Abstract
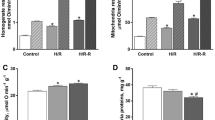
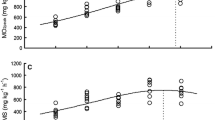
The effects of sex and training on mitochondrial function and its sensitivity to in vitro reactive oxygen species (ROS) exposure were investigated in male and female silver European eels. The critical swimming speed (Ucrit) was used as an index of training intensity and of overall swimming performance. After four days of training (10 h at 70% and 14 h at 50% of their Ucrit before training), Ucrit increased significantly in trained eels for both sexes. In untrained eels, basal mitochondrial respiration (V0) measured in the absence of ADP in permeabilized cardiac fibres was significantly higher and more resistant to in vitro ROS exposure in males than in females. No difference in ATP production (VATP) or hydroxyl radical release (\( {\mathrm{V}}_{\bullet_{\mathrm{OH}}} \)) was observed between male and female eels. In trained male eels, \( {\mathrm{V}}_{\bullet_{\mathrm{OH}}} \) decreased in red muscle fibres concomitantly to a trend in an increased ATP production. In trained female eels, a different trend in ATP decrease was observed. Training significantly enhanced respiratory rate Vmax and resistance to in vitro ROS exposure in cardiac fibres of male silver eels but not females. In male silver eels, but not females, training could induce an improvement of energy efficiency in red muscle and a greater resistance of mitochondrial function to ROS exposure in cardiac fibres. These beneficial effects could contribute to the highly efficient swimming performance of the male eel, which could serve to compensate their smaller size, helping them to achieve their transoceanic migration and synchronized arrival with the females at their reproduction area.




Similar content being viewed by others
References
Amérand A, Vettier A, Sébert P, Cann-Moisan C (2005) In vitro effect of hydrostatic pressure exposure on hydroxyl radical production in fish red muscle. Redox Rep 10:25–28
Amérand A, Vettier A, Moisan C et al (2010) Sex-related differences in aerobic capacities and reactive oxygen species metabolism in the silver eel. Fish Physiol Biochem 36:741–747
Aniagu SO, Day N, Chipman JK et al (2006) Does exhaustive exercise result in oxidative stress and associated DNA damage in the chub (Leuciscus cephalus)? Environ Mol Mutagen 47:616–623. https://doi.org/10.1002/em.20247
Birnie-Gauvin K, Costantini D, Cooke SJ, Willmore WG (2017) A comparative and evolutionary approach to oxidative stress in fish: A review. Fish Fish 18:928–942. https://doi.org/10.1111/faf.12215
Borrás C, Gambini J, López-Grueso R et al (2010) Direct antioxidant and protective effect of estradiol on isolated mitochondria. Biochim Biophys Acta (BBA) - Mol Basis Dis 1802:205–211. https://doi.org/10.1016/j.bbadis.2009.09.007
Boss O, Samec S, Desplanches D et al (1998) Effect of endurance training on mRNA expression of uncoupling proteins 1, 2, and 3 in the rat. FASEB J 12:335–339
Brand MD (2000) Uncoupling to survive? The role of mitochondrial inefficiency in ageing. Exp Gerontol 35:811–820
Brett JR (1964) The Respiratory Metabolism and Swimming Performance of Young Sockeye Salmon. J Fish Res Board Can 21:1183–1226. https://doi.org/10.1139/f64-103
Burgerhout E, Brittijn SA, Tudorache C et al (2013) Male European eels are highly efficient long distance swimmers: Effects of endurance swimming on maturation. Comp Biochem Physiol A Mol Integr Physiol 166:522–527. https://doi.org/10.1016/j.cbpa.2013.08.002
Castro V, Grisdale-Helland B, Helland SJ et al (2013) Cardiac Molecular-Acclimation Mechanisms in Response to Swimming-Induced Exercise in Atlantic Salmon. PLoS One 8:e55056. https://doi.org/10.1371/journal.pone.0055056
Davison W (1997) The effects of exercise training on teleost fish, a review of recent literature. Comp Biochem Physiol A Physiol 117:67–75
Egan B, Zierath JR (2013) Exercise Metabolism and the Molecular Regulation of Skeletal Muscle Adaptation. Cell Metab 17:162–184. https://doi.org/10.1016/j.cmet.2012.12.012
Farhat F, Dupas J, Amérand A et al (2014) Effect of exercise training on oxidative stress and mitochondrial function in rat heart and gastrocnemius muscle. Redox Rep 20:60–68. https://doi.org/10.1179/1351000214Y.0000000105
Farrell AP, Johansen JA, Steffensen JF et al (1990) Effects of exercise training and coronary ablation on swimming performance, heart size, and cardiac enzymes in rainbow trout, Oncorhynchus mykiss. Can J Zool 68:1174–1179. https://doi.org/10.1139/z90-174
Farrell AP, Johansen JA, Suarez RK (1991) Effects of exercise-training on cardiac performance and muscle enzymes in rainbow trout, Oncorhynchus mykiss. Fish Physiol Biochem 9:303–312. https://doi.org/10.1007/BF02265151
Fernström M, Tonkonogi M, Sahlin K (2004) Effects of acute and chronic endurance exercise on mitochondrial uncoupling in human skeletal muscle. J Physiol 554:755–763. https://doi.org/10.1113/jphysiol.2003.055202
Filho DW (2007) Reactive oxygen species, antioxidants and fish mitochondria. Front Biosci 12:1229. https://doi.org/10.2741/2141
Gamperl AK (2004) Cardiac plasticity in fishes: environmental influences and intraspecific differences. J Exp Biol 207:2539–2550. https://doi.org/10.1242/jeb.01057
Goglia F, Skulachev VP (2003) A function for novel uncoupling proteins: antioxidant defense of mitochondrial matrix by translocating fatty acid peroxides from the inner to the outer membrane leaflet. FASEB J 17:1585–1591. https://doi.org/10.1096/fj.03-0159hyp
Gomes EC, Silva AN, de Oliveira MR (2012) Oxidants, Antioxidants, and the Beneficial Roles of Exercise-Induced Production of Reactive Species. Oxidative Med Cell Longev 2012:1–12. https://doi.org/10.1155/2012/756132
He F, Li J, Liu Z et al (2016) Redox mechanism of reactive oxygen species in exercise. Front Physiol 7. https://doi.org/10.3389/fphys.2016.00486
Leichtweis SB, Leeuwenburgh C, Parmelee DJ et al (1997) Rigorous swim training impairs mitochondrial function in post-ischaemic rat heart. Acta Physiol Scand 160:139–148
Lumini-Oliveira J, Magalhães J, Pereira CV et al (2009) Endurance training improves gastrocnemius mitochondrial function despite increased susceptibility to permeability transition. Mitochondrion 9:454–462. https://doi.org/10.1016/j.mito.2009.08.005
Methling C, Tudorache C, Skov PV, Steffensen JF (2011) Pop Up Satellite Tags Impair Swimming Performance and Energetics of the European Eel (Anguilla anguilla). PLoS One 6:e20797. https://doi.org/10.1371/journal.pone.0020797
Misra HP, Fridovich I (1972) The Role of Superoxide Anion in the Autoxidation of Epinephrine and a Simple Assay for Superoxide Dismutase. J Biol Chem 247:3170–3175
Mortelette H, Amérand A, Sébert P et al (2010a) Effect of exercise training on respiration and reactive oxygen species metabolism in eel red muscle. Respir Physiol Neurobiol 172:201–205. https://doi.org/10.1016/j.resp.2010.05.023
Mortelette H, Moisan C, Sébert P et al (2010b) Fish as a model in investigations about the relationship between oxygen consumption and hydroxyl radical production in permeabilized muscle fibers. Mitochondrion 10:555–558
Moyes CD, Mathieu-Costello OA, Brill RW, Hochachka PW (1992) Mitochondrial metabolism of cardiac and skeletal muscles from a fast (Katsuwonus pelamis) and a slow (Cyprinus carpio) fish. Can J Zool 70:1246–1253. https://doi.org/10.1139/z92-172
Palstra AP, van den Thillart GEEJM (2010) Swimming physiology of European silver eels (Anguilla anguilla L.): energetic costs and effects on sexual maturation and reproduction. Fish Physiol Biochem 36:297–322. https://doi.org/10.1007/s10695-010-9397-4
Palstra A, van Ginneken V, van den Thillart G (2008) Cost of transport and optimal swimming speed in farmed and wild European silver eels (Anguilla anguilla). Comp Biochem Physiol A Mol Integr Physiol 151:37–44. https://doi.org/10.1016/j.cbpa.2008.05.011
Quintella BR, Mateus CS, Costa JL et al (2010) Critical swimming speed of yellow- and silver-phase European eel (Anguilla anguilla, L.): Critical swimming speed of yellow- and silver-phase European eel. J Appl Ichthyol 26:432–435. https://doi.org/10.1111/j.1439-0426.2010.01457.x
Ross SW, Dalton DA, Kramer S, Christensen BL (2001) Physiological (antioxidant) responses of estuarine fishes to variability in dissolved oxygen. Comp Biochem Physiol C Toxicol Pharmacol 130:289–303. https://doi.org/10.1016/S1532-0456(01)00243-5
Saks VA, Veksler VI, Kuznetsov AV et al (1998) Permeabilized cell and skinned fiber techniques in studies of mitochondrial function in vivo. In: Saks VA, Ventura-Clapier R, Leverve X et al (eds) Bioenergetics of the cell: quantitative aspects. Springer, Boston, pp 81–100
Scaion D, Sébert P (2008) Glycolytic fluxes in European silver eel, Anguilla anguilla: Sex differences and temperature sensitivity. Comp Biochem Physiol A Mol Integr Physiol 151:687–690. https://doi.org/10.1016/j.cbpa.2008.08.016
Scaion D, Belhomme M, Sébert P (2008) Pressure and temperature interactions on aerobic metabolism of migrating European silver eel. Respir Physiol Neurobiol 164:319–322. https://doi.org/10.1016/j.resp.2008.08.001
Sebert P (2013) Sex differences in energy metabolism. In: Trischitta F, TakeiY, Sebert P (eds) Eel physiology. CRC Press, Boca Raton, pp 98–117
Sébert P, Theron M (2001) Why can the eel, unlike the trout, migrate under pressure. Mitochondrion 1:79–85. https://doi.org/10.1016/S1567-7249(01)00009-5
Sfakianakis DG, Kentouri M (2010) Effect of temperature on muscle lactate metabolic recovery in sea bass (Dicentrarchus labrax, L.) juveniles exposed to exhaustive exercise. Fish Physiol Biochem 36:387–390. https://doi.org/10.1007/s10695-009-9305-y
Steinbacher P, Eckl P (2015) Impact of Oxidative Stress on Exercising Skeletal Muscle. Biomolecules 5:356–377. https://doi.org/10.3390/biom5020356
Steinhausen MF, Steffensen JF, Andersen NG (2005) Tail beat frequency as a predictor of swimming speed and oxygen consumption of saithe (Pollachius virens) and whiting (Merlangius merlangus) during forced swimming. Mar Biol 148:197–204. https://doi.org/10.1007/s00227-005-0055-9
Tesch F-W, Thorpe JE (2003) The eel. Blackwell Science, Oxford
Trischitta F, Takei Y, Sébert P (2013) Eel Physiology. CRC Press, Boca Raton
van Ginneken V, Dufour S, Sbaihi M et al (2007) Does a 5500-km swim trial stimulate early sexual maturation in the European eel (Anguilla anguilla L.)? Comp Biochem Physiol A Mol Integr Physiol 147:1095–1103. https://doi.org/10.1016/j.cbpa.2007.03.021
Vettier A, Sébert P (2004) Pressure resistance of aerobic metabolism in eels from different water environments. Mitochondrion 3:347–354. https://doi.org/10.1016/j.mito.2004.02.007
Walsh B, Tonkonogi M, Sahlin K (2001) Effect of endurance training on oxidative and antioxidative function in human permeabilized muscle fibres. Pflugers Arch 442:420–425. https://doi.org/10.1007/s004240100538
Yamamoto T, Ohkuwa T, Itoh H et al (2002) Effect of gender differences and voluntary exercise on antioxidant capacity in rats. Comp Biochem Physiol C Toxicol Pharmacol 132:437–444
Acknowledgements
We would like to thank Helen McCombie-Boudry of the “Bureau de Traduction de l’Université” (BTU) of Université de Bretagne Occidentale, Brest, France for her assistance with the improvement of the English of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farhat, F., Simon, B., Amérand, A. et al. Exercise training changes mitochondrial function and its vulnerability to reactive oxygen species exposure differently in male and female silver European eels. Environ Biol Fish 103, 363–375 (2020). https://doi.org/10.1007/s10641-020-00962-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-020-00962-z