Abstract
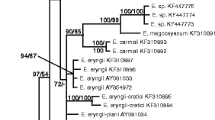
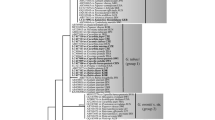
The phylogenetic revision of the genera Petrakia and Seifertia using LSU, ITS, RPB2 and TEF1 sequences and the re-evaluation of their morphological characteristics lead to several reclassifications: The genus Pseudodidymella as well as the genera Mycodidymella and Xenostigmina are synonymized with the genus Petrakia. Based on ITS sequence comparisons, it was previously suspected that the leaf spot pathogen Pseudodidymella fagi, which occurs on the Japanese beech Fagus crenata in Japan, is conspecific to the pathogen attacking the European beech Fagus sylvatica in Switzerland and Germany since 2008. Herein, we show that Japanese and European collections represent separate species and describe the European one as Petrakia liobae new to science. Apart from that, we make the new combinations Petrakia fagi and Petrakia minima. The names Petrakia aesculi and Petrakia aceris are validated. A 60-year-old collection from Wisconsin USA, designated as Petrakia echinata on leaves of silver maple (Acer saccharinum), proved to be another species new to science and is described here as Petrakia greenei. Consequently, there is currently no evidence of the European P. echinata to occur in North America. In contrast, P. echinata was found to infect the North American big leaf maple (Acer macrophyllum) in Europe. Antromycopsis alpina, described in 1914, was rediscovered in the Swiss Alps from dry fruits of Rhododendron ferrugineum. It is combined in Seifertia as S. alpina, based on molecular phylogenetic and morphological analyses. This anamorphic fungus appears to be native to Europe and does not cause a bud disease on Rhododendron in contrast to the closely related S. azaleae. Seifertia shangrilaensis is the third species of this genus that is closely related to Petrakia. Both genera belong to the family Melanommataceae.





Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Backus MP, Evans RI (1968) H. C. Greene (1904-1967). Mycologia 60(5):994–998
Baral H-O, Queloz V, Hosoya T (2014) Hymenoscyphus fraxineus, the correct scientific name for the fungus causing ash dieback in Europe. IMA Fungus 5(1):79–80. https://doi.org/10.5598/imafungus.2014.05.01.09
Bedlan G (2017) Petrakia juniperi sp. nov., a new fungus on Juniperus sp. J Kult pflanzen 69(5):173–174
Beenken L, Senn-Irlet B (2016) Neomyceten in der Schweiz, Stand des Wissens und Abschätzung des Schadpotentials der mit Pflanzen assoziierten gebietsfremden Pilze. WSL Berichte 50:1–93. https://www.dora.lib4ri.ch/wsl/islandora/object/wsl:9106
Beenken L, Zoller S, Berndt R (2012) Rust fungi on Annonaceae II: the genus Dasyspora Berk. & M.A. Curtis. Mycologia 104:659–681. https://doi.org/10.3852/11-068
Butin H (2011) Krankheiten der Wald- und Parkbäume, 4th edn. Ulmer, Stuttgart
Butin H, Holdenrieder O, Sieber TN (2013) The complete life cycle of Petrakia echinata. Mycol Prog 12:427–435
Castresana J (2000) Selection of conserved blocks from multiple align- ments for their use in phylogenetic analysis. Mol Biol Evol 17:540–552
Caudullo G, Welk E, San-Miguel-Ayanz J (2017) Chorological maps for the main European woody species. Data in brief 12:662–666. https://doi.org/10.1016/j.dib.2017.05.007
Caudullo G, Welk E, San-Miguel-Ayanz J (2018) Fagus sylvatica chorology. Figshare, Fileset. https://Figshare.com/articles/Fagus_sylvatica_chorology/5101144. Accessed 10 Sept 2019
Cech TL, Wiener L (2017) Pseudodidymella fagi, ein neuer Blattbräunepilz der Rotbuche in Österreich. Forstschutz Aktuell 62:22–25
Crous PW, Braun U, Wingfield MJ, Wood AR, Shin HD, Summerell BA, Alfenas AC, Cumagun CJ, Groenewald JZ (2009) Phylogeny and taxonomy of obscure genera of microfungi. Persoonia 22:139–161
Czachura P, Owczarek-Kościelniak M, Piątek M (2018) Pseudodidymella fagi in Slovakia: first detection, morphology and culture characteristics. Forest Pathol 2018:e12479. https://doi.org/10.1111/efp.12479
Davis WH (1939) A bud and twig blight of azaleas caused by Sporocybe azaleae. Phytopathology 29:517–529
Delgado G, Koukol O, Cáceres O, Piepenbring M (2017) The phylogenetic placement of Ernakulamia cochinensis within Pleosporales (Dothideomycetes, Ascomycota). Cryptogam Mycol 38(4):435–451
Den Bakker HC, Zuccarello GC, Kuyper TW, Noordeloos ME (2004) Evolution and host specificity in the ectomycorrhizal genus Leccinum. New Phytol 163:201–215
Denk T, Grimm GW (2009) The biogeographic history of beech trees. Rev Palaeobot Palynol 158:83–100
Desprez-Loustau ML (2009) Alien Fungi of Europe. In: Drake JA (ed) DAISIE, Handbook of Alien Species in Europe. Springer, pp 15–28
Endrestøl A (2017) Graphocephala fennahi Young, 1977 (Hemiptera, Cicadellidae) and Seifertia azaleae (Peck) Partr. & Morgan-Jones (Ascomycota, Dothideomycetes) in Norway. Norw J Entomol 64:112–129
Eschen R, O’Hanlon R, Santini A, Vannini A, Roques A, Kirichenko N, Kenis M (2019) Safeguarding global plant health: the rise of sentinels. J Pest Sci 92:29–36
Farr DF, Rossman AY (2019) Fungal databases, U.S. national fungus collections, ARS, USDA. https://nt.ars-grin.gov/fungaldatabases/. Accessed 10 Sept 2019
Fluri H, Senn-Irlet B, Graf U, Beenken L (2017) Geoglossum heuflerianum – Wiederentdeckung einer alpinen Art in der Schweiz. Osterr Z Pilzkd 26:87–97
Funk A (1986) Stigmina zilleri sp. nov., associated with brown leaf spot of broadleaf maple. Can J Bot 65(3):482–483
Greene HC (1960) Notes on Wisconsin parasitic fungi. XXVI. Trans Wis Acad Sci Arts Lett 49:85–111
Gross A, Hosoya T, Queloz V (2014) Population structure of the invasive forest pathogen Hymenoscyphus pseudoalbidus. Mol Ecol 23:2943–2960
Gross A, Beenken L, Dubach V, Queloz V, Tanaka K, Hashimoto A, Holdenrieder O (2017) Pseudodidymella fagi and Petrakia deviata: two closely related tree pathogens new to central. Eur For Pathol 00:e12351. https://doi.org/10.1111/efp.12351
Hashimoto A, Matsumura M, Hirayama K, Fujimoto R, Tanaka K (2017) Pseudodidymellaceae fam. nov.: phylogenetic affiliations of mycopappus-like genera in Dothideomycetes. Stud Mycol 87:187–206
Höhnel FV (1914) Fragmente zur Mykologie (XVI. Mitteilung, Nr. 813 bis 875). Sitz Ber Ak Wiss Wien M-N Klasse, Abt. 1 123:49–155
Howell PJ, Wood RKS (1962) Some factors affecting rhododendron bud blast and its control. Ann Appl Biol 50:723–733
Huelsenbeck JP, Ronquist F (2001) MrBayes: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755
Huson DH, Richter DC, Rausch C, Dezulian T, Franz M, Rupp R (2007) Dendroscope: an interactive viewer for large phylogenetic trees. BMC Bioinformatics 8:460
Index Herbariorum (2019) http://sweetgum.nybg.org/ih/. Accessed 10 Sept 2019
Jaklitsch WM, Voglmayr H (2017) Three former taxa of Cucurbitaria and considerations on Petrakia in the Melanommataceae. Sydowia 69:81–95
Kaneko S, Yokozawa Y, Kubono T (1988) Bud blight of Rhododendron trees caused by Pycnostysanus azaleae. Ann Phytopathol Soc Japan 54:323–326
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30(4):772–780
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier trans- form. Nucleic Acids Res 30:3059–3066
Kirisits T (2007) Die Petrakia-Blattbräune des Bergahorns. Forstschutz Aktuell 40:28–31
Li GJ, Hyde KD, Zhao RL et al (2016a) Fungal diversity notes 253–366: taxonomic and phylogenetic contributions to fungal taxa. Fungal Divers 78(1):1–237
Li J, Phookamsak R, Mapook A, Boonmee S, Bhat DJ, Hyde KD, Lumyong (2016b) Seifertia shangrilaensis sp. nov. (Melanommataceae), a new species from Southwest China. Phytotaxa 273(1):34–42. https://doi.org/10.11646/phytotaxa.273.1.3
Liu YJ, Whelen S, Hall BD (1999) Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit. Mol Biol Evol 16:1799–1808
Magri D, Vendramin GG, Comps B, Dupanloup I, Geburek T, Gömöry D, Latałowa M, Litt T, Paule L, Roure JM, Tantau I, van der Knaap WO, Petit RJ, de Beaulieu JL (2006) A new scenario for the Quaternary history of European beech populations: palaeobotanical evidence and genetic consequences. New Phytol 171:199–221. https://doi.org/10.1111/j.1469-8137.2006.01740.x
McMullan M, Rafiqi M, Kaithakottil G et al (2018) The ash dieback invasion of Europe was founded by two genetically divergent individuals. Nat Ecol Evol 2:1000–1008. https://doi.org/10.1038/s41559-018-0548-9
MyCoPortal (2019) http://mycoportal.org/portal/index.php. Accessed 10 Sept 2019
Nuytinck J, Verbeken A, Miller SL (2007) Worldwide Phylogeny of Lactarius Section Deliciosi Inferred from ITS and Glyceraldehyde-3-Phosphate Dehydrogenase Gene Sequences. Mycologia 99(6):820–832
Ogris N, Brglez A, Piškur B (2019) Pseudodidymella fagi in Slovenia: First Report and Expansion of Host Range. Forests 10(9):718. https://doi.org/10.3390/f10090718
Park J, Cho S, Lee SH, Shin HD (2013) First Report of Frosty Mildew on Salix koreensis Caused by Mycopappus alni in Korea. J Phytopathol 161:866–869. https://doi.org/10.1111/jph.12133
Partridge EC, Morgan-Jones G (2002) Notes on hyphomycetes LXXXVIII: New genera in which to classify Alysidium resinae and Pycnostysanus azaleae, with a consideration of Sorocybe. Mycotaxon 83:335–352
Peck CH (1873) [1874] Descriptions of new species of fungi. Bull Buffalo Soc Nat Sci 1:41–72
Peglion V (1895) Contributione alla conoscenza della Flora Micologica Avellinese. Malpighia 8:424–460
Petrak F (1966) [1968]. Über die Gattungen Petrakia Syd. und Echinosporium Woron. Sydowia 20:186–189
Queloz V, Grünig CR, Berndt R, Kowalski T, Sieber TN, Holdenrieder O (2011) Cryptic speciation in Hymenoscyphus albidus. Forest Pathol 41:133–142
Redhead SA, White GP (1985) Mycopappus, a new genus of leaf pathogens, and two parasitic Anguillospora species. Can J Bot 63(8):1429–1435
Rehner SA, Buckley E (2005) A Beauveria phylogeny inferred from nuclear ITS and EF1-a sequences: evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 97:84–98
Rehner SA, Samuels GJ (1994) Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol Res 98:625–634
Renner SS, Grimm GW, Kapli P, Denk T (2016) Species relationships and divergence times in beeches: new insights from the inclusion of 53 young and old fossils in a birth–death clock model. Philos T R Soc B 371. https://doi.org/10.1098/rstb.2015.0135
Ronquist F, Huelsenbeck JP (2005) MrBayes. Version 3.1.2. Bayesian analysis of phylogeny. GNU General Public License
Santini A, Ghelardini L, De Pace C et al (2013) Biogeographical patterns and determinants of invasion by forest pathogens in Europe. New Phytol 197:238–250
Santini A, Liebhold A, Migliorini D, Woodward S (2018) Tracing the role of human civilization in the globalization of plant pathogens. ISME J 12:647–652. https://doi.org/10.1038/s41396-017-0013-9
Schardl CL, Craven KD (2003) Interspecific hybridization in plant- associated fungi and oomycetes: A review. Mol Ecol 12:2861–2873
Schmitz H (1920) Observations on some common and important diseases of the Rhododendron. Phytopathology 10:273–278
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, Chen W, Fungal Barcoding Consortium (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for fungi. P Natl Acad Sci USA 109:6241–6246
Seifert KA, Hughes SJ, Boulay H, Louis-Seize G (2007) Taxonomy, nomenclature and phylogeny of three cladosporium-like hyphomycetes, Sorocybe resinae, Seifertia azaleae and the Hormoconis anamorph of Amorphotheca resinae. Stud Mycol 58:235–245
Sieber TM (2014) Neomyceten – eine anhaltende Bedrohung für den Schweizer Wald. Swiss For J 6:173–182
Stalpers JA, Seifertk A, Samsonr A (1991) A revision of the genera Antromycopsis, Sclerostilbum, and Tilachlidiopsis (Hyphomycetes). Can J of Bot 69:6–15
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313
Stukenbrock EH (2016) The role of hybridization in the evolution and emergence of new fungal plant pathogens. Phytopathology 106:104–112
Suto Y, Kawai M (2000) Mycopappus quercus sp. nov., causing frosty mildew in Quercus acutissima. Mycoscience 41(1):55–60
Suto Y, Suyama H (2005) Redheadia quercus gen. et sp. nov., the teleomorph of Mycopappus quercus, the frosty mildew fungus in Quercus acutissima. Mycoscience 46(4):227–234
Sydow H, Sydow P (1913) Novae fungorum species XI. Ann Mycol 11(5):402–408
Tian Q, Liu JK, Hyde KD et al (2015) Phylogenetic relationships and morphological reappraisal of Melanommataceae (Pleosporales). Fungal Divers 74:267–324
Turland NJ, Wiersema, JH, Barrie FR et al. (eds.) (2018). International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Vegetabile 159. Glashütten: Koeltz Botanical Books. https://doi.org/10.12705/Code.2018
Van der Aa HA (1968) Petrakia irregularis, a new fungus species. Acta Bot Neerl 17(3):221–225
Viennot-Bourgin G (1981) Observation simultanée en France du bud blast du Rhododendron et d’une cicadelle jouant le rôle de vecteur. Agronomie, EDP Sci 1(2):87–92
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
Watzl O (1937) Über die Bäume und Sträucher des von der Baramba entwässerten Gebietes der Chodschalgruppe und deren Blattkrankheiten, II Pilze an (lebenden) Blättern. Beih Bot Centralbl Abt B 57:431–439
Watzl O, Swoboda K, Singer R (1929) Bericht über eine Expedition in den Kaukasus. Anz Akad Wiss Wien 66(1):9–12
Wei CZ, Harada Y, Katumoto K (1997) Pseudodidymella fagi gen. et sp. nov. and its hyphomycete anamorph Pycnopleiospora fagi gen. et sp. nov. on Fagus crenata in Japan. Mycologia 89:494–502
Wei CZ, Harada Y, Katumoto K (1998) Mycodidymella aesculi gen. et sp. nov. and its synanamorphs Blastostroma aesculi gen. et sp. nov. and Mycopappus aesculi sp. nov. on Aesculus turbinata in Japan. Mycologia 90(2):334–345
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Wong MKM, Goh TK, McKenzie EHC, Hyde KD (2002) Fungi on grasses and sedges: Paratetraploa exappendiculata gen. et sp. nov., Petrakia paracochinensis sp. nov. and Spadicoides versiseptatis sp. nov. (dermatiaceous hyphomycetes). Cryptogamie Mycol 23(82):195–203
Acknowledgments
Ottmar Holdenrieder (Zurich) gave the impetus for this study. We are greatly obliged to him for important references in our study and manuscript. We thank Hermann Voglmayr (Vienna) for his Austrian collection of P. liobae, Thomas Cech (Vienna) and Marcin Piątek (Krakow) for data on their observations of P. liobae. Jörg Gilgen (Bern) and Beatrice Senn-Irlet (Bern) kindly provided their collections of S. alpina. We thank Anders Endrestøl (Oslo) for his decisive hint on the identity of S. alpina. Felix Neff kindly helped with photography using the Leica DVM6 Digital microscope at the WSL (Birmensdorf). We thank the curators of the herbaria in Harvard (FH), Vienna (W) and Wisconsin (WIS) for the possibility to study their collections. We are very grateful to the Harvard University Herbaria for the permission to publish the images of the type specimens of A. alpina kindly photographed by Genevieve E. Tocci. DNA extractions were performed at the WSL plant protection lab—many thanks for this to Quirin Kupper, Robin Winiger and Stephanie Pfister. Further molecular work and analyses were done at the GDC at ETH Zurich. The two newly described species were consciously named after their first finders in order to pay tribute to the important work of the field mycologists, without which many fungi would remain undiscovered. This study was financed by the WSL internal project “Pseudodydimella fagi, a new emerging disease of European beech.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Gerhard Rambold
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Taxonomic novelties: New species:Petrakia greenei Beenken, Andr. Gross & Queloz, Petrakia liobae Beenken, Andr. Gross & Queloz; New combinations:Petrakia fagi (C.Z. Wei, Y. Harada & Katum.) Beenken, Andr. Gross & Queloz, Petrakia minima (A. Hashim. & Kaz. Tanaka) Beenken, Andr. Gross & Queloz, Seifertia alpina (Höhn.) Beenken, Andr. Gross & Queloz.
Rights and permissions
About this article
Cite this article
Beenken, L., Gross, A. & Queloz, V. Phylogenetic revision of Petrakia and Seifertia (Melanommataceae, Pleosporales): new and rediscovered species from Europe and North America. Mycol Progress 19, 417–440 (2020). https://doi.org/10.1007/s11557-020-01567-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-020-01567-7