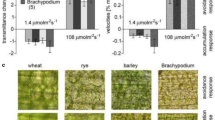
Abstract
Light-induced chloroplast movements control efficient light utilization in leaves, and thus, are essential for leaf photosynthesis and biomass production under fluctuating light conditions. Chloroplast movements have been intensively analyzed using wild-type and mutant plants of Arabidopsis thaliana. The molecular mechanism and the contribution to biomass production were elucidated. However, the knowledge of chloroplast movements is very scarce in other plant species, especially grass species including crop plants. Because chloroplast movements are efficient strategy to optimize light capture in leaves and thus promote leaf photosynthesis and biomass, analysis of chloroplast movements in crops is required for biomass production. Here, we analyzed chloroplast movements in a wide range of cultivated and wild species of genus Oryza. All examined Oryza species showed the blue-light-induced chloroplast movements. However, O. sativa and its ancestral species O. rufipogon, both of which are AA-genome species and usually grown in open condition where plants are exposed to full sunlight, showed the much weaker chloroplast movements than Oryza species that are usually grown under shade or semi-shade conditions, including O. officinalis, O. eichingeri, and O. granulata. Further detailed analyses of different O. officinalis accessions, including sun, semi-shade, and shade accessions, indicated that the difference in chloroplast movement strength between domesticated rice plants and wild species might result from the difference in habitat, and the shape of mesophyll chlorenchyma cells. The findings of this study provide useful information for optimizing Oryza growth conditions, and lay the groundwork for improving growth and yield in staple food crop Oryza sativa.




Similar content being viewed by others
References
Banaś AK, Aggarwal C, Labuz J, Sztatelman O, Gabryś H (2012) Blue light signaling in chloroplast movements. J Exp Bot 63:1559–1574. https://doi.org/10.1093/jxb/err429
Brugnoli E, Björkman O (1992) Chloroplast movements in leaves: influence on chlorophyll fluorescence and measurements of light-induced absorbance changes related to ΔpH and zeaxanthin formation. Photosynth Res 32:23–35. https://doi.org/10.1146/10.1007/BF00028795
Christie JM (2007) Phototropin blue-light receptors. Annu Rev Plant Biol 58:21–45. https://doi.org/10.1146/annurev.arplant.58.032806.103951
Davis PA, Caylor S, Whippo CW, Hangarter RP (2011) Changes in leaf optical properties associated with light-dependent chloroplast movements. Plant Cell Environ 34:2047–2059. https://doi.org/10.1111/j.1365-3040.2011.02402.x
Gotoh E, Suetsugu N, Higa T, Matsushita T, Tsukaya H, Wada M (2018a) Palisade cell shape affects the light-induced chloroplast movements and leaf photosynthesis. Sci Rep 8:1472. https://doi.org/10.1038/s41598-018-19896-9
Gotoh E, Suetsugu N, Yamori W, Ishishita K, Kiyabu R, Fukuda M, Higa T, Shirouchi B, Wada M (2018b) Chloroplast accumulation response enhances leaf photosynthesis and plant biomass production. Plant Physiol 178:1358–1369. https://doi.org/10.1104/pp.18.00484
Haga K, Takano M, Neumann R, Iino M (2005) The rice COLEOPTILE PHOTOTROPISM1 gene encoding an ortholog of Arabidopsis NPH3 is required for phototropism of coleoptiles and lateral tranlocation of auxin. Plant Cell 17:103–115. https://doi.org/10.1105/tpc.104.028357
Higa T, Wada M (2016) Chloroplast avoidance movement is not functional in plants grown under strong sunlight. Plant Cell Environ 39:871–882. https://doi.org/10.1111/pce.12681
Inoue Y, Shibata K (1973) Light-induced chloroplast rearrangements and their action spectra as measured by absorption spectrophotometry. Planta 114:341–358. https://doi.org/10.1007/BF00387947
Inoue Y, Shibata K (1974) Comparative examination of terrestrial plant leaves in terms of light-induced absorption changes due to chloroplast rearrangements. Plant Cell Physiol 15:717–721. https://doi.org/10.1093/oxfordjournals.pcp.a075058
Ishishita K, Suetsugu N, Hirose Y, Higa T, Doi M, Wada M, Matsushita T, Gotoh E (2016) Functional characterization of blue-light-induced responses and PHOTOTROPIN 1 gene in Welwitschia mirabilis. J Plant Res 129:175–187. https://doi.org/10.1007/s10265-016-0790-7
Jarillo JA, Gabrys H, Capel J, Alonso JM, Ecker JR, Cashmore AR (2001) Phototropin-related NPL1 controls chloroplast relocation induced by blue light. Nature 410:952–954. https://doi.org/10.1038/35073622
Kagawa T, Sakai T, Suetsugu N, Oikawa K, Ishiguro S, Kato T, Tabata S, Okada K, Wada M (2001) Arabidopsis NPL1: a phototropin homolog controlling the chloroplast high-light avoidance response. Science 291:2138–2141. https://doi.org/10.1126/science.291.5511.2138
Kanegae H, Tahir M, Savazzini F, Yamamoto K, Yano M, Sasaki T, Kanegae T, Wada M, Takano M (2000) Rice NPH1 homologues, OsNPH1a and OsNPH1b, are differently photoregulated. Plant Cell Physiol 41:415–423. https://doi.org/10.1093/pcp/41.4.415
Kasahara M, Kagawa T, Oikawa K, Suetsugu N, Miyao M, Wada M (2002a) Chloroplast avoidance movement reduces photodamage in plants. Nature 420:829–832. https://doi.org/10.1038/nature01213
Kasahara M, Swartz TE, Olney MA, Onodera A, Mochizuki N, Fukuzawa H, Asamizu E, Tabata S, Kanegae H, Takano M, Christie JM, Nagatani A, Briggs WR (2002b) Photochemical properties of the flavin mononucleotide-binding domains of the phototropins from Arabidopsis, rice, and Chlamydomonas reinhardtii. Plant Physiol 129:762–773. https://doi.org/10.1104/pp.002410
Kurata N, Yamazaki Y (2006) Oryzabase. An integrated biological and genome information database for rice. Plant Physiol 140:12–17. https://doi.org/10.1104/pp.105.063008
Li FW, Rothfels CJ, Melkonian M, Villarreal JC, Stevenson DW, Graham SW, Wong GK, Mathews S, Pryer KM (2015) The origin and evolution of phototropins. Front Plant Sci 6:637. https://doi.org/10.3389/fpls.2015.00637
Neumann R, Iino M (1997) Phototropism of rice (Oryza sativa L.) coleoptiles: fluence-response relationships, kinetics and photogravitropic equilibrium. Planta 201:288–292. https://doi.org/10.1007/s004250050068
Ohyanagi H, Ebata T, Huang X, Gong H, Fujita M, Mochizuki T, Toyoda A, Fujiyama A, Kaminuma E, Nakamura Y, Feng Q, Wang ZX, Han B, Kurata N (2016) OryzaGenome: genomic diversity database of wild Oryza species. Plant Cell Physiol 57:e1. https://doi.org/10.1093/pcp/pcv171
Oi T, Enomoto S, Nakao T, Arai S, Yamane K, Taniguchi M (2017) Three-dimensional intracellular structure of a whole rice mesophyll cell observed with FIB-SEM. Ann Bot 120:21–28. https://doi.org/10.1093/aob/mcx036
Park YI, Chow WS, Anderson JM (1996) Chloroplast movement in the shade plant Tradescantia albiflora helps protect photosystem II against light stress. Plant Physiol 111:867–875. https://doi.org/10.1104/pp.111.3.867
Sage TL, Sage RF (2009) The functional anatomy of rice leaves: implications for refixation of photorespiratory CO2 and efforts to engineer C4 photosynthesis into rice. Plant Cell Physiol 50:756–772. https://doi.org/10.1093/pcp/pcp033
Sakai T, Kagawa T, Kasahara M, Swartz TE, Christie JM, Briggs WR, Wada M, Okada K (2001) Arabidopsis nph1 and npl1: blue light receptors that mediate both phototropism and chloroplast relocation. Proc Natl Acad Sci USA 98:6969–6974. https://doi.org/10.1073/pnas.101137598
Sakamoto T, Morinaka Y, Ohnishi T, Sunohara H, Fujioka S, Ueguchi-Tanaka M, Mizutani M, Sakata K, Tak-atsuto S, Yoshida S, Tanaka H, Kitano H, Matsuoka M (2006) Erect leaves caused by brassinosteroid deficiency increase biomass production and grain yield in rice. Nat Biotechnol 24:105–109. https://doi.org/10.1038/nbt1173
Sanchez PL, Wing RA, Brar DS (2013) The wild relative of rice: genomes and genomics. In: Zhang Q, Wing RA (eds) Genetics and genomics of rice, plant genetics and genomics. Springer Science+Business Media, New York, pp 9–25
Shang B, Zang Y, Zhao X, Zhu J, Fan C, Guo X, Zhang X (2019) Functional characterization of GhPHOT2 in chloroplast avoidance of Gossypium hirsutum. Plant Physiol Biochem 135:51–60. https://doi.org/10.1016/j.plaphy.2018.11.027
Shimazaki K, Doi M, Assmann SM, Kinoshita T (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58:219–247. https://doi.org/10.1146/annurev.arplant.57.032905.105434
Sinclair TR, Sheehy JE (1999) Erect leaves and photosynthesis in rice. Science 283:1455. https://doi.org/10.1126/science.283.5407.1455c
Suetsugu N, Wada M (2007) Chloroplast photorelocation movement mediated by phototropin family proteins in green plants. Biol Chem 388:927–935. https://doi.org/10.1515/BC.2007.118
Suetsugu N, Wada M (2012) Chloroplast photorelocation movement: a sophisticated strategy for chloroplasts to perform efficient photosynthesis. In: Najafpour MM (ed) Advances in photosynthesis-fundamental aspects. InTech, Rijeka, pp 215–234
Suetsugu N, Wada M (2016) Evolution of the cp-actin-based motility system of chloroplasts in green plants. Front Plant Sci 7:561. https://doi.org/10.3389/fpls.2016.00561
Sztatelman O, Waloszek A, Banaś AK, Gabryś H (2010) Photoprotective function of chloroplast avoidance movement: in vivo chlorophyll fluorescence study. J Plant Physiol 167:709–716. https://doi.org/10.1016/j.jplph.2009.12.015
The 3,000 rice genomes project (2014) The 3,000 rice genomes project. GigaScience 3:7. https://doi.org/10.1186/2047-217X-3-7
Toda Y, Wang Y, Takahashi A, Kawai Y, Tada Y, Yamaji N, Feng Ma J, Ashikari M, Kinoshita T (2016) Oryza sativa H+-ATPase (OSA) is involved in the regulation of dumbbell-shaped guard cells of rice. Plant Cell Physiol 15:717–721. https://doi.org/10.1093/pcp/pcw070
Vaughan D (1994) The wild relatives of rice: a genetic resources handbook. The International Rice Research Institute, Manila
Wada M, Kong SG (2011) Analysis of chloroplast movement and relocation in Arabidopsis. Methods Mol Biol 774:87–102. https://doi.org/10.1007/978-1-61779-234-2_6
Walczak T, Gabryś H (1980) New type of photometer for measurements of transmission changes corresponding to chloroplast movement. Photosynthetica 14:65–72
Xiong D, Chen J, Yu T, Gao W, Ling X, Li Y, Peng S, Huang J (2015) SPAD-based leaf nitrogen estimation is impacted by environmental factors and crop leaf characteristics. Sci Rep 5:13389. https://doi.org/10.1038/srep13389
Yanai H (2011) Statcel 3—the useful add-in software forms on Excel, 3rd edn. OMS, Tokyo
Acknowledgements
We thank Tomonao Matsushita (Kyoto University, Japan) and Bungo Shirouchi (Kyushu University, Japan) for research supports, and Taiki Akasaka (Kyushu University, Japan) and Hiroyasu Furuumi (National Institute of Genetics, Japan) for research assistance. We thank the Center for Advanced Instrumental and Educational Support of the Faculty of Agriculture, Kyushu University, Japan for confocal microscopy. This work was supported in part by the Grant-in-Aid for Scientific Research to EG (no. JP15K18713, JP18K14491 and JP19H04729), NS (no. JP15KK0254 and JP19K06721), YY (no. JP18K05576), and MW (no. JP20227001, JP23120523, JP25120721, and JP25251033) from the Japan Society for the Promotion of Science, by a Grant-in-Aid for a JSPS Research Fellow to TH (no. JP17J06717), by the Research Grant for Young Investigators of Kyushu University to EG, by the Research Grant for Ichimura Fundation for New Technology to EG, and by the Research Grant for Ohsumi Frontier Science Foundation to MW. The wild rice accessions used in this study were distributed from the National Institute of Genetics supported by the National Bioresource Project (NBRP), AMED, Japan.
Author information
Authors and Affiliations
Contributions
EG conceived the study. EG and NS designed the study. MK, TU, YY, YT, TH, TA, TK, and EG performed the experiments. MK, TU, YY, TK, MW, NS, and EG analyzed the data. NS and EG wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 6010 kb)
Supplementary file3 (AVI 13864 kb)
Rights and permissions
About this article
Cite this article
Kihara, M., Ushijima, T., Yamagata, Y. et al. Light-induced chloroplast movements in Oryza species. J Plant Res 133, 525–535 (2020). https://doi.org/10.1007/s10265-020-01189-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-020-01189-w