Abstract
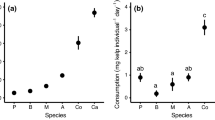
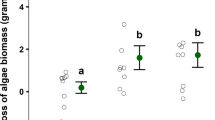
Ecologists have long wondered how plants and algae persist under constant herbivory, and studies have shown that factors like chemical defense and morphology can protect these species from consumption. However, grazers are also highly diverse and exert varying top-down control over primary producers depending on traits such as body size. Moreover, susceptibility of plants and algae to herbivory may vary across life stages and size classes, with juveniles potentially the most vulnerable. Here, we focus on diverse grazing communities within giant kelp forests and compared consumption on two size classes of juvenile giant kelp (Macrocystis pyrifera) across four herbivore species ranging in size. We also integrated field and literature densities to estimate impacts on populations of juvenile kelp. We found that purple sea urchins, a species known for exerting strong control over adult M. pyrifera, had weak per capita impact on microscopic kelp, on par with a much smaller crustacean species. While urchin consumption increased with macroscopic juvenile kelp, it never surpassed the smaller brown turban snail, suggesting that feeding morphology, in addition to herbivore body size, is a predictor of consumption at these small size classes. The smaller herbivores also occurred in high densities in the field, increasing their predicted population-level impacts on juvenile kelp compared to urchins and perhaps other larger, but less abundant, herbivores. This study highlights the variation in species’ roles within an herbivore guild and the importance of age-related changes in grazing vulnerability to better understand herbivore control on plant and algae population dynamics.



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Andersen RA (2005) Algal culturing techniques. Elsevier, Burlington
Andrews HL (1945) The kelp beds of the Monterey region. Ecology 26:24–37. https://doi.org/10.2307/1931912
Angelini C, Altieri AH, Silliman BR, Bertness MD (2011) Interactions among foundation species and their consequences for community organization, biodiversity, and conservation. Bioscience 61:782–789. https://doi.org/10.1525/bio.2011.61.10.8
Barker KM, Chapman ARO (1990) Feeding preferences of periwinkles among four species of Fucus. Mar Biol 106:113–118. https://doi.org/10.1007/bf02114681
Barton KE, Koricheva J (2010) The ontogeny of plant defense and herbivory: characterizing general patterns using meta-analysis. Am Nat 175:481–493. https://doi.org/10.1086/650722
Bernays EA (1998) Evolution of feeding behavior in insect herbivores. Bioscience 48:35–44. https://doi.org/10.2307/1313226
Bernstein BB, Jung N (1979) Selective pressures and coevolution in a kelp canopy community in southern California. Ecol Monogr 49:335–355. https://doi.org/10.2307/1942488
Boege K, Marquis RJ (2005) Facing herbivory as you grow up: the ontogeny of resistance in plants. Trends Ecol Evol 20:441–448. https://doi.org/10.1016/j.tree.2005.05.001
Booth JAT, McPhee-Shaw EE, Chua P, Kingsley E, Denny M, Phillips R, Bograd SJ, Zeidberg LD, Gilly WF (2012) Natural intrusions of hypoxic, low pH water into nearshore marine environments on the California coast. Cont Shelf Res 45:108–115. https://doi.org/10.1016/j.csr.2012.06.009
Bray RN, Ebeling AW (1975) Food, activity, and habitat of 3 “picker-type” microcarnivorous fishes in kelp forests off Santa Barbara, California. Fish Bull 73:815–829
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789. https://doi.org/10.1890/03-9000
Burkepile DE, Hay ME (2008) Herbivore species richness and feeding complementarity affect community structure and function on a coral reef. Proc Natl Acad Sci 105:16201–16206. https://doi.org/10.1073/pnas.0801946105
Cates RG (1980) Feeding patterns of monophagous, oligophagous, and polyphagous insect herbivores: the effect of resource abundance and plant chemistry. Oecologia 46:22–31. https://doi.org/10.1007/bf00346961
Davenport AC, Anderson TW (2007) Positive indirect effects of reef fishes on kelp performance: the importance of mesograzers. Ecology 88:1548–1561. https://doi.org/10.1890/06-0880
Dayton PK (1972) Toward an understanding of community resilience and the potential effects of enrichments to the benthos at McMurdo Sound, Antarctica Proceedings of the Colloquium on Conservation Problems in Antarctica. Allen Press Lawrence, Kansas, USA, pp 81–96
Estes JE, Smith NS, Palmisano JF (1978) Sea otter predation and community organization in the western Aleutian Islands, Alaska. Ecology 59:822–833. https://doi.org/10.2307/1938786
Field CB, Behrenfeld MJ, Randerson JT, Falkowski P (1998) Primary production of the biosphere: integrating terrestrial and oceanic components. Science 281:237–240. https://doi.org/10.1126/science.281.5374.237
Games PA, Howell JF (1976) Pairwise multiple comparison procedures with unequal n’s and/or variances: a Monte Carlo study. J Educ Stat 1:113–125. https://doi.org/10.2307/1164979
Gillooly JF, Brown JH, West GB, Savage VM, Charnov EL (2001) Effects of size and temperature on metabolic rate. Science 293:2248–2251. https://doi.org/10.1126/science.1061967
Graham MH (2002) Prolonged reproductive consequences of short-term biomass loss in seaweeds. Mar Biol 140:901–911. https://doi.org/10.1007/s00227-001-0761-x
Graham MH (2004) Effects of local deforestation on the diversity and structure of southern California giant kelp forest food webs. Ecosystems 7:341–357. https://doi.org/10.1007/s10021-003-0245-6
Graham MH, Vasquez JA, Buschmann AH (2007) Global ecology of the giant kelp Macrocystis: from ecotypes to ecosystems. Oceanogr Mar Biol Annu Rev 45:39
Gullström M, Bodin M, Nilsson PG, Öhman MC (2008) Seagrass structural complexity and landscape configuration as determinants of tropical fish assemblage composition. Mar Ecol Prog Ser 363:241–255. https://doi.org/10.3354/meps07427
Hairston NG, Smith FE, Slobodkin LB (1960) Community structure, population control, and competition. Am Nat 94:421–425. https://doi.org/10.1086/282146
Hammer RM, Zimmerman RC (1979) Species of demersal zooplankton inhabiting a kelp forest ecosystem off Santa Catalina Island, California. Bull South Calif Acad Sci 78:199–206
Hanley ME, Lamont BB, Fairbanks MM, Rafferty CM (2007) Plant structural traits and their role in anti-herbivore defence. Perspect Plant Ecol Evol Syst 8:157–178. https://doi.org/10.1016/j.ppees.2007.01.001
Hardy RA (1973) A survey of the marine environment near the city of Monterey ocean outfall. Marine Resources Administrative Report California Department of Fish and Game, Long Beach
Harrold C, Reed DC (1985) Food availability, sea urchin grazing, and kelp forest community structure. Ecology 66:1160–1169. https://doi.org/10.2307/1939168
Hay ME, Fenical W (1988) Marine plant-herbivore interactions: the ecology of chemical defense. Annu Rev Ecol Syst 19:111–145. https://doi.org/10.1146/annurev.ecolsys.19.1.111
Hillebrand H et al (2009) Herbivore metabolism and stoichiometry each constrain herbivory at different organizational scales across ecosystems. Ecol Lett 12:516–527. https://doi.org/10.1111/j.1461-0248.2009.01304.x
Hochuli DF (2001) Insect herbivory and ontogeny: how do growth and development influence feeding behaviour, morphology and host use? Austral Ecol 26:563–570. https://doi.org/10.1111/j.1442-9993.2001.tb00137.x
Hooper DU, Vitousek PM (1997) The effects of plant composition and diversity on ecosystem processes. Science 277:1302–1305. https://doi.org/10.1126/science.277.5330.1302
Huntly N (1991) Herbivores and the dynamics of communities and ecosystems. Annu Rev Ecol Syst 22:477–503. https://doi.org/10.1146/annurev.ecolsys.22.1.477
Kearsley MJ, Whitham TG (1989) Developmental changes in resistance to herbivory: implications for individuals and populations. Ecology 70:422–434. https://doi.org/10.2307/1937547
Leighton DL (1966) Studies of food preference in algivorous invertebrates of southern California kelp beds. Pac Sci 20:104–113
Light SF (2007) The Light and Smith manual: intertidal invertebrates from central California to Oregon. University of California Press, Berkeley
Ling SD et al (2015) Global regime shift dynamics of catastrophic sea urchin overgrazing. Philos Trans Royal Soc B 370:20130269. https://doi.org/10.1098/rstb.2013.0269
Lowry L, Pearse JS (1973) Abalones and sea urchins in an area inhabited by sea otters. Mar Biol 23:213–219. https://doi.org/10.1007/bf00389487
Lubchenco J (1983) Littorina and Fucus: effects of herbivores, substratum heterogeneity, and plant escapes during succession. Ecology 64:1116–1123. https://doi.org/10.2307/1937822
Ma Y, Cohen SR, Addadi L, Weiner S (2008) Sea urchin tooth design: an “All-Calcite” polycrystalline reinforced fiber composite for grinding rocks. Adv Mater 20:1555–1559. https://doi.org/10.1002/adma.200702842
MacArthur RH (1972) Strong, or weak, interactions. Trans Conn Acad Arts Sci 44:177–188
Mann KH (1977) Destruction of kelp-beds by sea-urchins: a cyclical phenomenon or irreversible degradation? Helgoländer Wissenschaftliche Meeresuntersuchungen 30:455. https://doi.org/10.1007/bf02207854
Milchunas DG, Noy-Meir I (2002) Grazing refuges, external avoidance of herbivory and plant diversity. Oikos 99:113–130. https://doi.org/10.1034/j.1600-0706.2002.990112.x
Ng CA, Micheli F (2020) Short-term effects of hypoxia are more important than effects of ocean acidification on grazing interactions with juvenile giant kelp (Macrocystis pyrifera). Sci Rep 10:5403. https://doi.org/10.1038/s41598-020-62294-3
Norton TA, Manley NL (1990) The characteristics of algae in relation to their vulnerability to grazing snails. Behavioural mechanisms of food selection. Springer, New York, pp 461–478
Novotny V et al (2010) Guild-specific patterns of species richness and host specialization in plant–herbivore food webs from a tropical forest. J Anim Ecol 79:1193–1203. https://doi.org/10.1111/j.1365-2656.2010.01728.x
Parker MA (1985) Size-dependent herbivore attack and the demography of an arid grassland shrub. Ecology 66:850–860. https://doi.org/10.2307/1940547
Parker GG et al (2004) Three-dimensional structure of an old-growth Pseudotsuga-Tsuga canopy and its implications for radiation balance, microclimate, and gas exchange. Ecosystems 7:440–453. https://doi.org/10.1007/s10021-004-0136-5
Pearse JS (2006) Ecological role of purple sea urchins. Science 314:940–941. https://doi.org/10.1126/science.1131888
Poore AGB et al (2012) Global patterns in the impact of marine herbivores on benthic primary producers. Ecol Lett 15:912–922. https://doi.org/10.1111/j.1461-0248.2012.01804.x
Sala E, Boudouresque C, Harmelin-Vivien M (1998) Fishing, trophic cascades, and the structure of algal assemblages: evaluation of an old but untested paradigm. Oikos. https://doi.org/10.2307/3546364
Sala E, Graham MH (2002) Community-wide distribution of predator–prey interaction strength in kelp forests. Proc Natl Acad Sci 99:3678–3683. https://doi.org/10.1073/pnas.052028499
Shulman MJ (1990) Aggression among sea urchins on Caribbean coral reefs. J Exp Mar Biol Ecol 140:197–207. https://doi.org/10.1016/0022-0981(90)90127-x
Silliman BR, McCoy MW, Angelini C, Holt RD, Griffin JN, van de Koppel J (2013) Consumer fronts, global change, and runaway collapse in ecosystems. Annu Rev Ecol Evol Syst 44:503–538. https://doi.org/10.1146/annurev-ecolsys-110512-135753
Silliman BR, Newell SY (2003) Fungal farming in a snail. Proc Natl Acad Sci 100:15643–15648. https://doi.org/10.1073/pnas.2535227100
Silliman BR, Zieman JC (2001) Top-down control of Spartina alterniflora production by periwinkle grazing in a Virginia salt marsh. Ecology 82:2830–2845. https://doi.org/10.2307/2679964
Steneck RS, Watling L (1982) Feeding capabilities and limitation of herbivorous molluscs: a functional group approach. Mar Biol 68:299–319. https://doi.org/10.1007/bf00409596
Taylor CR, Maloiy GMO, Weibel ER, Langman VA, Kamau JMZ, Seeherman HJ, Heglund NC (1981) Design of the mammalian respiratory system. III. Scaling maximum aerobic capacity to body mass: wild and domestic mammals. Respir Physiol 44:25–37. https://doi.org/10.1016/0034-5687(81)90075-x
Tegner MJ, Dayton PK, Edwards PB, Riser KL (1995) Sea urchin cavitation of giant kelp (Macrocystis pyrifera C. Agardh) holdfasts and its effects on kelp mortality across a large California forest. J Exp Mar Biol Ecol 191:83–99. https://doi.org/10.1016/0022-0981(95)00053-t
Tracey SR, Baulch T, Hartmann K, Ling SD, Lucieer V, Marzloff MP, Mundy C (2015) Systematic culling controls a climate driven, habitat modifying invader. Biol Invasions 17:1885–1896. https://doi.org/10.1007/s10530-015-0845-z
Valentine JF, Heck KL (1999) Seagrass herbivory: evidence for the continued grazing of marine grasses. Mar Ecol Prog Ser 176:291–302. https://doi.org/10.3354/meps176291
Van Alstyne KL, Whitman SL, Ehlig JM (2001) Differences in herbivore preferences, phlorotannin production, and nutritional quality between juvenile and adult tissues from marine brown algae. Mar Biol 139:201–210. https://doi.org/10.1007/s002270000507
Viejo RM, Åberg P, Cervin G, Lindegarth M (1999) The interactive effects of adult canopy, germling density and grazing on germling survival of the rockweed Ascophyllum nodosum. Mar Ecol Prog Ser 187:113–120. https://doi.org/10.3354/meps187113
Watanabe JM (1984) The influence of recruitment, competition, and benthic predation on spatial distributions of three species of kelp forest gastropods (Trochidae: Tegula). Ecology 65:920–936. https://doi.org/10.2307/1938065
Wilkinson DM, Sherratt TN (2016) Why is the world green? The interactions of top–down and bottom–up processes in terrestrial vegetation ecology. Plant Ecol Divers 9:127–140. https://doi.org/10.1080/17550874.2016.1178353
Wittstock U, Agerbirk N, Stauber EJ, Olsen CE, Hippler M, Mitchell-Olds T, Gershenzon J, Vogel H (2004) Successful herbivore attack due to metabolic diversion of a plant chemical defense. Proc Natl Acad Sci 101:4859–4864. https://doi.org/10.1073/pnas.0308007101
Wootton JT (1997) Estimates and tests of per capita interaction strength: diet, abundance, and impact of intertidally foraging birds. Ecol Monogr 67:45–64. https://doi.org/10.2307/2963504
Acknowledgements
We thank A. Muth and M. Graham for support in developing the kelp culturing methods; N. Low, P. Leary, and J. Lee for help with the aquarium system; K. Chang-Haines, A. Wachtell, M. Pobis, J. Salazar, T. Leggett, L. Anderson, A. Charlesworth, C. Aragon, and A. Meislin for help in the field and laboratory; J. Cohen for the illustrations; and J. Barry, L. Crowder, and R. Dirzo for advice on the experimental design and data analyses.
Funding
This work was supported by the National Science Foundation (DEB-1212124, DGE-114747, BioOce 1736830 and 1722513), The Women Divers Hall of Fame, and the Myers Oceanographic and Marine Biology Trust.
Author information
Authors and Affiliations
Contributions
CAN and FM conceived and designed the experiments. CAN performed the experiments, collected and analyzed the data, and wrote the first draft. Both authors contributed equally to subsequent revisions of the analyses and manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Communicated by Pablo Munguia.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ng, C.A., Micheli, F. Size-dependent vulnerability to herbivory in a coastal foundation species. Oecologia 193, 199–209 (2020). https://doi.org/10.1007/s00442-020-04655-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04655-3