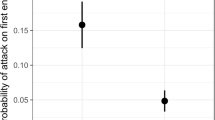
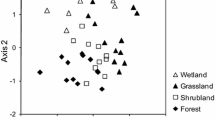
Abstract
Much of our understanding of mutualism is derived from pairwise obligate interactions between specialized participants. These observations may not adequately inform conservation efforts for facultative mutualist species, especially for areas in which invasive species are present and affect the mutualism. We evaluate the effects of ant attendance on larval survival for the endangered Fender’s blue butterfly (Icaricia icarioides fenderi Macy) in situ and examine the effects of biotic and abiotic factors—specifically ant community composition and microhabitat structure—on this mutualistic interaction in Oregon, USA. Fender’s blue larval survival was as much as three times higher in plots with a high proportion of ant attendance compared to plots in which larvae were rarely tended, and ant recruitment was reduced by dense, invasive European grasses. Thatch from these dense invasives, specifically tall oatgrass (Arrhenatherum elatius Beauv.) and tall fescue (Schedonorus arundinaceus Schreb.), are a threat to larval survival. In addition, variation in microhabitat structure determined which ant species occurred in our plots. Larvae were associated with ten ant species but primarily tended by only two: Prenolepis imparis Say and Aphaenogaster occidentalis Emery, and we observed no difference in butterfly larval survival between the two predominant ant species. Conservation efforts for this species would benefit from actions that reduce invasive grasses and facilitate ant tending interactions. Our study demonstrates how invasive grasses and forbs in degraded prairie systems can negatively impact the conservation of an at-risk species by disrupting an important mutualistic interaction.





Similar content being viewed by others
References
Axén AH (2000) Variation in behavior of lycaenid larvae when attended by different ant species. Evol Ecol 14:611–625
Bächtold A, Alves-Silva E, Kaminski LA, Del-Claro K (2014) The role of tending ants in host plant selection and egg parasitism of two facultative myrmecophilous butterflies. Naturwissenschaften 101:913–919
Ballmer GR, Pratt GF (1988) A survey of the last instar larvae of the Lycaenidae (Lepidoptera) of California. J Res Lepid 27:1–81
Barton K (2018) MuMIn: Multi-model inference. R package version 1(40):4
Bascompte J (2009) Mutualistic networks. Front Ecol Environ 7:429–436
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Bennion LD, Ferguson JA, New L, Schultz CB (2020) Community-level effects of herbicide-based restoration treatments: structural benefits but at what cost? Restor Ecol. https://doi.org/10.1111/rec.13118
Beyer LJ, Schultz CB (2010) Oviposition selection by a rare grass skipper Polites mardon in montane habitats: advancing ecological understanding to develop conservation strategies. Biol Conserv 143:862–872
Billick I, Tonkel K (2003) The relative importance of spatial vs. temporal variability in generating a conditional mutualism. Ecology 84:289–295
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Henry M, Stevens H, White JS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evolut 24:127–135
Bonney S, Andersen A, Schlesinger C (2017) Biodiversity impacts of an invasive grass: ant community responses to Cenchrus ciliaris in arid Australia. Biol Invasions 19:57–72
DeVries PJ (1991) Call production by myrmecophilous riodinid and lycaenid butterfly caterpillars (Lepidoptera): morphological, acoustical, functional, and evolutionary patterns. Am Mus Novit 3025:1–23
Duarte M, de Almeida GL, Casagrande MM, Mielke OHH (2001) Notes on the last instar larva and pupa of Hemiargus hanno (Stoll) (Lepidoptera, Lycaenidae, Polyommatinae). Rev Bras Zool 18:1097–1105
Eastwood R, Fraser AM (1999) Associations between lycaenid butterflies and ants in Australia. Aust J Ecol 24:503–537
Fiedler K (1991) Systematic, evolutionary, and ecological implications of myrmecophily within the Lycaenidae (Insecta: Lepidoptera: Papilionoidea). Bonn Zool Monogr 31:1–210
Fiedler K (2006) Ant-associates of Palaearctic lycaenid butterfly larvae (Hymenoptera: Formicidae; Lepidoptera: Lycaenidae)—a review. Myrmecol News 9:77–87
Fiedler K, Maschwitz U (1988) Functional analysis of the myrmecophilous relationships between ants (Hymenoptera: Formicidae) and lycaenids (Lepidoptera: Lycaenidae). II. Lycaenid larvae as trophobiotic partners of ants: a quantitative approach. Oecologia 75:204–206
Fiedler K, Saam C (1995) Ants benefit from attending facultatively myrmecophilous Lycaenidae caterpillars: evidence from a survival study. Oecologia 104:316–322
Fiedler K, Hölldobler B, Seufert P (1996) Butterflies and ants: the communicative domain. Experientia 52:14–24
Fraser AM, Axén AH, Pierce NE (2001) Assessing the quality of different ant species as partners of a myrmecophilous butterfly. Oecologia 129:452–460
Friendly M, Fox J (2017) Candisc: visualizing generalized canonical discriminant and canonical correlation analysis. R package version 0.8-0.
Gammans N, Drummond F, Groden E (2018) Impacts of the invasive European Red Ant (Myrmica rubra (L.): Hymenoptera; Formicidae) on a myrmecochorous system in the northeastern United States. Environ Entomol 47:908–917
Glaeser RM, Schultz CB (2014) Characterizing a contentious management tool: the effects of a grass-specific herbicide on the silvery blue butterfly. J Insect Conserv 18:1047–1058
Gnatzy W, Jatho M, Kleinteich T, Gorb SN, Hustert R (2017) The eversible tentacle organs of Polyommatus caterpillars (Lepidoptera, Lycaenidae): Morphology, fine structure, sensory supply and functional aspects. Arthropod Struct Dev 46:788–804
Hays DW, Potter AE, Thompson CW, Dunn PV (2000) Critical habitat components for four rare South Puget Sound grassland butterflies. A report prepared for the Washington Department of Fish and Wildlife and The Nature Conservancy of Washington, Seattle.
Kaminski LA, Rodrigues D (2011) Species-specific levels of ant attendance mediate performance costs in a facultative myrmecophilous butterfly. Physiol Entomol 36:208–214
Kjar D, Park Z (2016) Increased ant (Hymenoptera: Formicidae) incidence and richness are associated with alien plant cover in a small mid-Atlantic riparian forest. Myrmecol News 22:109–117
LaBar CC, Schultz CB (2012) Investigating the role of herbicides in controlling invasive grasses in prairie habitats: effects on non-target butterflies. Nat Areas J 32:177–189
Lane CP (1999) Benefits of heterogeneous habitat: oviposition preference and immature performance of Lycaeides melissa samuelis Nabokov (Lepidoptera: Lycaenidae). PhD dissertation. University of Minnesota: St. Paul.
Lane CP, Andow DA (2003) Oak savannah subhabitat variation and the population biology of Lycaeides melissa samuelis (Lepidoptera: Lycaenidae). Ann Entomol Soc Am 96:799–809
LeVan KE, Barney SK, Wilson Rankin EE (2018) Introduced ants reduce interaction diversity in a multi-species ant-aphid mutualism. Oikos 127:1132–1141
Lindzey S, Connor EF (2011) Monitoring the mission blue butterfly using immature stages. J Insect Conserv 15:765–773
Longino JT, Coddington J, Colwell RK (2002) The ant fauna of a tropical rainforest: estimating species richness three different ways. Ecology 83:689–702
Maxwell JA (1998) The conservation of the Karner blue butterfly (Lycaeides melissa samuelis Nobokov): ecological studies on habitat creation and management. PhD dissertation. Department of Entomology; University of Wisconsin: Madison.
Murata K, Matsuura A (2013) Effect of grazing intensity on the interaction between Shijimiaeoides divinus asonis (Matsumura, 1929) and its attendant ants. Entomol Sci 16:390–399
New TR (2014) Insects, fire and conservation. Springer International Publishing, Basel
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2018) Vegan: community ecology package. R package version 2.4–6.
Pascale EG, Thiet RK (2016) The relationship between ants and Lycaeides melissa samuelis (Lepidoptera: Lycaenidae) at Concord Pine Barrens, NH, USA. Environ Entomol 45:633–641
Pellissier L, Litsios G, Fiedler K, Pottier J, Dubuis A, Pradervand J, Salamin N, Guisan A (2012) Loss of interactions with ants under cold climate in a regional myrmecophilous butterfly fauna. J Biogeogr 39:1782–1790
Peterson MA (1993) The nature of ant attendance and the survival of larval Icaricia acmon (Lycaenidae). J Lepid Soc 47:8–16
Peterson MA (1995) Unpredictability in the facultative association between larvae of Euphilotes enoptes (Lepidoptera, Lycaenidae) and ants. Biol J Linnean Soc 55:209–233
Pierce NE, Easteal S (1986) The selective advantage of attendant ants for larvae of a lycaenid butterfly, Glaucopsyche lygdamus. J Anim Ecol 55:451–462
Pierce NE, Braby MF, Heath A, Lohman DJ, Mathew J, Rand DB, Travassos MA (2002) The ecology and evolution of ant association in the Lycaenidae (Lepidoptera). Annu Rev Entomol 47:733–771
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Russell C, Schultz CB (2010) Effects of grass-specific herbicides on butterflies: an experimental investigation to advance conservation efforts. J Insect Conserv 14:53–63
Ruxton GD, Neuhäuser MN (2010) Good practice in testing for an association in contingency tables. Behav Ecol Sociobiol 64:1505–1513
Saarinen EV, Daniels JC (2006) Miami blue butterfly larvae (Lepidoptera: Lycaenidae) and ants (Hymenoptera: Formicidae): new information on the symbionts of an endangered taxon. Fla Entomol 89:69–74
Savignano DA (1994) Benefits to Karner blue butterfly larvae from association with ants. In: Andow DA, Baker RJ, Lane CP (eds.) Karner blue butterfly: a symbol of vanishing landscape. University of Minnesota, Minneapolis.
Scheiner SM, Gurevitch J (2001) Design and analysis of ecological experiments. Oxford University Press, Oxford
Schultz CB, Crone EE (1998) Burning prairie to restore butterfly habitat? A modeling approach for management tradeoffs for the Fender’s blue. Restor Ecol 6:244–252
Schultz CB, Hammond PC, Wilson MV (2003) Biology of the Fender’s blue butterfly (Icaricia icarioides fenderi Macy), an endangered species in of western Oregon native prairies. Nat Area J 23:61–71
Schultz CB, Henry E, Carleton A, Hicks T, Thomas R, Potter A, Collins M, Linders M, Fimbel C, Black S, Anderson HE, Diehl G, Hamman S, Gilbert R, Foster J, Hays D, Wilderman D, Davenport R, Steel E, Page N, Lilley PL, Heron J, Kroeker N, Webb C, Reader B (2011) Conservation of prairie-oak butterflies in Oregon, Washington, and British Columbia. BioOne 85:361–388
Stanley AG, Dunwiddie PW, Kaye TN (2011) Restoring invaded Pacific Northwest prairies: management recommendations from a region-wide experiment. Northwest Sci 85:233–246
Therneau T (2015) A package for survival analysis in S. R package version 2:38
Thomas RC, Schultz CB (2016) Resource selection in an endangered butterfly: females select native nectar species. J Wildl Manag 80:171–180
Thomas JA, Simcox DJ, Clarke RT (2009) Successful conservation of a threatened Maculinea butterfly. Science 325:80–83
Trager MD, Daniels JC (2009) Ant tending of Miami blue butterfly larvae (Lepidoptera: Lycaenidae): partner diversity and effects on larval performance. Fla Entomol 92:474–482
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. New York, Springer
Wagner D, del Rio CM (1997) Experimental tests of the mechanism for ant-enhanced growth in an ant-tended lycaenid butterfly. Oecologia 112:424–429
Warchola N, Bastianelli C, Schultz CB, Crone EE (2015) Fire increases ant-tending and survival of the Fender’s blue butterfly larvae. J Insect Conserv 19:1063–1073
Weeks JA (2003) Parasitism and ant protection alter the survival of the lycaenid Hemiargus isola. Ecol Entomol 28:228–232
Williams BK (1983) Some observations of the use of discriminant analysis in ecology. Ecology 64:1283–1291
Winfree R, MacLeod M, Harrison T, Cariveau DP (2015) Conserving and restoring mutualisms. In: Bronstein JL (ed.) Mutualism. Oxford University Press, Oxford.
Zuur AF (2012) A beginner’s guide to generalized additive models with R. Highland Statistics Ltd., Newburgh
Acknowledgements
We thank W. Messinger (Army Core of Engineers), S. Villegas-Moore (Bureau of Land Management), and M. Benotsch (The Nature Conservancy) for logistical support and permitting us to work on their land. We are also grateful to J. L. Smokey, A. B. Thomas, J. A. Ferguson, and many others for assistance in the field, with pitfall sampling, and with sorting specimens; L. F. New for advice on analyses; and R. E. Bonoan for valuable comments on a draft of this manuscript. This work was funded by the US Bureau of Land Management, the DeWind Award from the Xerces Society for Invertebrate Conservation, the Linfield College Student-Faculty Collaborative Research Grant, the Jane Clair Dirks-Edmunds Research Fellowship, and Washington State University.
Author information
Authors and Affiliations
Contributions
CCT, CVT, and CBS conceived and designed the experiments and CBS secured funding. CVT conducted the pitfall sampling fieldwork; CCT conducted all other fieldwork. CCT and CVT identified specimens collected in the study. CCT analyzed the data and wrote the manuscript; CBS and CVT edited drafts of the manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thomas, C.C., Tillberg, C.V. & Schultz, C.B. Facultative mutualism increases survival of an endangered ant-tended butterfly. J Insect Conserv 24, 385–395 (2020). https://doi.org/10.1007/s10841-020-00218-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-020-00218-2