Abstract
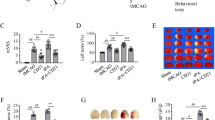
The macrophage scavenger receptor 1 (MSR1)-induced resolution of neuroinflammation may be a novel therapeutic strategy for ischemic stroke. Our previous study showed that the neuroprotective and anti-inflammatory effects of phthalide are associated with the inhibition of the post-ischemic damage-associated molecular pattern (DAMP)/Toll-like receptor 4 (TLR4) pathway. This study investigated the effects of the phthalide derivative CD21 on ischemic brain injury and the mechanism underlying MSR1-induced resolution of neuroinflammation. Using a rat model of 2 h transient middle cerebral artery occlusion (MCAO), MSR1-induced peroxiredoxin1 (PRX1) clearance in RAW264.7 macrophages were investigated. We show here that CD21 significantly ameliorated infarct volumes and neurological deficits in a dose-dependent manner with a ≥ 12 h therapeutic time window. Moreover, administration of 5 mg/kg/day CD21 over 24 h significantly reduced pathological damages, with associated inhibition of PRX1 expression, reduced TLR4/nuclear factor-κB activation and the suppression of the inflammatory response in MCAO rats. Furthermore, the expression of MAFB/MSR1 in the ischemic brain was elevated and the phagocytosis of PRX1 in CD68-positive macrophages isolated from the ischemic brain was enhanced. Further in vitro studies show that CD21 (20 μM) strongly enhanced the Msr1 mRNA and MSR1 protein levers in RAW264.7 cells and PRX1 internalization in cellular lysosomes, which were significantly reversed by N-acetylcysteine treatment. These results suggest that CD21 may exert neuroprotective and anti-inflammatory effects with a wide time window for the treatment of ischemic stroke. The anti-stroke effects of CD21 appear to be mediated partially via the induction of MSR1-promoted DAMP (PRX1) clearance, TLR4/nuclear factor-κB pathway inhibition, and the resolution of inflammation.

The neuroprotective action of CD21 was associated with the resolution of neuroinflammation through enhancement of the MAFB-MSR1 pathway that leads to DAMP (PRX1) phagocytosis and TLR4 pathway inhibition. Red solid arrows represent promotion, red dotted arrow represents the positive correlation, green arrows represent inhibition.







Similar content being viewed by others
References
Angel C, Dirnagl U, Urra X, Planas AM (2016) Neuroprotection in acute stroke: targeting Excitotoxicity, oxidative and Nitrosative stress, and inflammation. Lancet Neurol 15:869–881. https://doi.org/10.1016/S1474-4422(16)00114-9
Aziz A, Vanhille L, Mohideen P, Kelly LM, Otto C, Bakri Y, Mossadegh N, Sarrazin S, Sieweke MH (2006) Development of macrophages with altered actin organization in the absence of MAFB. Mol Cell Biol 26:6808–6818. https://doi.org/10.1128/MCB.00245-06
Billod JM, Lacetera A, Guzman CJ, Martin SS (2016) Computational approaches to toll-like receptor 4 modulation. Molecules 21:994–1018. https://doi.org/10.3390/molecules21080994
Brea D, Blanco M, Ramos-Cabrer P, Moldes O, Arias S, Pérez-Mato M, Leira R, Sobrino T, Castillo J (2011) Toll-like receptors 2 and 4 in ischemic stroke: outcome and therapeutic values. J Cereb Blood Flow Metab 31:1424–1431. https://doi.org/10.1038/jcbfm.2010.231
Buckley CD, Gilroy DW, Serhan CN, Stockinger B, Tak PP (2013) The resolution of inflammation. Nat Rev Immunol 13:59–66. https://doi.org/10.1038/nri3362
Chen C, Du JR, Shi MQ, Miao XN, San N, Wang PH (2017) The preparation methods and applications of 3-alkyl-5, 6-dioxide-substituted phthalein compounds. Patent, CN106432161 a
Dayon L, Turck N, Garcí-Berrocoso T, Walter N, Burkhard PR, Vilalta A, Sahuquillo J, Montaner J, Sanchez JC (2011) Brain extracellular fluid protein changes in acute stroke patients. J Proteome Res 10:1043–1051. https://doi.org/10.1021/pr101123t
Dirnagl U, Endres M (2014) Found in translation: preclinical stroke research predicts human pathophysiology, clinical phenotypes, and therapeutic outcomes. Stroke 45:1510–1518. https://doi.org/10.1161/STROKEAHA.113.004075
Enrique ZL, Philip RW, Sara S, Robert C (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91. https://doi.org/10.1161/01.str.20.1.84
Gan Y, Ji X, Hu X, Luo Y, Zhang L, Li P, Liu X, Yan F, Vosler P, Gao Y, Stetler RA, Chen J (2012) Transgenic overexpression of peroxiredoxin-2 attenuates ischemic neuronal injury via suppression of a redox-sensitive pro-death signaling pathway. Antioxid Redox Signal 17:719–732. https://doi.org/10.1089/ars.2011.4298
Goulopoulou S, McCarthy CG, Webb RC (2016) Toll-like receptors in the vascular system: sensing the dangers within. Pharmacol Rev 68:142–167. https://doi.org/10.1124/pr.114.010090
Iadecola C, Anrather J (2011a) The immunology of stroke: from mechanisms to translation. Nat Med 17:796–808. https://doi.org/10.1038/nm.2399
Iadecola C, Anrather J (2011b) Stroke research at a crossroad: asking the brain for directions. Nat Neurosci 14:1363–1368. https://doi.org/10.1038/nn.2953
Jin MH, Lee YH, Kim JM, Sun HN, Moon EY, Shong MH, Kim SU, Lee SH, Lee TH, Yu DY, Lee DS (2005) Characterization of neural cell types expressing peroxiredoxins in mouse brain. Neurosci Lett 381:252–257. https://doi.org/10.1016/j.neulet.2005.02.048
Joseph EK, Suzanne EH, Christian AT, Long C, Samuel CS, John DL (1996) Scavenger receptor-mediated adhesion of microglia to β-amyloid fibrils. Nature 382:716–719. https://doi.org/10.1038/382716a0
Kuang X, Yao Y, Du JR, Liu YX, Wang CY, Qian ZM (2006) Neuroprotective role of Z-ligustilide against forebrain ischemic injury in ICR mice. Brain Res 1102:145–153. https://doi.org/10.1016/j.brainres.2006.04.110
Kuang X, Du JR, Liu YX, Zhang GY, Peng HY (2008) Postischemic administration of Z-Ligustilide ameliorates cognitive dysfunction and brain damage induced by permanent forebrain ischemia in rats. Pharmacol Biochem Behav 88:213–221. https://doi.org/10.1016/j.pbb.2007.08.006
Kuang X, Chen YS, Wang LF, Li YJ, Liu K, Zhang MX, Li LJ, Chen C, He Q, Wang Y, du JR (2014a) Klotho upregulation contributes to the neuroprotection of ligustilide in an Alzheimer's disease mouse model. Neurobiol Aging 35:169–178. https://doi.org/10.1016/j.neurobiolaging.2013.07.019
Kuang X, Wang LF, Yu L, Li YJ, Wang YN, He Q, Chen C, du JR (2014b) Ligustilide ameliorates neuroinflammation and brain injury in focal cerebral ischemia/reperfusion rats: involvement of inhibition of TLR4/peroxiredoxin 6 signaling. Free Radic Biol Med 71:165–175. https://doi.org/10.1016/j.freeradbiomed.2014.03.028
Lifshitz V, Weiss R, Levy H, Frenkel D (2013) Scavenger receptor a deficiency accelerates cerebrovascular amyloidosis in an animal model. J Mol Neurosci 50:198–203. https://doi.org/10.1007/s12031-012-9909-z
Liu DL, Zhao LX, Zhang S, Du JR (2016) Peroxiredoxin 1-mediated activation of TLR4/NF-kappaB pathway contributes to neuroinflammatory injury in intracerebral hemorrhage. Int Immunopharmacol 41:82–89. https://doi.org/10.1016/j.intimp.2016.10.025
Macrez R, Ali C, Toutirais O, Le Mauff B, Defer G, Dirnagl U, Vivien D (2011) Stroke and the immune system: from pathophysiology to new therapeutic strategies. Lancet Neurol 10:471–480. https://doi.org/10.1016/s1474-4422(11)70066-7
Mao XN, Zhou HJ, Chen C, Liu DL, Yang XJ, Du JR (2017) Neuroprotective effect of a novel gastrodin derivative against ischemic brain injury involvement of peroxiredoxin and TLR4 signaling inhibition. Oncotarget 8:90979–90995. https://doi.org/10.18632/oncotarget.18773
Molteni M, Bosi A, Rossetti C (2018) Natural products with toll-like receptor 4 antagonist activity. Int J Inflamm 2018:1–9. https://doi.org/10.1155/2018/2859135
Monty K (1997) The other side of scavenger receptors: pattern recognition for host defense. Curr Opin Lipido 8:275–280. https://doi.org/10.1097/00041433-199710000-00006
Moskowitz MA, Lo EH, Iadecola C (2010) The science of stroke: mechanisms in search of treatments. Neuron 67:181–198. https://doi.org/10.1016/j.neuron.2010.07.002
Peng HY, Dun JR, Zhang GY, Kuang X, Liu YX, Qian ZM, Wang CY (2007) Neuroprotective effect of Z-ligustilide against permanent focal ischemic damage in rats. Biol Pharm Bull 30:309–312. https://doi.org/10.1248/bpb.30.309
Poornima V, Shen Y, Chopp M, Chen J (2018) Cell-based and pharmacological neurorestorative therapies for ischemic stroke. Neuropharmacology 15:310–322. https://doi.org/10.1016/j.neuropharm.2017.08.036
Riddell JR, Wang XY, Minderman H, Gollnick SO (2010) Peroxiredoxin 1 stimulates secretion of proinflammatory cytokines by binding to TLR4. J Immunol 184:1022–1030. https://doi.org/10.4049/jimmunol.0901945
Rock FL, Hardiman G, Timans JC, Kastelein RA, Bazan JF (1998) A family of human receptors structurally related to Drosophila toll. Proc Natl Acad Sci U S A 95:588–593. https://doi.org/10.1073/pnas.95.2.588
Sarrazin S, Mossadegh-Keller N, Fukao T, Aziz A, Mourcin F, Vanhille L, Kelly Modis L, Kastner P, Chan S, Duprez E, Otto C, Sieweke MH (2009) MAFB restricts M-CSF-dependent myeloid commitment divisions of hematopoietic stem cells. Cell 138:300–313. https://doi.org/10.1016/j.cell.2009.04.057
Shichita T, Hasegawa E, Kimura A, Morita R, Sakaguchi R, Takada I, Sekiya T, Ooboshi H, Kitazono T, Yanagawa T, Ishii T, Takahashi H, Mori S, Nishibori M, Kuroda K, Akira S, Miyake K, Yoshimura A (2012) Peroxiredoxin family proteins are key initiators of post-ischemic inflammation in the brain. Nat Med 18:911–917. https://doi.org/10.1038/nm.2749
Shichita T, Minako I, Yoshimura A (2014) Post-ischemic inflammation regulates neural damage and protection. Front Cell Neurosci 8:319–327. https://doi.org/10.3389/fncel.2014.00319
Shichita T, Ito M, Morita R, Komai K, Noguchi Y, Ooboshi H, Koshida R, Takahashi S, Kodama T, Yoshimura A (2017) MAFB prevents excess inflammation after ischemic stroke by accelerating clearance of damage signals through MSR1. Nat Med 23:723–732. https://doi.org/10.1038/nm.4312
Svensson L, Noren K, Wiklund O, Lindmark H, Ohlsson B, Hultén LM (2002) Inhibitory effects of N-acetylcysteine on scavenger receptor classs a expression in human macrophages. J Intern Med 251:437–446. https://doi.org/10.1046/j.1365-2796.2002.00983.x
Yang XJ (2018) Neuroprotection of a novel compound CD21 against ischemia stroke in rats and the involved mechanisms. Sichuan University, Dissertation
Zeng CY, Yang TT, Zhou HJ, Zhao Y, Kuang X, Duan W, Du JR (2019) Lentiviral vector-mediated overexpression of Klotho in the brain improves Alzheimer's disease-like pathology and cognitive deficits in mice. Neurobiol Aging 78:18–28. https://doi.org/10.1016/j.neurobiolaging.2019.02.003
Zhao LX, Du JR, Zhou HJ, Liu DL, Gu MX, Long FY (2016) Differences in Proinflammatory property of six subtypes of Peroxiredoxins and anti-inflammatory effect of Ligustilide in macrophages. PLoS One 11:e0164586. https://doi.org/10.1371/journal.pone.0164586
Zhou HJ et al (2017) Protective effect of Klotho against ischemic brain injury is associated with inhibition of RIG-I/NF-kappaB signaling. Front Pharmacol 8:950–962. https://doi.org/10.3389/fphar.2017.00950
Acknowledgements
This work was supported by National Natural Science Foundation of China (81072636 and 81473219), Sichuan Science and Technology Programs (2014JY0151 and 2018SZ0015), the Fundamental Research Funds for the Central Universities (YJ201561), Science Foundation for Excellent Youth Scholars in Sichuan Province (2017JQ0014), 111 Project of the National Ministry of Education (B18035).
Author information
Authors and Affiliations
Contributions
Jun-Rong Du conceptualized and designed this study. Xiao-Jia Yang, Xiao Zou, Yu-Miao Gan and Dong-Ling Liu performed this experiment.Chu Chen, Xiao-Jia Yang and Yu-Miao Gan contributed reagents and materials. Xiao Zou and Xiao-Jia Yang conducted the data analysis and prepared the draft. Jun-Rong Du rewrote the manuscript and Wei Duan assisted in the revision.
Corresponding author
Ethics declarations
Ethics Approval
All of the animal experiments were performed in accordance with China Animal Welfare Legislation and the Guidelines of Laboratory Animal Care and Use of Sichuan University.
Conflict of Interest
All authors have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zou, X., Yang, XJ., Gan, YM. et al. Neuroprotective Effect of Phthalide Derivative CD21 against Ischemic Brain Injury:Involvement of MSR1 Mediated DAMP peroxiredoxin1 Clearance and TLR4 Signaling Inhibition. J Neuroimmune Pharmacol 16, 306–317 (2021). https://doi.org/10.1007/s11481-020-09911-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-020-09911-0