Abstract
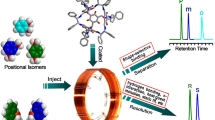
Porous organic cages (POCs) are an emerging class of porous molecular materials which self-assembled by discrete, shape-persistent, cage-like molecules and have recently received intensive interest in diverse fields. In this work, a hydroxyl-functionalized homochiral POC was synthesized by [4 + 6] condensation of 2-hydroxy-1,3,5-triformylbenzene with (1R, 2R)-diaminocyclohexane and coated on a capillary column for gas chromatography (GC) separations. Forty-one pairs of enantiomers belonging to various classes have been resolved on the column, including alcohols, diols, esters, lactones, halohydrocarbons, ethers, epoxides, ketones and sulfoxides. Compared with β-cyclodextrin derivative-based commercial β-DEX 120 column, previously reported chiral POCs- (CC9 and CC10) based columns, there are 12, 27 and 19 pairs of studied enantiomers cannot be resolved on β-DEX 120 column, CC9 column and CC10 column, respectively. Besides, both separation factors and resolution values of some racemates are higher on this column than those on β-DEX 120 column, CC9 column and CC10 column. The results demonstrate that the column exhibits good chiral recognition complementarity or superior chiral resolution capability to those columns which used for comparison, and the introduction of hydroxyl group enhances hydrogen-bonding interaction to some racemates, leading to better enantioselectivity. The column also shows excellent separation performance toward positional isomers of dichlorobenzene, dibromobenzene, nitrotoluene, chlorotoluene, nitrobromobenzene and nitrochlorobenzene. The retention time and selectivity of analytes have no significant changes after more than 500 injections and 260 °C conditioned for 6 h, showing the good repeatability and thermal stability of the column. This work indicates the promising prospect of POCs for GC separation, especially for the separation of enantiomers and also demonstrates the significance of design and synthesis of more functionalized chiral POCs for GC enantioseparation that broaden the enantiomer separation scope and applicability of POCs-based columns through their chiral recognition complementarities.





Similar content being viewed by others
References
Li H, Eddaoudi M, O'Keeffe M, Yaghi OM (1999) Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature 402:276–279
Côté AP, Benin AI, Ockwig NW, O’Keeffe M, Matzger AJ, Yaghi OM (2005) Porous, crystalline, covalent organic frameworks. Science 310:1166–1170
Wu DC, Xu F, Sun B, Fu RW, He HK, Matyjaszewski K (2012) Design and preparation of porous polymers. Chem Rev 112:3959–4015
Holst JR, Trewin A, Cooper AI (2010) Porous organic molecules. Nat Chem 2:915–920
Tian J, Thallapally PK, McGrail BP (2012) Porous organic molecular materials. CrystEngComm 14:1909–1919
McKeown NB (2010) Nanoporous molecular crystals. J Mater Chem 20:10588–10597
Mastalerz M (2010) Shape-persistent organic cage compounds by dynamic covalent bond formation. Angew Chem Int Ed 49:5042–5053
Zhang G, Mastalerz M (2014) Organic cage compounds-from shape-persistency to function. Chem Soc Rev 43:1934–1947
Jin Y, Zhu Y, Zhang W (2013) Development of organic porous materials through Schiff-base chemistry. CrystEngComm 15:1484–1499
Tozawa T, Jones JTA, Swamy SI, Jiang S, Adams DJ, Shakespeare S, Clowes R, Bradshaw D, Hasell T, Chong SY, Tang C, Thompson S, Parker J, Trewin A, Bacsa J, Slawin AMZ, Steiner A, Cooper AI (2009) Porous organic cages. Nat Mater 8:973–978
Mastalerz M, Schneider MW, Oppel IM, Presly O (2011) A salicylbisimine cage compound with high surface area and selective CO2/CH4 adsorption. Angew Chem Int Ed 50:1046–1051
Jin Y, Voss BA, Noble RD, Zhang W (2010) A shape-persistent organic molecular cage with high selectivity for the adsorption of CO2 over N2. Angew Chem Int Ed 49:6348–6351
Chen GJ, Xin WL, Wang JS, Cheng JY, Dong YB (2019) Visible-light triggered selective reduction of nitroarenes to azo compounds catalysed by Ag@organic molecular cages. Chem Commun 55:3586–3589
Yang X, Sun JK, Kitta M, Pang H, Xu Q (2018) Encapsulating highly catalytically active metal nanoclusters inside porous organic cages. Nat Catal 1:214–220
Mitra T, Jelfs KE, Schmidtmann M, Ahmed A, Chong SY, Adams DJ, Cooper AI (2013) Molecular shape sorting using molecular organic cages. Nat Chem 5:276–281
Zhang JH, Zhu PJ, Xie SM, Zi M, Yuan LM (2018) Homochiral porous organic cage used as stationary phase for open tubular capillary electrochromatography. Anal Chim Acta 999:169–175
Zhang C, Wang Q, Long H, Zhang W (2011) A highly C70 selective shape-persistent rectangular prism constructed through one-step alkyne metathesis. J Am Chem Soc 133:20995–21001
Chen L, Reiss PS, Chong SY, Holden D, Jelfs KE, Hasell T, Little MA, Kewley A, Briggs ME, Stephenson A, Thomas KM, Armstrong JA, Bell J, Busto J, Noel R, Liu J, Strachan DM, Thallapally PK, Cooper AI (2014) Separation of rare gases and chiral molecules by selective binding in porous organic cages. Nat Mater 13:954–960
Lu Z, Lu X, Zhong Y, Hu Y, Li G, Zhang R (2019) Carbon dot-decorated porous organic cage as fluorescent sensor for rapid discrimination of nitrophenol isomers and chiral alcohols. Anal Chim Acta 1050:146–153
Duan AH, Wang BJ, Xie SM, Zhang JH, Yuan LM (2017) A chiral, porous, organic cage-based, enantioselective potentiometric sensor for 2-aminobutanol. Chirality 29:172–177
McCaffrey R, Long H, Jin Y, Sanders A, Park W, Zhang W (2014) Template synthesis of gold nanoparticles with an organic molecular cage. J Am Chem Soc 136:1782–1785
Uemura T, Nakanishi R, Mochizuki S, Kitagawa S, Mizuno M (2016) Radical polymerization of vinyl monomers in porous organic cages. Angew Chem Int Ed 55:6443–6447
Tao ZR, Wu JX, Zhao YJ, Xu M, Tang WQ, Zhang QH, Gu L, Liu DH, Gu ZY (2019) Untwisted restacking of two-dimensional metal-organic framework nanosheets for highly selective isomer separations. Nat Commun 10:1–8
Gu ZY, Yan XP (2010) Metal-organic framework MIL-101 for high-resolution gas-chromatographic separation of xylene isomers and ethylbenzene. Angew Chem Int Ed 49:1477–1480
Huang J, Han X, Yang S, Cao Y, Yuan C, Liu Y, Wang J, Cui Y (2019) Microporous 3D covalent organic frameworks for liquid chromatographic separation of xylene isomers and ethylbenzene. J Am Chem Soc 141:8996–9003
Han X, Huang J, Yuan C, Liu Y, Cui Y (2018) Chiral 3D covalent organic frameworks for high performance liquid chromatographic enantioseparation. J Am Chem Soc 140:892–895
Zhang K, Cai SL, Yan YL, He ZH, Lin HM, Huang XL, Zheng SR, Fan J, Zhang WG (2017) Construction of a hydrazone-linked chiral covalent organic framework-silica composite as the stationary phase for high performance liquid chromatography. J Chromatogr A 1519:100–109
Lu CM, Liu SQ, Xu JQ, Ding YJ, Ouyang GF (2016) Exploitation of a microporous organic polymer as a stationary phase for capillary gas chromatography. Anal Chim Acta 902:205–211
Maya F, Cabello CP, Figuerola A, Palomino GT, Cerdà V (2019) Immobilization of metal-organic frameworks on supports for sample preparation and chromatographic separation. Chromatographia 82:361–375
Zhang JH, Zhang M, Xie SM, He PG, Yuan LM (2015) A novel inorganic mesoporous material with a nematic structure derived from nanocrystalline cellulose as the stationary phase for high-performance liquid chromatography. Anal Methods 7:3448–3453
Xie SM, Zhang ZJ, Wang ZY, Yuan LM (2011) Chiral metal-organic frameworks for high-resolution gas chromatographic separations. J Am Chem Soc 133:11892–11895
Zhang XH, Xie SM, Duan AH, Wang BJ, Yuan LM (2013) Separation performance of MOFs Zn(ISN)2·2H2O as stationary phase for high-resolution GC. Chromatographia 76:831–836
Qian HL, Yang CX, Yan XP (2016) Bottom-up synthesis of chiral covalent organic frameworks and their bound capillaries for chiral separation. Nat Commun 7:12104
Dong JQ, Liu Y, Cui Y (2014) Chiral porous organic frameworks for asymmetric heterogeneous catalysis and gas chromatographic separation. Chem Commun 50:14949–14952
Zhang JH, Xie SM, Zhang M, Zi M, He PG, Yuan LM (2014) Novel inorganic mesoporous material with chiral nematic structure derived from nanocrystalline cellulose for high-resolution gas chromatographic separations. Anal Chem 86:9595–9602
Zhang JH, Xie SM, Chen L, Wang BJ, He PG, Yuan LM (2015) Homochiral porous organic cage with high selectivity for the separation of racemates in gas chromatography. Anal Chem 87:7817–7824
Kewley A, Stephenson A, Chen L, Briggs ME, Hasell T, Cooper AI (2015) Porous organic cages for gas chromatography separations. Chem Mater 27:3207–3210
Xie SM, Zhang JH, Fu N, Wang BJ, Chen L, Yuan LM (2016) A chiral porous organic cage for molecular recognition using gas chromatography. Anal Chim Acta 903:156–163
Zhang JH, Xie SM, Wang BJ, He PG, Yuan LM (2015) Highly selective separation of enantiomers using a chiral porous organic cage. J Chromatogr A 1426:174–182
Zhang JH, Xie SM, Wang BJ, He PG, Yuan LM (2018) A homochiral porous organic cage with large cavity and pore windows for the efficient gas chromatography separation of enantiomers and positional isomers. J Sep Sci 41:1385–1394
Petryk M, Szymkowiak J, Gierczyk B, Spólnik G, Janiak A, Kwit M (2016) Chiral, triformylphenol-derived salen-type [4+6] organic cages. Org Biomol Chem 14(31):7495–7499
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 21705142, 91856123 and 21765025), the Applied Basic Research Foundation of Yunnan Province (No. 2019FB011) and University Student Innovation and Entrepreneurship Training Program of Yunnan Province (No. 208).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, HX., Xie, TP., Xie, SM. et al. Enantiomeric Separation on a Homochiral Porous Organic Cage-Based Chiral Stationary Phase by Gas Chromatography. Chromatographia 83, 703–713 (2020). https://doi.org/10.1007/s10337-020-03895-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-020-03895-y