Abstract
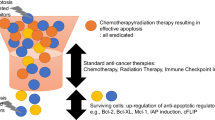
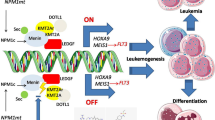
The successful use of PARP1 inhibitors like olaparib (Loparza®) in the treatment of BRCA1/2-deficient breast cancer has provided clinical proof-of-concept for applying personalized medicine based on synthetic lethality to the treatment of cancer. Unfortunately, all marketed PARP1 inhibitors act by competing with the cofactor NAD+ and resistance is already developing to this anticancer mechanism. Allosteric PARP1 inhibitors could provide a means of overcoming this resistance. A high throughput screen performed by Tulin et al. identified 5F02 as an allosteric PARP inhibitor that acts by preventing the enzymatic activation of PARP1 by histone H4. 5F02 demonstrated anticancer activity in several cancer cell lines and was more potent than olaparib and synergistic with olaparib in these assays. In the present study, we explored the structure–activity relationship of 5F02 by preparing analogs that possessed structural variation in four regions of the chemical scaffold. Our efforts led to lead molecule 7, which demonstrated potent anti-clonogenic activity against BRCA-deficient NALM6 leukemia cells in culture and a therapeutic index for the BRCA-deficient cells over their BRCA-proficient isogenic counterparts.






Similar content being viewed by others
References
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zeigler K, Guldberg P, Sehested M, Nesland JM, Lukas C, Orntoft T, Lkas J, Bartek J (2005) DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434:864–870
Dasgupta Y, Golovine K, Nieborowska-Skorska M (2017) Drugging DNA repair to target T-ALL cells. Leuk Lymphoma 59:1746–1749
Fujita M, Sasanuma H, Yamamoto KN, Harada H, Kurosawa A, Adachi N, Omura M, Hiraoka M, Takeda S, Hirota K (2013) Inerference in DNA replication can cause mitotic chromosomal breakage unassociated with double-strand breaks. PLoS ONE 8:e60043
Jain PG, Patel BD (2019) Medicinal chemistry approaches of poly ADP-ribose polymerase 1 (PARP1) inhibiors as anticancer agents—a recent update. Eur J Med Chem 165:198–215
Jin L, Wang W, Fang G (2014) Targeting protein-protein interaction by small molecules. Ann Rev Pharmacol Toxicol 54:435–456
Karanam K, Kafri R, Loewer A, Layay G (2012) Quantitative live cell imaging reveals a gradual shift between DNA repair mechanisms and a maximal use of HR in mid S phase. Mol Cell 47:320–329
Karpova Y, Wu C, Divan A, McDonnell ME, Hewlett E, Makhov P, Gordon J, Ye M, Reitz AB, Childers WE, Skorski T, Kolenko V, Tulin AV (2019) Non-NAD-like PARP-1 inhibitors in prostate cancer treatment. Biochem Pharmacol 167:149–162
Kirsanov KI, Kotova E, Makhov P, Golovine K, Lesovaya EA, Kolenko VM, Yakubovskaya MG, Tulin AV (2014) Minor groove binding ligands disrupt PARP-1 activation pathways. Oncotarget 5:428–437
Koslov NS, Kovaleva VN, Vecher EI, Boreiko EI, Vladyko GV, Korobchenko LV (1989) Synthesis and antiviral properties of cyclohexanol amino esters. Vestsi Akadėmii navuk BSSR. Seryîa khimichnykh navuk 2::66–68
Lewin AH, Frucht M (1975) Restricted rotationin amides VII. Methods of resonance assignment in tertiary amides—an evaluation. Org Mag Res 7:26–225
Lord CJ, Tutt AN, Ashworth A (2015) Synthetic lethality and cancer therapy: lessons learned from the development of PARP inhibitors. Annu Rev Med 66:455–470
Maifrede S, Martin K, Podszywalow-Bartnicka P, Sullivan-Reed K, Langer SK, Nejati R, Dasgupta Y, Hulse M, Gritsyuk D, Nieborowska-Skorska M, Lupey-Green LN, Zhao H, Piwocka K, Wasik MA, Tempera I, Skorski T (2017) Mol Cancer Res 15:967–972
Malyuchenko NV, Lotova EY, Kulaeva OI, Kirpichnikov MP, Studitskiy VM (2015) PARP1 inhibitors: antitumor drug design. Acta Nat 7:27–37
Metzger MJ, Stoddard BL, Monnat Jr RJ (2013) PARP-mediated repair, homologous recombination, and back-up no-homologous end joining-like repair of single-strand breaks. DNA Repair 12:529–534
Nickoloff JA, Jones D, Lee S-K, Williamson EA, Hromas R (2017) Drugging the cancers addicted to DNA repair. J Natl Can Inst 109:djx059
Nieborowska-Skorska M, Maifrede S, Ye M, Toma M, Hewlett E, Gordon J, Le BV, Sliwinski T, Zhao H, Piwocka K, Valent P, Tulin AV, Childers W, Skorski T (2019) Non-NAD-like PARP1 inhibitors enhanced synthetic lethal effect of NAD-like PARP inhibitors against BRCA1-deficient leukemia. Leuk Lymphoma 60:1098–1101
Nieborowska-Skorska M, Sullivan K, Dasgupta Y, Podszywalow-Bartnicka P, Hosner G, Maifrede S, Martinez E, Di Marcantonio D, Bolton-Gillespie E, Cramer-Morales K, Lee J, Li M, Slupianek A, Gritsyuk D, Cerny-Reiterer S, Seferynska I, Stoklosa T, Bullinger L, Zhao H, Gorbunova V, Piwocka K, Valent P, Civin CI, Muschen M, Dick JE, Want JC, Bhatia S, Bhatia R, Eppert K, Minden MD, Sykes SM, Skorski T (2017) Gene expression and mutation-guided synthetic lethality eradicates proliferating and quiescent leukemia cells. J Clin Investig 127:2392–2406
Schenker PK, Gunthard HH (1952) Carbon rings LX. The ten-membered ring. V. Helv Chem Acta 35:1598–1615
Sullivan-Reed K, Bolton-Gillespie E, Dasgupta Y, Langer S, Siciliano M, Nieborowska-Skorska M, Hanamshet K, Belyaeva EA, Bernhardy AJ, Lee J, Moore M, Zhao H, Valent P, Metlawska-Wasowska K, Muschen M, Bhatia S, Bhatia R, Johnson N, Wasik MA, Mazin AV, Skorski T (2018) Simultaneous targeting of PARP1 and RAD52 triggers dual synthetic lethality in BRCA-deficient tumor cells. Cell Rep 23:3127–3136
Thomas C, Ji Y, Lodhi N, Kotova E, Pinnola AD, Golovine K, Makhov P, Pechenkina K, Kolenko V, Tulin AV (2016) Non-NAD-like poly(ADP-ribose) polymerase-1 inhibitors effectively eliminate cancer in vivo. EBioMedicine 13:90–98
Ying S, Hamdy FC, Helleday T (2012) Mre11-dependent degradation of stalled DNA replication forks is prevented by BRCA2 and PARP1. Cancer Res 72:2814–2821
Acknowledgements
The authors would like to thank Dr Furong Sun and the staff at the Mass Spectrometry Lab, School of Chemical Sciences, University of Illinois at Urbana-Champaign for providing the high resolution mass spectrometry data.
Funding
This work was funded by the National Institutes of Health/National Cancer Institute under R01 CA186238 to TS. MT was supported by an Etiuda6 scholarship awarded to her by the Polish National Science Centre.
Author information
Authors and Affiliations
Contributions
EH and MT contributed equally to the paper. EH designed and synthesized the molecules described in the paper and contributed to the preparation of the paper. MT performed pharmacological experiments, prepared figures and contributed to the preparation of the paper. KS-R performed pharmacological experiments. JG performed the in vitro ADME studies. TS oversaw the efforts of MT. WEC supervised the medicinal chemistry efforts, helped design the molecules described in the paper and revised the paper. TS supervised the pharmacology efforts, designed the pharmacology protocols used in the paper and revised the paper. AT contributed to the preparation and revision of the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All experiments carried out in the course of this work were performed in compliance with OSHA regulations and under protocols that were approved by the Temple University Institutional Biosafety Committee.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary material
Rights and permissions
About this article
Cite this article
Hewlett, E., Toma, M., Sullivan-Reed, K. et al. Novel allosteric PARP1 inhibitors for the treatment of BRCA-deficient leukemia. Med Chem Res 29, 962–978 (2020). https://doi.org/10.1007/s00044-020-02537-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02537-0