Abstract
Key message
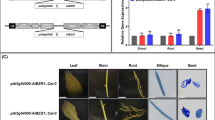
Seed-specific down-regulation of AtCESA1 and AtCESA9, which encode cellulose synthase subunits, differentially affects seed storage compound accumulation in Arabidopsis.
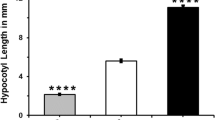
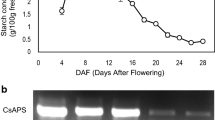
High amounts of cellulose can negatively affect crop seed quality, and, therefore, diverting carbon partitioning from cellulose to oil, protein and/or starch via molecular breeding may improve seed quality. To determine the effect of seed cellulose content reduction on levels of storage compounds, Arabidopsis thaliana CELLULOSE SYNTHASE1 (AtCESA1) and AtCESA9 genes, which both encode cellulose synthase subunits, were individually down-regulated using seed-specific intron-spliced hairpin RNA (hpRNAi) constructs. The selected seed-specific AtCESA1 and AtCESA9 Arabidopsis RNAi lines displayed reduced cellulose contents in seeds, and exhibited no obvious visual phenotypic growth defects with the exception of a minor effect on early root development in AtCESA1 RNAi seedlings and early hypocotyl elongation in the dark in both types of RNAi line. The seed-specific down-regulation of AtCESA9 resulted in a reduction in seed weight compared to empty vector controls, which was not observed in AtCESA1 RNAi lines. In terms of effects on carbon partitioning, AtCESA1 and AtCESA9 RNAi lines exhibited distinct effects. The down-regulation of AtCESA1 led to a ~ 3% relative increase in seed protein content (P = 0.04) and a ~ 3% relative decrease in oil content (P = 0.02), but caused no alteration in soluble glucose levels. On the contrary, AtCESA9 RNAi lines did not display a significant reduction in seed oil, protein or soluble glucose content. Taken together, our results indicate that the seed-specific down-regulation of AtCESA1 causes alterations in seed storage compound accumulation, while the effect of AtCESA9 on carbon partitioning is absent or minor in Arabidopsis.










Similar content being viewed by others
Abbreviations
- CESA:
-
CELLULOSE SYNTHASE
- AtCESA :
-
Arabidopsis CELLULOSE SYNTHASE
- hpRNAi:
-
Intron-spliced hairpin RNA
- PP2AA3 :
-
PROTEIN PHOSPHATASE 2A SUBUNIT 3
- FAME:
-
Fatty acid methyl esters
- DAF:
-
Days after flowering
- TAG:
-
Triacylglycerol
References
An D, Suh MC (2015) Overexpression of Arabidopsis WRI1 enhanced seed mass and storage oil content in Camelina sativa. Plant Biotechnol Rep 9:137–148. https://doi.org/10.1007/s11816-015-0351-x
Annison G, Choct M (2005) Anti-nutritive activities of cereal non-starch polysaccharides in broiler diets and strategies minimizing their effects. Worlds Poult Sci J 47:232–242. https://doi.org/10.1079/wps19910019
Arioli T, Peng L, Betzner AS et al (1998) Molecular analysis of cellulose biosynthesis in Arabidopsis. Science 279:717–720. https://doi.org/10.1016/S1369-5266(98)80058-7
Barratt DHP, Derbyshire P, Findlay K et al (2009) Normal growth of Arabidopsis requires cytosolic invertase but not sucrose synthase. Proc Natl Acad Sci USA 106:13124–13129. https://doi.org/10.1073/pnas.0900689106
Baud S, Dubreucq B, Miquel M et al (2008) Storage reserve accumulation in Arabidopsis: metabolic and developmental control of seed filling. Arabidopsis Book. https://doi.org/10.1199/tab.0113
Beeckman T, Przemeck G, Stamatiou G et al (2002) Genetic complexity of cellulose synthase A gene function in Arabidopsis embryogenesis. Plant Physiol 130:1883–1893. https://doi.org/10.1104/pp.102.010603
Bhuiyan NH, Friso G, Rowland E et al (2016) The plastoglobule-localized metallopeptidase PGM48 is a positive regulator of senescence in arabidopsis thaliana. Plant Cell 28:3020–3037. https://doi.org/10.1105/tpc.16.00745
Burn JE, Hocart CH, Birch RJ et al (2002) Functional analysis of the cellulose synthase genes. Plant Physiol 129:797–807. https://doi.org/10.1104/pp.010931.1
Burton RA, Gibeaut DM, Bacic A et al (2000) Virus-induced silencing of a plant cellulose synthase gene. Plant Cell 12:691–706. https://doi.org/10.1105/tpc.12.5.691
Campbell L, Rempel C, Wanasundara J (2016) Canola/rapeseed protein: future opportunities and directions—workshop proceedings of IRC 2015. Plants 5:17. https://doi.org/10.3390/plants5020017
Caño-Delgado A, Penfield S, Smith C et al (2003) Reduced cellulose synthesis invokes lignification and defense responses in Arabidopsis thaliana. Plant J 34:351–362. https://doi.org/10.1046/j.1365-313X.2003.01729.x
Chandrasekharan MB, Bishop KJ, Hall TC (2003) Module-specific regulation of the β-phaseolin promoter during embryogenesis. Plant J 33:853–866. https://doi.org/10.1046/j.1365-313X.2003.01678.x
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743. https://doi.org/10.1046/j.1365-313X.1998.00343.x
Corrales AR, Carrillo L, Nebauer SG et al (2014) Salinity assay in Arabidopsis. Bio-protocol. https://doi.org/10.21769/BioProtoc.1216
DeBolt S, Scheible W-R, Schrick K et al (2009) Mutations in UDP-Glucose:sterol glucosyltransferase in Arabidopsis cause transparent testa phenotype and suberization defect in seeds. Plant Physiol 151:78–87. https://doi.org/10.1104/pp.109.140582
Desprez T, Juraniec M, Crowell EF et al (2007) Organization of cellulose synthase complexes involved in primary cell wall synthesis in Arabidopsis thaliana. Proc Natl Acad Sci USA 104:15572–15577. https://doi.org/10.1073/pnas.0706569104
Ekman Å, Hayden DM, Dehesh K et al (2008) Carbon partitioning between oil and carbohydrates in developing oat (Avena sativa L.) seeds. J Exp Bot 59:4247–4257. https://doi.org/10.1093/jxb/ern266
Foster CE, Martin TM, Pauly M (2010) Comprehensive compositional analysis of plant cell walls (lignocellulosic biomass) part I: lignin. J Vis Exp. https://doi.org/10.3791/1837
Frisch DA, Van Der GAHM, Dias K, Hall TC (1995) Chromosomal integration is required for spatial regulation of expression from the phaseolin promoter. Plant J 7:503–512
Fujii S, Hayashi T, Mizuno K (2010) Sucrose synthase is an integral component of the cellulose synthesis machinery. Plant Cell Physiol 51:294–301. https://doi.org/10.1093/pcp/pcp190
Gacek K, Bartkowiak-Broda I, Batley J (2018) Genetic and molecular regulation of seed storage proteins (SSPs) to improve protein nutritional value of oilseed rape (Brassica napus L.) seeds. Front Plant Sci 9:1–9. https://doi.org/10.3389/fpls.2018.00890
Gillmor C, Poindexter P, Lorieau J et al (2002) α-glucosidase I is required for cellulose biosynthesis and morphogenesis in Arabidopsis. J Cell Biol 156:1003–1013. https://doi.org/10.1083/jcb.200111093
Goderis IJWM, De Bolle MFC, François IEJA et al (2002) A set of modular plant transformation vectors allowing flexible insertion of up to six expression units. Plant Mol Biol 50:17–27. https://doi.org/10.1023/A:1016052416053
Griffiths JS, Tsai AY-L, Xue H et al (2014) SALT-OVERLY SENSITIVE5 mediates Arabidopsis seed coat mucilage adherence and organization through pectins. Plant Physiol 165:991–1004. https://doi.org/10.1104/pp.114.239400
Haigler CH, Ivanova-Datcheva M, Hogan PS et al (2001) Carbon partitioning to cellulose synthesis. Plant Mol Biol 47:29–51. https://doi.org/10.1023/A:1010615027986
Hernández-Blanco C, Feng DX, Hu J et al (2007) Impairment of cellulose synthases required for Arabidopsis secondary cell wall formation enhances disease resistance. Plant Cell 19:890–903. https://doi.org/10.1105/tpc.106.048058
Iyer VV, Sriram G, Fulton DB et al (2008) Metabolic flux maps comparing the effect of temperature on protein and oil biosynthesis in developing soybean cotyledons. Plant Cell Environ 31:506–517. https://doi.org/10.1111/j.1365-3040.2008.01781.x
Jia W, Mikulski D, Rogiewicz A et al (2012) Low-fiber Canola. Part II: nutritive value of the meal. J Agric Food Chem 60:12231–12237
Jiang Y, Deyholos MK (2010) Transcriptome analysis of secondary-wall-enriched seed coat tissues of canola (Brassica napus L.). Plant Cell Rep 29:327–342. https://doi.org/10.1007/s00299-010-0824-x
Knowles PF (1983) Genetics and breeding of sunflower. J Am Oil Chem Soc 60:1252–1253. https://doi.org/10.1007/BF02702092
Lam HM, Wong P, Chan HK et al (2003) Overexpression of the ASN1 gene enhances nitrogen status in seeds of Arabidopsis. Plant Physiol 132:926–935. https://doi.org/10.1104/pp.103.020123
Le MHA, Buchet ADG, Beltranena E et al (2017) Digestibility and intestinal fermentability of canola meal from Brassica juncea and Brassica napus fed to ileal-cannulated grower pigs. Anim Feed Sci Technol 234:43–53. https://doi.org/10.1016/j.anifeedsci.2017.09.005
Li N, Gugel IL, Giavalisco P et al (2015) FAX1, a novel membrane protein mediating plastid fatty acid export. PLoS Biol 13:1–37. https://doi.org/10.1371/journal.pbio.1002053
Li F, Xie G, Huang J et al (2017) OsCESA9 conserved-site mutation leads to largely enhanced plant lodging resistance and biomass enzymatic saccharification by reducing cellulose DP and crystallinity in rice. Plant Biotechnol J 15:1093–1104. https://doi.org/10.1111/pbi.12700
Liu H, Zhai Z, Kuczynski K et al (2019) BIOTIN ATTACHMENT DOMAIN-CONTAINING proteins, inhibitors of ACCase, are regulated by WRINKLED1. bioRxiv. https://doi.org/10.1101/634550
Lock Y-Y, Snyder CL, Zhu W et al (2009) Antisense suppression of type 1 diacylglycerol acyltransferase adversely affects plant development in Brassica napus. Physiol Plant 137:61–71. https://doi.org/10.1111/j.1399-3054.2009.01258.x
Matthäus B (1998) Effect of dehulling on the composition of antinutritive compounds in various cultivars of rapeseed. Lipid 92:139–144
Mcallister CH, Beatty PH, Good AG (2012) Engineering nitrogen use efficient crop plants: the current status. Plant Biotechnol J 10:1011–1025. https://doi.org/10.1111/j.1467-7652.2012.00700.x
Mendu V, Griffiths JS, Persson S et al (2011) Subfunctionalization of cellulose synthases in seed coat epidermal cells mediates secondary radial wall synthesis and mucilage attachment. Plant Physiol 157:441–453. https://doi.org/10.1104/pp.111.179069
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Pan X, Siloto RMP, Wickramarathna AD et al (2013) Identification of a pair of phospholipid: diacylglycerol acyltransferases from developing flax (Linum usitatissimum L.) seed catalyzing the selective production of trilinolenin. J Biol Chem 288:24173–24188. https://doi.org/10.1074/jbc.M113.475699
Persson S, Paredez A, Carroll A et al (2007) Genetic evidence for three unique components in primary cell-wall cellulose synthase complexes in Arabidopsis. Proc Natl Acad Sci USA 104:15566–15571. https://doi.org/10.1073/pnas.0706592104
Sanjaya DTP, Weise SE, Benning C (2011) Increasing the energy density of vegetative tissues by diverting carbon from starch to oil biosynthesis in transgenic Arabidopsis. Plant Biotechnol J 9:874–883. https://doi.org/10.1111/j.1467-7652.2011.00599.x
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108. https://doi.org/10.1038/nprot.2008.73
Schrick K, Fujioka S, Takatsuto S et al (2004) A link between sterol biosynthesis, the cell wall, and cellulose in Arabidopsis. Plant J 38:227–243. https://doi.org/10.1111/j.1365-313X.2004.02039.x
Schrick K, Debolt S, Bulone V (2012) Deciphering the molecular functions of sterols in cellulose biosynthesis. Front Plant Sci 3:84. https://doi.org/10.3389/fpls.2012.00084
Shewry PR (2007) Improving the protein content and composition of cereal grain. J Cereal Sci 46:239–250. https://doi.org/10.1016/j.jcs.2007.06.006
Shewry P, Casey R (1999) Introduction: seed proteins. Springer Science, New York, pp 1–10
Shi L, Katavic V, Yu Y et al (2012) Arabidopsis glabra2 mutant seeds deficient in mucilage biosynthesis produce more oil. Plant J 69:37–46. https://doi.org/10.1111/j.1365-313X.2011.04768.x
Simbaya J, Slominski BA, Rakow G et al (1995) Quality characteristics of yellow-seeded Brassica seed meals: protein, carbohydrates, and dietary fiber components. J Agric Food Chem 43:2062–2066. https://doi.org/10.1021/jf00056a020
Singer SD, Chen G, Mietkiewska E et al (2016) Arabidopsis GPAT9 contributes to synthesis of intracellular glycerolipids but not surface lipids. J Exp Bot 67:4627–4638. https://doi.org/10.1093/jxb/erw242
Slominski BA, Meng X, Jia W, et al (2004) Chemical composition and nutritive value of yellow-seeded Brassica napus canola. Feed Ind Raw Mater Feed, pp 253–255
Somerville C (2006) Cellulose synthesis in higher plants. Annu Rev Cell Dev Biol 22:53–78. https://doi.org/10.1146/annurev.cellbio.22.022206.160206
Stork J, Harris D, Griffiths J et al (2010) CELLULOSE SYNTHASE9 serves a nonredundant role in secondary cell wall synthesis in Arabidopsis epidermal testa cells. Plant Physiol 153:580–589. https://doi.org/10.1104/pp.110.154062
Tan SH, Mailer RJ, Blanchard CL, Agboola SO (2011) Extraction and characterization of protein fractions from Australian canola meals. Food Res Int 44:1075–1082. https://doi.org/10.1016/j.foodres.2011.03.023
Tomlinson KL, McHugh S, Labbe H et al (2004) Evidence that the hexose-to-sucrose ratio does not control the switch to storage product accumulation in oilseeds: Analysis of tobacco seed development and effects of overexpressing apoplastic invertase. J Exp Bot 55:2291–2303. https://doi.org/10.1093/jxb/erh251
Ufaz S, Galili G (2008) Improving the content of essential amino acids in crop plants: goals and opportunities. Plant Physiol 147:954–961. https://doi.org/10.1104/pp.108.118091
Updegraff DM (1969) Semimicro determination of cellulose in biological materials. Anal Biochem 32:420–424. https://doi.org/10.1016/S0003-2697(69)80009-6
van Erp H, Kelly AA, Menard G, Eastmond PJ (2014) Multigene engineering of triacylglycerol metabolism boosts seed oil content in Arabidopsis. Plant Physiol 165:30–36. https://doi.org/10.1104/pp.114.236430
Vanhercke T, El Tahchy A, Liu Q et al (2014) Metabolic engineering of biomass for high energy density: oilseed-like triacylglycerol yields from plant leaves. Plant Biotechnol J 12:231–239. https://doi.org/10.1111/pbi.12131
Vishwanath SJ, Kosma DK, Pulsifer IP et al (2013) Suberin-associated fatty alcohols in Arabidopsis: distributions in roots and contributions to seed coat barrier properties. Plant Physiol 163:1118–1132. https://doi.org/10.1104/pp.113.224410
Weber H, Borisjuk L, Wobus U (1997) Sugar import and metabolism during seed development. Trends Plant Sci 2:169–174. https://doi.org/10.1016/S1360-1385(97)01030-3
Weichert N, Saalbach I, Weichert H et al (2010) Increasing sucrose uptake capacity of wheat grains stimulates storage protein synthesis. Plant Physiol 152:698–710. https://doi.org/10.1104/pp.109.150854
Weselake RJ, Taylor DC, Rahman MH et al (2009) Increasing the flow of carbon into seed oil. Biotechnol Adv 27:866–878. https://doi.org/10.1016/j.biotechadv.2009.07.001
Wesley SV, Helliwell CA, Smith NA et al (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27:581–590. https://doi.org/10.1046/j.1365-313X.2001.01105.x
Xu Y, Caldo KMP, Pal-Nath D et al (2018) Properties and biotechnological applications of Acyl-CoA:diacylglycerol acyltransferase and phospholipid:diacylglycerol acyltransferase from terrestrial plants and microalgae. Lipids 53:663–688. https://doi.org/10.1002/lipd.12081
Yoshie-Stark Y, Wada Y, Wasche A (2008) Chemical composition, functional properties, and bioactivities of rapeseed protein isolates. Food Chem 107:32–39. https://doi.org/10.1016/j.foodchem.2007.07.061
Zakharov A, Giersberg M, Hosein F et al (2004) Seed-specific promoters direct gene expression in non-seed tissue. J Exp Bot 55:1463–1471. https://doi.org/10.1093/jxb/erh158
Acknowledgements
We thank Dr. Kelvin Lien for his technical support in protein analysis carried out in this study as well Dr. Elzbieta Mietkiewska for her valuable discussion throughout. The research was supported by the University of Alberta Start-up Research Grant (G.C.), Genome Canada, Genome Prairie, Dow AgroScience (Corteva Agriscience, Agriculture division of DowDupont), Alberta Innovates (R.J.W. and G.C.), Alberta Agriculture and Forestry (G.C.), the Natural Science and Engineering Research Council of Canada (NSERC) Discovery Grants (Discovery grant number RGPIN-2018-05850 to J.O.; RGPIN-2016-05926 to G.C.; and RGPIN-2014-04585 to R.J.W.) and the Canada Research Chairs Program (R.J.W. and G.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Neal Stewart.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jayawardhane, K.N., Singer, S.D., Ozga, J.A. et al. Seed-specific down-regulation of Arabidopsis CELLULOSE SYNTHASE 1 or 9 reduces seed cellulose content and differentially affects carbon partitioning. Plant Cell Rep 39, 953–969 (2020). https://doi.org/10.1007/s00299-020-02541-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-020-02541-z