Abstract
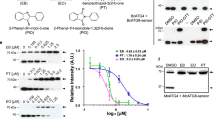
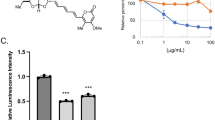
Serine is an essential component in organisms as a building block of biomolecules, a precursor of metabolites, an allosteric regulator of an enzyme, etc. This amino acid is thought to be a key metabolite in human diseases including cancers and infectious diseases. To understand the consequence of serine catabolism, we screened natural products to identify a fungal metabolite chaetoglobosin D (ChD) as a specific inhibitor of fission yeast cell growth when cultivated with serine as a sole nitrogen source. ChD targets actin, and actin mutant cells showed severe growth defect on serine medium. ROS accumulated in cells when cultivated in serine medium, while actin mutant cells showed increased sensitivity to oxidative stress. ROS production is a new aspect of serine metabolism, which might be involved in disease progression, and actin could be the drug target for curing serine-dependent symptoms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yeung KS, Paterson I. Actin-binding marine macrolides: total synthesis and biological importance. Angew Chem Int Ed. 2002;41:4632–53.
Kita M, Kigoshi H. Marine natural products that interfere with multiple cytoskeletal protein interactions. Nat Prod Rep. 2015;32:534–42.
Allingham JS, Klenchin VA, Rayment I. Actin-targeting natural products: structures, properties and mechanisms of action. Cell Mol Life Sci. 2006;63:2119–34.
Scherlach K, Boettger D, Remme N, Hertweck C. The chemistry and biology of cytochalasans. Nat Prod Rep. 2010;27:869–86.
de Koning TJ, et al. L-serine in disease and development. Biochem J. 2003;371:653–61.
Maddocks OD, et al. Serine starvation induces stress and p53-dependent metabolic remodelling in cancer cells. Nature. 2013;493:542–6.
Locasale JW. Serine, glycine and one-carbon units: cancer metabolism in full circle. Nat Rev Cancer. 2013;13:572–83.
Locasale JW, et al. Phosphoglycerate dehydrogenase diverts glycolytic flux and contributes to oncogenesis. Nat Genet. 2011;43:869–74.
Possemato R, et al. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature. 2011;476:346–50.
Pollari S, et al. Enhanced serine production by bone metastatic breast cancer cells stimulates osteoclastogenesis. Breast Cancer Res Treat. 2011;125:421–30.
Chaneton B, et al. Serine is a natural ligand and allosteric activator of pyruvate kinase M2. Nature 2012;491:458–62.
Kitamoto S, et al. Dietary L-serine confers a competitive fitness advantage to Enterobacteriaceae in the inflamed gut. Nat Microbiol. 2019:116–25.
Hayles J, Nurse P. A journey into space. Nat Rev Mol Cell Biol. 2001;2:647–56.
Hercyk BS, Onwubiko UN, Das ME. Coordinating septum formation and the actomyosin ring during cytokinesis in Schizosaccharomyces pombe. Mol Microbiol. 2019;112:1645–57.
Takahashi H, Sun X, Hamamoto M, Yashiroda Y, Yoshida M. The SAGA histone acetyltransferase complex regulates leucine uptake through the Agp3 permease in fission yeast. J Biol Chem. 2012;287:38158–67.
Sun X, et al. Identification of novel secreted fatty acids that regulate nitrogen catabolite repression in fission yeast. Sci Rep. 2016;6:20856.
Reidman S, Cohen A, Kupiec M, Weisman R. The cytosolic form of aspartate aminotransferase is required for full activation of TOR complex 1 in fission yeast. J Biol Chem. 2019;294:18244–55.
Fantes P, Nurse P. Control of cell size at division in fission yeast by a growth-modulated size control over nuclear division. Exp Cell Res. 1977;107:377–86.
Petersen J, Nurse P. TOR signalling regulates mitotic commitment through the stress MAP kinase pathway and the Polo and Cdc2 kinases. Nat Cell Biol. 2007;9:1263–72.
Ishiguro J, Kobayashi W. An actin point-mutation neighboring the ‘hydrophobic plug’ causes defects in the maintenance of cell polarity and septum organization in the fission yeast Schizosaccharomyces pombe. FEBS Lett. 1996;392:237–41.
Huang J, et al. Nonmedially assembled F-actin cables incorporate into the actomyosin ring in fission yeast. J Cell Biol. 2012;199:831–47.
Moreno S, Klar A, Nurse P. Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 1991;194:795–823.
Sekita S, Yoshihira K, Natori S. Chetoglobosins, cyto-toxic 10-(indol-3-yl)-[13]cytochalasans from Chaetomium Spp. 4. C-13-nuclear magnetic-resonance spectra and their application to a biosynthetic study. Chem Pharm Bull. 1983;31:490–8.
Marchetti MA, Weinberger M, Murakami Y, Burhans WC, Huberman JA. Production of reactive oxygen species in response to replication stress and inappropriate mitosis in fission yeast. J Cell Sci. 2006;119:124–31.
Sekita S, et al. Chaetoglobosins, cytotoxic 10-(indol-3-yl)-[13]cytochalasans from Chaetomium spp. I. Production, isolation and some cytological effects of chaetoglobosins A-J. Chem Pharm Bull. 1982;30:1609–17.
Yahara I, Harada F, Sekita S, Yoshihira K, Natori S. Correlation between effects of 24 different cytochalasins on cellular structures and cellular events and those on actin in vitro. J Cell Biol. 1982;92:69–78.
Maruyama K, et al. Effects of chaetoglobosin J on the G-F transformation of actin. Biochim Biophys Acta. 1986;874:137–43.
Kashman Y, Groweiss A, Shmueli J. Latrunculin, a New 2-thiazolidinone macrolide from the marine sponge Latrunculia-magnifica. Tetrahedron Lett. 1980;21:3629–32.
Gourlay CW, Carpp LN, Timpson P, Winder SJ, Ayscough KR. A role for the actin cytoskeleton in cell death and aging in yeast. J Cell Biol. 2004;164:803–9.
Farah ME, Amberg DC. Conserved actin cysteine residues are oxidative stress sensors that can regulate cell death in yeast. Mol Biol Cell. 2007;18:1359–65.
Leadsham JE, Kotiadis VN, Tarrant DJ, Gourlay CW. Apoptosis and the yeast actin cytoskeleton. Cell Death Differ. 2010;17:754–62.
Kobayashi D, et al. Endogenous reactive oxygen species is an important mediator of miconazole antifungal effect. Antimicrob Agents Chemother. 2002;46:3113–7.
Avery SV. Molecular targets of oxidative stress. Biochem J. 2011;434:201–10.
Fujiwara I, Zweifel ME, Courtemanche N, Pollard TD. Latrunculin A accelerates actin filament depolymerization in addition to sequestering actin monomers. Curr Biol 2018;28:3183–92. e2.
Lassing I, et al. Molecular and structural basis for redox regulation of beta-actin. J Mol Biol. 2007;370:331–48.
Lucas S, Chen G, Aras S, Wang J. Serine catabolism is essential to maintain mitochondrial respiration in mammalian cells. Life Sci Alliance. 2018;1:e201800036.
Acknowledgements
We thank J. Ishiguro (Konan University), M. Balasubramanian (University of Warwick) and National Bio-Resource Project of the MEXT, Japan for yeast strains, and M. Sasaki (The University of Tokyo) for reading the paper. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology, Japan (26102726 (SN), 17H06401 (HK and SN), 19H02840 (HK)) and Japan Society for the Promotion of Science (19H05640 (SN and MY)).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
This work is dedicated to Professor William Fenical for his great contribution to marine natural products chemistry.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Kanou, A., Nishimura, S., Tabuchi, T. et al. Serine catabolism produces ROS, sensitizes cells to actin dysfunction, and suppresses cell growth in fission yeast. J Antibiot 73, 574–580 (2020). https://doi.org/10.1038/s41429-020-0305-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41429-020-0305-6