Abstract
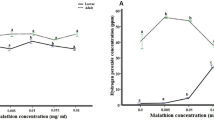
Pesticides exert a wide variety of functions for controlling pests. At low doses the pests are not much affected and parallel to that a chance of tolerance has been expected. In this study a population of fruit flies Drosophila melanogaster was grown under various doses of insecticide Malathion and herbicide Paraquat. Lower than the sub-lethal doses (0.2 micromolar), various biomolecular alterations in terms of protein oxidation (Advanced Oxidation Protein Products, AOPP), lipid peroxidation (Malondialdehyde, MDA), sialic acids, total thiols, Cupric Ion Reducing Antioxidant Capacity (CUPRAC) and 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) cation radical scavenging activity have been observed. Positive alteration in AOPP for both Malathion and Paraquat treated fruit flies as well as increased MDA content in Paraquat treated flies are the alterations in the fruit fly population because of oxidative stress. The unaffected MDA, sialic acid content and CUPRAC antioxidant capacity for Malathion treated flies demonstrates the diminished effects of oxidative stress exerted through Malathion. There is increase sialic acid and CUPRAC antioxidant capacity after treatment of Paraquat. There is also an increase in total thiols and ABTS cation radical scavenging activity after treatment of both Malathion and Paraquat. These results evidently show that improved antioxidant mechanisms effectively alleviate the oxidative stress exerted by pesticides. This study is clearly reflecting that oxidative stress generated by the sub lethal doses of pesticides can be diminished and certain tolerance level is also achieved by the insects in terms of increased antioxidant defence and longevity.

Graphical abstract






Similar content being viewed by others
References
Agrawal A, Pandey RS, Sharma B (2010) Water pollution with special reference to pesticide contamination in India. J Water Res Prot 2(05):432–448. https://doi.org/10.4236/jwarp.2010.25050
Apak R, Güçlü K, Özyürek M, Karademir SE, Altun M (2005) Total antioxidant capacity assay of human serum using copper (II)-neocuproine as chromogenic oxidant: the CUPRAC method. Free Radic Res 39(9):949–961. https://doi.org/10.1080/10715760500210145
Araujo SM, de Paula MT, Poetini MR, Meichtry L, Bortolotto VC, Zarzecki MS, Jesse CR, Prigol M (2015) Effectiveness of γ-oryzanol in reducing neuromotor deficits, dopamine depletion and oxidative stress in a Drosophila melanogaster model of Parkinson's disease induced by rotenone. Neurotoxicology 51:96–105. https://doi.org/10.1016/j.neuro.2015.09.003
Auluck PK, Chan HE, Trojanowski JQ, Lee VM, Bonini NM (2002) Chaperone suppression of α-synuclein toxicity in a Drosophila model for Parkinson's disease. Science 295(5556):865–868. https://doi.org/10.1126/science.1067389
Barres B, Corio-Costet MF, Debieu D, Délye C, Fillinger-David S, Grosman J, Micoud A, Siegwart M, Walker AS (2016) Trends and challenges in pesticide resistance detection. Trends Plant Sci 21(10):834–853. https://doi.org/10.1016/j.tplants.2016.06.006
Bonilla E, Contreras R, Medina-Leendertz S, Mora M, Villalobos V, Bravo Y (2012) Minocycline increases the life span and motor activity and decreases lipid peroxidation in manganese treated Drosophila melanogaster. Toxicology 294(1):50–53. https://doi.org/10.1016/j.tox.2012.01.016
Bonilla E, Medina-Leendertz S, Villalobos V, Molero L, Bohorquez A (2006) Paraquat-induced oxidative stress in Drosophila melanogaster: effects of melatonin, glutathione, serotonin, minocycline, lipoic acid and ascorbic acid. Neurochem Res 31(12):1425–1432. https://doi.org/10.1007/s11064-006-9194-8
Broughton A (1990) Chronic health effects and immunological alternations associated with exposures to pesticide. Comments Toxicol 4:59–71
Büyükgüzel E (2009) Evidence of oxidative and antioxidative responses by galleria mellonella larvae to malathion. J Econ Entomol 102(1):152–159. https://doi.org/10.1603/029.102.0122
Büyükgüzel K (2006) Malathion-induced oxidative stress in a parasitoid wasp: effect on adult emergence, longevity, fecundity, and oxidative and antioxidative response of Pimpla turionellae (Hymenoptera: Ichneumonidae). J Econ Entomol 99(4):1225–1234. https://doi.org/10.1603/029.102.0122
Chitra KC, Abdu SU (2013) Impact of Malathion on the biochemical composition of the freshwater fish, Oreochromis mossambicus. Int J Zoo Res 3:31–42
Cochemé HM, Murphy MP (2008) Complex I is the major site of mitochondrial superoxide production by paraquat. J Biol Chem 283(4):1786–1798. https://doi.org/10.1074/jbc.M708597200
Costa MD, Freitas ML, Soares FA, Carratu VS, Brandão R (2011) Potential of two new oximes in reactivate human acetylcholinesterase and butyrylcholinesterase inhibited by organophosphate compounds: an in vitro study. Toxicol in Vitro 25(8):2120–2123. https://doi.org/10.1016/j.tiv.2011.09.018
Cui X, Wang L, Zuo P, Han Z, Fang Z, Li W, Liu J (2004) D-galactose-caused life shortening in Drosophila melanogaster and Musca domestica is associated with oxidative stress. Biogerontology 5(5):317–325
Dang K, Doggett SL, Singham GV, Lee CY (2017) Insecticide resistance and resistance mechanisms in bed bugs, Cimex spp.(Hemiptera: Cimicidae). Parasit. Vectors 10(1):318. https://doi.org/10.1186/s13071-017-2232-3
Das S (2013) A review of dichlorvos toxicity in fish. Curr. World environ 8(1):143. https://doi.org/10.12944/CWE.8.1.08
de Aguiar LM, Figueira FH, Gottschalk MS, da Rosa CE (2016) Glyphosate-based herbicide exposure causes antioxidant defence responses in the fruit fly Drosophila melanogaster. Comp. Biochem. Physiol., part C: Toxicol. Pharmacol. 185:94–101. https://doi.org/10.1016/j.cbpc.2016.03.006
Esterbauer H and Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. In methods in enzymology (Vol. 186, pp. 407-421). Academic press
Feany MB, Bender WW (2000) A Drosophila model of Parkinson's disease. Nature 404(6776):394–398
Giri S, Prasad SB, Giri A, Sharma GD (2002) Genotoxic effects of malathion: an organophosphorus insecticide, using three mammalian bioassays in vivo. Mutat Res 514(1–2):223–231. https://doi.org/10.1016/S1383-5718(01)00341-2
Heinrichsen ET, Zhang H, Robinson JE, Ngo J, Diop S, Bodmer R, Joiner WJ, Metallo CM, Haddad GG (2014) Metabolic and transcriptional response to a high-fat diet in Drosophila melanogaster. Mol Metab 3(1):42–54. https://doi.org/10.1016/j.molmet.2013.10.003
Hua J, Jones DK, Relyea RA (2014) Induced tolerance from a sublethal insecticide leads to cross-tolerance to other insecticides. Environ Sci Technol 48(7):4078–4085. https://doi.org/10.1021/es500278f
İçen E, Armutçu F, Büyükgüzel K, Gürel A (2005) Biochemical stress indicators of greater wax moth exposure to organophosphorus insecticides. J Econ Entomol 98(2):358–366. https://doi.org/10.1093/jee/98.2.358
Jadavji NM, Murray LK, Emmerson JT, Rudyk CA, Hayley S, Smith PD (2019) Paraquat exposure increases oxidative stress within the dorsal striatum of male mice with a genetic deficiency in one-carbon metabolism. Toxicol Sci 169(1):25–33. https://doi.org/10.1093/toxsci/kfz034
Jebali J, Banni M, Guerbej H, Almeida EA, Bannaoui A, Boussetta H (2006) Effects of malathion and cadmium on acetylcholinesterase activity and metallothionein levels in the fish Seriola dumerilli. Fish Physiol. Biochem 32(1):93. https://doi.org/10.1007/s10695-006-0041-2
Keifer MC, Firestone J (2007) Neurotoxicity of pesticides. J Agromed 12(1):17–25. https://doi.org/10.1300/J096v12n01_03
Kim K, Lawrence SM, Park J, Pitts L, Vann WF, Betenbaugh MJ, Palter KB (2002) Expression of a functional Drosophila melanogaster N-acetylneuraminic acid (Neu5Ac) phosphate synthase gene: evidence for endogenous sialic acid biosynthetic ability in insects. Glycobiology 12(2):73–83. https://doi.org/10.1093/glycob/12.2.73
Krůček T, Korandová M, Šerý M, Frydrychová RČ, Krůček T, Korandová M, Szakosová K (2015) Effect of low doses of herbicide paraquat on antioxidant defense in Drosophila. Arch Insect Biochem Physiol 88(4):235–248. https://doi.org/10.1002/arch.21222
Kumar V and Kumar P (2019). Pesticides in agriculture and environment: Impacts on human health. https://doi.org/10.26832/AESA-2019-CAE-0160-07
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mahmood I, Imadi SR, Shazadi K, Gul A and Hakeem KR (2016) Effects of pesticides on environment. In plant, soil and microbes (pp. 253-269). Springer, Cham
Mehdi SH, Qamar A (2011) Neuropathological studies of Drosophila melanogaster with sublethal doses of malathion. Biol and Med 3(2):265–269
Miller CS, Mitzel HC (1995) Chemical sensitivity attributed to pesticide exposure versus remodeling. Arch Environ Health 50(2):119–129. https://doi.org/10.1080/00039896.1995.9940889
Naqqash MN, Gökçe A, Bakhsh A, Salim M (2016) Insecticide resistance and its molecular basis in urban insect pests. Parasitol Res 115(4):1363–1373. https://doi.org/10.1007/s00436-015-4898-9
Ong C, Yung LY, Cai Y, Bay BH, Baeg GH (2015) Drosophila melanogaster as a model organism to study nanotoxicity. Nanotoxicology 9(3):396–403. https://doi.org/10.3109/17435390.2014.940405
Pandey KB, Mishra N, Rizvi SI (2010) Protein oxidation biomarkers in plasma of type 2 diabetic patients. Clin Biochem 43(4–5):508–511. https://doi.org/10.1016/j.clinbiochem.2009.11.011
Patil VK, David M (2013) Oxidative stress in freshwater fish, Labeo rohita as a biomarker of malathion exposure. Environ Monit Assess 185(12):10191–10199. https://doi.org/10.1007/s10661-013-3323-z
Possamai FP, Fortunato JJ, Feier G, Agostinho FR, Quevedo J, Wilhelm Filho D, Dal-Pizzol F (2007) Oxidative stress after acute and sub-chronic malathion intoxication in Wistar rats. Environ Toxicol Pharmacol 23(2):198–204. https://doi.org/10.1016/j.etap.2006.09.003
Randall C, Hock W, Crow E, Hudak-Wise C, Kasai J (2008) National pesticide applicator certification core manual. National Association of State Departments of Agriculture Research Foundation, Washington, DC, pp 149–162
Rashid B, Husnain T and Riazuddin S (2010) Herbicides and pesticides as potential pollutants: a global problem. In plant adaptation and phytoremediation (pp. 427-447). Springer, Dordrecht
Schmolke A, Thorbek P, Chapman P, Grimm V (2010) Ecological models and pesticide risk assessment: current modeling practice. Environ Toxicol Chem 29(4):1006–1012. https://doi.org/10.1002/etc.120
Shieh P, Jan CR, Liang WZ (2019) The protective effects of the antioxidant N-acetylcysteine (NAC) against oxidative stress-associated apoptosis evoked by the organophosphorus insecticide malathion in normal human astrocytes. Toxicology 417:1–14. https://doi.org/10.1016/j.tox.2019.02.004
Sifakis S, Mparmpas M, Soldin OP, Tsatsakis A (2011) Pesticide exposure and health related issues in male and female reproductive system. In Pesticides-Formulations, Effects, Fate. IntechOpen, Pesticide Exposure and Health Related Issues in Male and Female Reproductive System
Speck-Planche A, Kleandrova VV, Scotti MT (2012) Fragment-based approach for the in silico discovery of multi-target insecticides. Chemom Intell Lab Syst, 111(1), 39–45. https://doi.org/10.1016/j.chemolab.2011.11.010
Spyridaki MH, Siskos PA (1996) An improved spectrophotometric method for the determination of free, bound and total N-acetylneuraminic acid in biological fluids. Anal Chim Acta 327(3):277–285. https://doi.org/10.1016/0003-2670(96)00073-6
Sudati JH, Vieira FA, Pavin SS, Dias GR, Seeger RL, Golombieski R, Athayde ML, Soares FA, Rocha JB, Barbosa NV (2013) Valeriana officinalis attenuates the rotenone-induced toxicity in Drosophila melanogaster. Neurotoxicology 37:118–126. https://doi.org/10.1016/j.neuro.2013.04.006
Uversky VN (2004) Neurotoxicant-induced animal models of Parkinson’s disease: understanding the role of rotenone, maneb and paraquat in neurodegeneration. Cell Tissue Res 318(1):225–241. https://doi.org/10.1007/s00441-004-0937-z
Witko-Sarsat V, Friedlander M, Capeillère-Blandin C, Nguyen-Khoa T, Nguyen AT, Zingraff J, Jungers P, Descamps-Latscha B (1996) Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int 49(5):1304–1313. https://doi.org/10.1038/ki.1996.186
Wu H, Zhang Y, Shi X, Zhang J, Ma E (2017) Overexpression of Mn-superoxide dismutase in Oxya chinensis mediates increased malathion tolerance. Chemosphere 181:352–359. https://doi.org/10.1016/j.chemosphere.2017.04.087
Yadav IS and Devi NL (2017) Pesticides classification and its impact on human and environment. Environ Sci Engg, 6
Zhu KY (2008) Insecticide bioassay. Encyclopedia of Entomology:1974–1976. https://doi.org/10.1007/978-1-4020-6359-6_1541
Acknowledgements
We are thankful to Dr. Jasneet Grewal for her critical reading of the manuscript. Dr. Surender has provided a great help for the analyses of results. Department of Zoology, School of Bioengineering and Bioscience is highly acknowledged for providing funding for the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Tolerance against pesticides is observed when fruit flies are exposed to lower doses of Malathion and Paraquat
• Antioxidant defence system is observed to respond in affirmative manner against the exposure
• Few biomolecules are not affected by the oxidative stress produced
• Comparative longevity is increased in pesticide exposed fruit flies
• Total thiols are positively corroborated against the exposure
Rights and permissions
About this article
Cite this article
Mehdi, M.M., Layqat, M. & Majid, S. Biomolecular alterations by chronic sub-lethal exposure of Malathion and Paraquat in Drosophila melanogaster: study on pesticide tolerance in insects. Int J Trop Insect Sci 40, 869–876 (2020). https://doi.org/10.1007/s42690-020-00143-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-020-00143-6