.
Gomel, M. A., R. Lee, and K. J. Grande-Allen. Comparing the role of mechanical forces in vascular and valvular calcification progression. Front. Cardiovasc. Med. 5:197, 2019. https://doi.org/10.3389/fcvm.2018.00197.
Gregory, C. A., W. G. Gunn, A. Peister, and D. J. Prockop. An Alizarin red-based assay of mineralization by adherent cells in culture: comparison with cetylpyridinium chloride extraction. Anal. Biochem. 329:77–84, 2004. https://doi.org/10.1016/j.ab.2004.02.002.
Gu, X., and K. S. Masters. Role of the Rho pathway in regulating valvular interstitial cell phenotype and nodule formation. Am. J. Physiol. 2011. https://doi.org/10.1152/ajpheart.01178.2009.
Hjortnaes, J., C. Goettsch, J. D. Hutcheson, G. Camci-Unal, L. Lax, K. Scherer, S. Body, F. J. Schoen, J. Kluin, A. Khademhosseini, and E. Aikawa. Simulation of early calcific aortic valve disease in a 3D platform: a role for myofibroblast differentiation. J. Mol. Cell. Cardiol. 94:13–20, 2016. https://doi.org/10.1016/j.yjmcc.2016.03.004.
Hoffman, B. D., C. Grashoff, and M. A. Schwartz. Dynamic molecular processes mediate cellular mechanotransduction. Nature. 475:316–323, 2011. https://doi.org/10.1038/nature10316.
Huk, D. J., B. F. Austin, T. E. Horne, R. B. Hinton, W. C. Ray, D. D. Heistad, and J. Lincoln. Valve endothelial cell-derived Tgfβ1 signaling promotes nuclear localization of Sox9 in interstitial cells associated with attenuated calcification significance. Arterioscler. Thromb. Vasc. Biol. 36:328–338, 2016. https://doi.org/10.1161/ATVBAHA.115.306091.
Iannotti, J. P., S. Naidu, Y. Noguchi, R. M. Hunt, and C. T. Brighton. Growth plate matrix vesicle biogenesis. The role of intracellular calcium. Clin. Orthop. Relat. Res. 306:222–229, 1994.
Jeong, H., Y. H. Kim, Y. Lee, S. J. Jung, and S. B. Oh. TRPM2 contributes to LPC-induced intracellular Ca2+ influx and microglial activation. Biochem. Biophys. Res. Commun. 485:301–306, 2017. https://doi.org/10.1016/j.bbrc.2017.02.087.
Jono, S., Y. Nishizawa, A. Shioi, and H. Morii. 1,25-Dihydroxyvitamin D3 increases in vitro vascular calcification by modulating secretion of endogenous parathyroid hormone related peptide. Circulation. 98:1302–1306, 1998. https://doi.org/10.1161/01.CIR.98.13.1302.
Kim, K. M. Apoptosis and calcification. Scanning Microsc. 9:1137–1175, 1995.
Krause, T., M. U. Gerbershagen, M. Fiege, R. Weisshorn, and F. Wappler. Dantrolene—a review of its pharmacology, therapeutic use and new developments. Anaesthesia. 59:364–373, 2004. https://doi.org/10.1111/j.1365-2044.2004.03658.x.
Li, X. H., and Y. J. Wu. Characteristics of lysophosphatidylcholine-induced Ca2+ response in human neuroblastoma SH-SY5Y cells. Life Sci. 80:886–892, 2007. https://doi.org/10.1016/j.lfs.2006.11.017.
Li, C., S. Xu, and A. I. Gotlieb. The progression of calcific aortic valve disease through injury, cell dysfunction, and disruptive biologic and physical force feedback loops. Cardiovasc. Pathol. 22:1–8, 2013. https://doi.org/10.1016/j.carpath.2012.06.005.
Liu, A. C., V. R. Joag, and A. I. Gotlieb. The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am. J. Pathol. 171:1407–1418, 2007. https://doi.org/10.2353/ajpath.2007.070251.
Mahmut, A., M.-C. Boulanger, D. El Husseini, D. Fournier, R. Bouchareb, J.-P. Després, P. Pibarot, Y. Bossé, and P. Mathieu. Elevated expression of lipoprotein-associated phospholipase A2 in calcific aortic valve disease. J. Am. Coll. Cardiol. 63:460–469, 2014. https://doi.org/10.1016/j.jacc.2013.05.105.
Massaeli, H., J. A. Austria, and G. N. Pierce. Lesions in ryanodine channels in smooth muscle cells exposed to oxidized low density lipoprotein. Arter. Thromb Vasc Biol. 20:328–334, 2000.
Mathieu, P., and M. C. Boulanger. Basic mechanisms of calcific aortic valve disease. Can. J. Cardiol. 30:982–993, 2014. https://doi.org/10.1016/j.cjca.2014.03.029.
Meissner, G. Ryanodine receptor/Ca2+ release channels and their regulation by endogenous effectors. Annu. Rev. Physiol. 56:485–508, 1994. https://doi.org/10.1146/annurev.ph.56.030194.002413.
Mohty, D., P. Pibarot, J. P. Després, C. Côté, B. Arsenault, A. Cartier, P. Cosnay, C. Couture, and P. Mathieu. Association between plasma LDL particle size, valvular accumulation of oxidized LDL, and inflammation in patients with aortic stenosis. Arterioscler. Thromb. Vasc. Biol. 28:187–193, 2008. https://doi.org/10.1161/ATVBAHA.107.154989.
Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65:55–63, 1983. https://doi.org/10.1016/0022-1759(83)90303-4.
Nakamura, Y., M. Yasukochi, S. Kobayashi, K. Uehara, A. Honda, R. Inoue, I. Imanaga, and A. Uehara. Cell membrane-derived lysophosphatidylcholine activates cardiac ryanodine receptor channels. Pflugers Arch. 453:455–462, 2007. https://doi.org/10.1007/s00424-006-0141-y.
Parisi, V., D. Leosco, G. Ferro, A. Bevilacqua, G. Pagano, C. de Lucia, P. Perrone Filardi, A. Caruso, G. Rengo, and N. Ferrara. The lipid theory in the pathogenesis of calcific aortic stenosis. Nutr. Metab. Cardiovasc. Dis. 25:519–525, 2015. https://doi.org/10.1016/j.numecd.2015.02.001.
Puperi, D. S., R. W. O’Connell, Z. E. Punske, Y. Wu, J. L. West, and K. J. Grande-Allen. Hyaluronan hydrogels for a biomimetic spongiosa layer of tissue engineered heart valve scaffolds. Biomacromolecules. 17:1766–1775, 2016. https://doi.org/10.1021/acs.biomac.6b00180.
Rajamannan, N. M., F. J. Evans, E. Aikawa, K. J. Grande-Allen, L. L. Demer, D. D. Heistad, C. A. Simmons, K. S. Masters, P. Mathieu, K. D. O’Brien, F. J. Schoen, D. A. Towler, A. P. Yoganathan, and C. M. Otto. Calcific aortic valve disease: not simply a degenerative process: a review and agenda for research from the National Heart and Lung and Blood Institute Aortic Stenosis Working Group. Executive summary: calcific aortic valve disease-2011 update. Circulation. 124:1783–1791, 2011. https://doi.org/10.1161/CIRCULATIONAHA.110.006767.
Rodriguez, K. J., L. M. Piechura, and K. S. Masters. Regulation of valvular interstitial cell phenotype and function by hyaluronic acid in 2-D and 3-D culture environments. Matrix Biol. 30:70–82, 2011. https://doi.org/10.1016/j.matbio.2010.09.001.
Rodriguez, K. J., L. M. Piechura, A. M. Porras, and K. S. Masters. Manipulation of valve composition to elucidate the role of collagen in aortic valve calcification. BMC Cardiovasc. Disord. 14:29, 2014. https://doi.org/10.1186/1471-2261-14-29.
Song, R., Q. Zeng, L. Ao, J. A. Yu, J. C. Cleveland, K. S. Zhao, D. A. Fullerton, and X. Meng. Biglycan induces the expression of osteogenic factors in human aortic valve interstitial cells via toll-like receptor-2. Arterioscler. Thromb. Vasc. Biol. 32:2711–2720, 2012. https://doi.org/10.1161/ATVBAHA.112.300116.
Stephens, E. H., J. L. Carroll, and K. J. Grande-Allen. The use of collagenase III for the isolation of porcine aortic valvular interstitial cells: rationale and optimization. J. Heart Valve Dis. 16:175–183, 2007.
Stephens, E. H., J. G. Saltarrelli, L. S. Baggett, I. Nandi, J. J. Kuo, A. R. Davis, E. A. Olmsted-Davis, M. J. Reardon, J. D. Morrisett, and K. J. Grande-Allen. Differential proteoglycan and hyaluronan distribution in calcified aortic valves. Cardiovasc. Pathol. 20:334–342, 2011. https://doi.org/10.1016/j.carpath.2010.10.002.
Sutko, J. L., J. A. Airey, W. Welch, and L. Ruest. The pharmacology of ryanodine and related compounds. Pharmacol. Rev. 49:53–98, 1997.
Sylvester, P. W. Optimization of the tetrazolium dye (MTT) colorimetric assay for cellular growth and viability. Methods Mol. Biol. 716:157–168, 2011. https://doi.org/10.1007/978-1-61779-012-6_9.
Thayer, P., K. Balachandran, S. Rathan, C. H. Yap, S. Arjunon, H. Jo, and A. P. Yoganathan. The effects of combined cyclic stretch and pressure on the aortic valve interstitial cell phenotype. Ann. Biomed. Eng. 39:1654–1667, 2011. https://doi.org/10.1007/s10439-011-0273-x.
Tillman, T. S., and M. Cascio. Effects of membrane lipids on ion channel structure and function. Cell Biochem. Biophys. 38:161–190, 2003. https://doi.org/10.1385/cbb:38:2:161.
Towler, D. A. Molecular and cellular aspects of calcific aortic valve disease. Circ. Res. 113:198–208, 2013. https://doi.org/10.1161/CIRCRESAHA.113.300155.
Van Petegem, F. Ryanodine receptors: structure and function. J. Biol. Chem. 287:31624–31632, 2012. https://doi.org/10.1074/jbc.R112.349068.
Vater, C., P. Kasten, and M. Stiehler. Culture media for the differentiation of mesenchymal stromal cells. Acta Biomater. 7:463–477, 2011. https://doi.org/10.1016/j.actbio.2010.07.037.
Wang, H., M. W. Tibbitt, S. J. Langer, L. A. Leinwand, and K. S. Anseth. Hydrogels preserve native phenotypes of valvular fibroblasts through an elasticity-regulated PI3K/AKT pathway. Proc. Natl. Acad. Sci. U. S. A. 110:19336–19341, 2013. https://doi.org/10.1073/pnas.1306369110.
Wiltz, D. C., R. I. Han, R. L. Wilson, A. Kumar, J. D. Morrisett, and K. J. Grande-Allen. Differential aortic and mitral valve interstitial cell mineralization and the induction of mineralization by lysophosphatidylcholine in vitro. Cardiovasc. Eng. Technol. 5:371–383, 2014. https://doi.org/10.1007/s13239-014-0197-3.
Wu, Y., D. S. Puperi, K. J. Grande-Allen, and J. L. West. Ascorbic acid promotes extracellular matrix deposition while preserving valve interstitial cell quiescence within 3D hydrogel scaffolds. J. Tissue Eng. Regen. Med. 11:1963–1973, 2017. https://doi.org/10.1002/term.2093.
Yip, C. Y. Y., J. H. Chen, R. Zhao, and C. A. Simmons. Calcification by valve interstitial cells is regulated by the stiffness of the extracellular matrix. Arterioscler. Thromb. Vasc. Biol. 29:936–942, 2009. https://doi.org/10.1161/ATVBAHA.108.182394.
Zeng, Q., R. Song, D. A. Fullerton, L. Ao, Y. Zhai, S. Li, D. B. Ballak, J. C. Cleveland, T. B. Reece, T. A. McKinsey, D. Xu, C. A. Dinarello, and X. Meng. Interleukin-37 suppresses the osteogenic responses of human aortic valve interstitial cells in vitro and alleviates valve lesions in mice. Proc. Natl. Acad. Sci. 114:1631–1636, 2017. https://doi.org/10.1073/pnas.1619667114.
Zhang, X., B. Xu, D. S. Puperi, A. L. Yonezawa, Y. Wu, H. Tseng, M. L. Cuchiara, J. L. West, and K. J. Grande-Allen. Integrating valve-inspired design features into poly(ethylene glycol) hydrogel scaffolds for heart valve tissue engineering. Acta Biomater. 14:11–21, 2015. https://doi.org/10.1016/j.actbio.2014.11.042.
Acknowledgments
This work was supported by the National Heart, Lung and Blood Institute [R21HL104377 to K.J.G.A. and T32HL007812 to J.D.M.] and the National Institute of Diabetes and Digestive and Kidney Disease [F30DK108541 to R.L.W.] of the National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The authors thank Jennifer P. Connell for her critical reading of the manuscript and Marci Kang and Vaidya Parthasarathy for their suggestions regarding research directions.
Conflict of interest
The authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Craig Alexander Simmons oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wilson, R.L., Sylvester, C.B., Wiltz, D.C. et al. The Ryanodine Receptor Contributes to the Lysophosphatidylcholine-Induced Mineralization in Valvular Interstitial Cells. Cardiovasc Eng Tech 11, 316–327 (2020). https://doi.org/10.1007/s13239-020-00463-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-020-00463-1