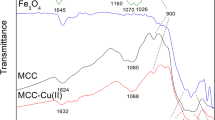
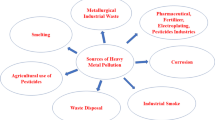
Abstract
Environmental pollution by toxic metals causes serious health complications, thus requiring advanced remediation methods for waters and effluents. In particular, chitosan-based magnetic materials have been recently developed to remove metals from aqueous solutions, industrial wastewater and water from lakes and rivers. Here, we review the adsorption of lead (Pb), cadmium (Cd), mercury (Hg) and arsenic (As) using magnetic chitosan. The manuscript presents recent experimental findings on the synthesis of magnetic adsorbents, focusing on magnetization methods, the main aspects of adsorption and adsorbent regeneration. The major findings are: (1) Kinetic patterns are mostly correlated by pseudo-second-order equations. (2) Langmuir isotherm model provides satisfactory estimations of monolayer capacity, the highest reported values being 341.7 mg/g for lead, 152 mg/g for mercury, 321.9 mg/g for cadmium and 65.5 mg/g for arsenic. (3) Most magnetic chitosan-based adsorbents keep their magnetic features and adsorption efficiency in consecutive adsorption–desorption runs. Overall, most chitosan-based magnetic adsorbents provide effective uptake of toxic metals ions from aqueous media and have a high degree of reusability.



Similar content being viewed by others
References
Abdollahi SB (2015) A new approach for analysis of adsorption from liquid phase: a critical review. J Pollut Eff Control. https://doi.org/10.4172/2375-4397.1000139
Ayawei N, Ebelegi AN, Wankasi D (2017) Modelling and interpretation of adsorption isotherms. J Chem. https://doi.org/10.1155/2017/3039817
Azari A, Gharibi H, Kakavandi B et al (2017) Magnetic adsorption separation process: an alternative method of mercury extracting from aqueous solution using modified chitosan coated Fe3O4 nanocomposites. J Chem Technol Biotechnol 92:188–200. https://doi.org/10.1002/jctb.4990
Azizian S (2004) Kinetic models of sorption: a theoretical analysis. J Colloid Interface Sci 276:47–52. https://doi.org/10.1016/j.jcis.2004.03.048
Babu BR, Bhanu SU, Meera KS (2009) Waste minimization in electroplating industries: a review. J Environ Sci Health Part C Environ Carcinog Ecotoxicol Rev 27:155–177. https://doi.org/10.1080/10590500903124158
Barakat MA (2011) New trends in removing heavy metals from industrial wastewater. Arab J Chem 4:361–377. https://doi.org/10.1016/j.arabjc.2010.07.019
Bolisetty S, Peydayesh M, Mezzenga R (2019) Sustainable technologies for water purification from heavy metals: review and analysis. Chem Soc Rev 48:463–487. https://doi.org/10.1039/c8cs00493e
Branger C, Meouche W, Margaillan A (2013) Recent advances on ion-imprinted polymers. React Funct Polym 73:859–875. https://doi.org/10.1016/j.reactfunctpolym.2013.03.021
Carlos L, Garcia Einschlag FS, González MC, Mártire DO (2013) Applications of magnetite nanoparticles for heavy metal removal from wastewater. Waste Water Treat Technol Recent Anal Dev. https://doi.org/10.5772/54608
Chung JY, Do YuS, Hong YS (2014) Environmental source of arsenic exposure. J Prev Med Public Health 47:253–257. https://doi.org/10.3961/jpmph.14.036
Deliyanni EA, Kyzas GZ, Triantafyllidis KS, Matis KA (2015) Activated carbons for the removal of heavy metal ions: a systematic review of recent literature focused on lead and arsenic ions. Open Chem 13:699–708. https://doi.org/10.1515/chem-2015-0087
Dewangan BJP, Yenkie MN (2018) Novel applications in polymers and waste management. Apple Academic Press, Toronto
Divya K, Jisha MS (2018) Chitosan nanoparticles preparation and applications. Environ Chem Lett 16:101–112. https://doi.org/10.1007/s10311-017-0670-y
Duffus JH (2002) “heavy metals” - A meaningless term? (IUPAC technical report). Pure Appl Chem 74:793–807
Egorova KS, Ananikov VP (2017) Toxicity of metal compounds: knowledge and myths. Organometallics 36:4071–4090. https://doi.org/10.1021/acs.organomet.7b00605
El-Azazy M, El-Shafie AS, Issa AA et al (2019) potato peels as an adsorbent for heavy metals from aqueous solutions: eco-structuring of a green adsorbent operating Plackett–Burman design. J Chem. https://doi.org/10.1155/2019/4926240
El-Naas MH, Al-Zuhair S, Alhaija MA (2010) Reduction of COD in refinery wastewater through adsorption on date-pit activated carbon. J Hazard Mater 173:750–757. https://doi.org/10.1016/J.JHAZMAT.2009.09.002
Elwakeel KZ (2014) Removal of arsenate from aqueous media by magnetic chitosan resin immobilized with molybdate oxoanions. Int J Environ Sci Technol 11:1051–1062. https://doi.org/10.1007/s13762-013-0307-z
Engwa GA, Ferdinand PU, Nwalo FN, Unachukwu MN (2019) Mechanism and health effects of heavy metal toxicity in humans. In: Karciolgu O (ed) Poisoning model world new tricks an old dog?. IntechOpen, London. https://doi.org/10.5772/intechopen.82511
EPA (2001) Drinking water standard for arsenic, vol 2. Environmental Protection Agency, Washington, DC
Escudero LB, Quintas PY, Wuilloud RG, Dotto GL (2019) Recent advances on elemental biosorption. Environ Chem Lett 17:409–427. https://doi.org/10.1007/s10311-018-0816-6
Fan L, Luo C, Sun M et al (2013) Highly selective adsorption of lead ions by water-dispersible magnetic chitosan/graphene oxide composites. Colloids Surfaces B Biointerfaces 103:523–529. https://doi.org/10.1016/j.colsurfb.2012.11.006
Fan HL, Zhou SF, Jiao WZ et al (2017) Removal of heavy metal ions by magnetic chitosan nanoparticles prepared continuously via high-gravity reactive precipitation method. Carbohydr Polym 174:1192–1200. https://doi.org/10.1016/j.carbpol.2017.07.050
Farghali AA, Abdel Tawab HA, Abdel Moaty SA, Khaled R (2017) Functionalization of acidified multi-walled carbon nanotubes for removal of heavy metals in aqueous solutions. J Nanostruct Chem 7:101–111. https://doi.org/10.1007/s40097-017-0227-4
Flora G, Gupta D, Tiwari A (2012) Toxicity of lead: a review with recent updates. Interdiscip Toxicol 5:47–58. https://doi.org/10.2478/v10102-012-0009-2
Freundlich HMF (1906) Over the adsorption in solution. Urnal Phys Chem 57:385–470
Fristachi A, Choudhury H (2017) Cadmium. In: Quah SR (ed) International encyclopedia of public health, 2nd edn. Elsevier, Oxford, pp 316–319
Fu F, Wang Q (2011) Removal of heavy metal ions from wastewaters: a review. J Environ Manag 92:407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
Garelick H, Jones H, Dybowska A, Valsami-Jones E (2008) Arsenic pollution sources. In: Whitacre DM (ed) Reviews of environmental contamination and toxicology. Springer, New York, pp 17–60
Ghanizadeh G, Asgari G (2011) Adsorption kinetics and isotherm of methylene blue and its removal from aqueous solution using bone charcoal. React Kinet Mech Catal 102:127–142. https://doi.org/10.1007/s11144-010-0247-2
Ghazi AM, Millette JR (1964) Lead. In: Morrison RD, Murphy BL (eds) Environmental forensics. Elsevier, pp 55–79
Guinee VF (1972) Lead poisoning. Am J Med 52:283–288. https://doi.org/10.1016/0002-9343(72)90015-0
Gupta VK, Srivastava SK, Mohan D, Sharma S (1998) Design parameters for fixed bed reactors of activated carbon developed from fertilizer waste for the removal of some heavy metal ions. Waste Manag 17:517–522. https://doi.org/10.1016/S0956-053X(97)10062-9
Hargreaves AJ, Constantino C, Dotro G et al (2018) Fate and removal of metals in municipal wastewater treatment: a review. Environ Technol Rev 7:1–18. https://doi.org/10.1080/21622515.2017.1423398
Hauptman M, Bruccoleri R, Woolf AD (2017) An update on childhood lead poisoning. Clin Pediatr Emerg Med 18:181–192. https://doi.org/10.1016/j.cpem.2017.07.010
Hayat MT, Nauman M, Nazir N et al (2019) Environmental hazards of cadmium: past, present, and future. In: Hasanuzzaman M, Prasad MNV, Fujita M (eds) Cadmium toxicity and tolerance in plants: from physiology to remediation. Elsevier Inc., pp 163–183
He YQ, Zhang NN, Wang XD (2011) Adsorption of graphene oxide/chitosan porous materials for metal ions. Chin Chem Lett 22:859–862. https://doi.org/10.1016/j.cclet.2010.12.049
He Y, Wu P, Xiao W et al (2019) Efficient removal of Pb(II) from aqueous solution by a novel ion imprinted magnetic biosorbent: adsorption kinetics and mechanisms. PLoS ONE 14:1–17. https://doi.org/10.1371/journal.pone.0213377
Ho YS, McKay G (1998) A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf Environ Prot 76:332–340. https://doi.org/10.1205/095758298529696
Honarkar H, Barikani M (2009) Applications of biopolymers I: chitosan. Monatshefte fur Chemie 140:1403–1420. https://doi.org/10.1007/s00706-009-0197-4
Hong YS, Kim YM, Lee KE (2012) Methylmercury exposure and health effects. J Prev Med Public Health 45:353–363. https://doi.org/10.3961/jpmph.2012.45.6.353
Hong M, Yu L, Wang Y et al (2019) Heavy metal adsorption with zeolites: the role of hierarchical pore architecture. Chem Eng J 359:363–372. https://doi.org/10.1016/j.cej.2018.11.087
Hughes MF (2002) Arsenic toxicity and potential mechanisms of action. Toxicol Lett 133:1–16. https://doi.org/10.1016/S0378-4274(02)00084-X
Jaishankar M, Tseten T, Anbalagan N et al (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7:60–72. https://doi.org/10.2478/intox-2014-0009
Jang Y-C, Somanna Y, Kim H (2016) Source, distribution, toxicity and remediation of arsenic in the environment—a review. Int J Appl Environ Sci ISSN 11:973–6077
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68:167–182. https://doi.org/10.1093/bmb/ldg032
Kadirvelu K, Goal J (2007) Eco-friendly technologies for removal of hazardous heavy metals from water and industrial wastewater. In: Lewinsky AA (ed) Hazardous material and wastewater. Nova Science Publisher, Hauppauge, pp 127–148
Kadouche S, Farhat M, Lounici H et al (2017) Low cost chitosan biopolymer for environmental use made from abundant shrimp wastes. Waste Biomass Valoriz 8:401–406. https://doi.org/10.1007/s12649-016-9593-2
Kahlon SK, Sharma G, Julka JM et al (2018) Impact of heavy metals and nanoparticles on aquatic biota. Environ Chem Lett 16:919–946. https://doi.org/10.1007/s10311-018-0737-4
Keng P-S, Lee S-L, Ha S-T et al (2014) Removal of hazardous heavy metals from aqueous environment by low-cost adsorption materials. Environ Chem Lett 12:15–25. https://doi.org/10.1007/s10311-013-0427-1
Khan S, Edathil AA, Banat F (2019) Sustainable synthesis of graphene-based adsorbent using date syrup. Sci Rep. https://doi.org/10.1038/s41598-019-54597-x
Kretsinger RH, Uversky VN, Permyakov EA (2013) Encyclopedia of metalloproteins, 2013th edn. Springer, New York
Kumar AKR, Saikia K, Neeraj G et al (2019) Remediation of bio-refinery wastewater containing organic and inorganic toxic pollutants by adsorption onto chitosan-based magnetic nanosorbent. Water Qual Res J. https://doi.org/10.2166/wqrj.2019.003
Kyzas GZ, Bikiaris DN (2015) Recent modifications of chitosan for adsorption applications: a critical and systematic review. Mar Drugs 13:312–337. https://doi.org/10.3390/md13010312
Kyzas GZ, Deliyanni EA (2013) Mercury(II) removal with modified magnetic chitosan adsorbents. Molecules 18:6193–6214. https://doi.org/10.3390/molecules18066193
Kyzas GZ, Matis KA (2015) Nanoadsorbents for pollutants removal: a review. J Mol Liq 203:159–168. https://doi.org/10.1016/j.molliq.2015.01.004
Lagergren S (1898) Zur Theorie der Sogenannten Adsorption Gelöster Stoffe. Kungliga Svenska Vetenskapsakademiens. Handlingar 24:139
Lambert M, Leven B, Green R (2000) New methods of cleaning up heavy metal in soils and water innovative solutions to an environmental problem. In: Environ. Sci. Technol. Briefs citizens. http://www.engg.ksu.edu/HSRC/Tosc/metals.pdf. Accessed 5 Jan 2020
Langford NJ, Ferner RE (1999) Toxicity of mercury. J Hum Hypertens 13:651–656. https://doi.org/10.1038/sj.jhh.1000896
Langmuir I (1918) The adsorption of gases on plane surfaces of glass mic and platinum. J Am Chem Soc 40:1361–1403
Lasheen MR, El-Sherif IY, Tawfik ME et al (2016) Preparation and adsorption properties of nano magnetite chitosan films for heavy metal ions from aqueous solution. Mater Res Bull 80:344–350. https://doi.org/10.1016/j.materresbull.2016.04.011
Laurent S, Forge D, Port M et al (2010) Erratum: magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications (Chemical Reviews (2008) 108 (2064)). Chem Rev 110:2574. https://doi.org/10.1021/cr900197g
Lettinga G (1995) Anaerobic digestion and wastewater treatment systems. Antonie Van Leeuwenhoek 67:3–28. https://doi.org/10.1007/BF00872193
Li B, Zhou F, Huang K et al (2017) Environmentally friendly chitosan/PEI-grafted magnetic gelatin for the highly effective removal of heavy metals from drinking water. Sci Rep 7:1–9. https://doi.org/10.1038/srep43082
Li R, Liang W, Huang H et al (2018) Removal of cadmium(II) cations from an aqueous solution with aminothiourea chitosan strengthened magnetic biochar. J Appl Polym Sci 135:1–11. https://doi.org/10.1002/app.46239
Limousin G, Gaudet JP, Charlet L et al (2007) Sorption isotherms: a review on physical bases, modeling and measurement. Appl Geochem 22:249–275. https://doi.org/10.1016/j.apgeochem.2006.09.010
Liu C, Wang B, Deng Y et al (2015) Performance of a new magnetic chitosan nanoparticle to remove arsenic and its separation from water. J Nanomater. https://doi.org/10.1155/2015/191829
Liu T, Han X, Wang Y et al (2017) Magnetic chitosan/anaerobic granular sludge composite: synthesis, characterization and application in heavy metal ions removal. J Colloid Interface Sci 508:405–414. https://doi.org/10.1016/j.jcis.2017.08.067
Lockitch G (1993) Perspectives on lead toxicity. Clin Biochem 26:371–381. https://doi.org/10.1016/0009-9120(93)90113-K
Malwal D, Gopinath P (2017) Silica stabilized magnetic-chitosan beads for removal of arsenic from water. Colloids Interface Sci Commun 19:14–19. https://doi.org/10.1016/j.colcom.2017.06.003
Mamtani R, Stern P, Dawood I, Cheema S (2011) Metals and disease: a global primary health care perspective. J Toxicol. https://doi.org/10.1155/2011/319136
Masel RI (1996) Principles of adsorption and reaction on solid surfaces. Wiley, New York
Masindi V, Muedi KL (2018) Environmental contamination by heavy metals. Heavy Met. https://doi.org/10.5772/intechopen.76082
Mccabe WL, Smith JC, Harriott P (2000) Unit operations of chemical engineering, 6th edn. Mc Graw Hill, New York
McHugh S, O’Reilly C, Mahony T et al (2003) Anaerobic granular sludge bioreactor technology. Rev Environ Sci Biotechnol 2:225–245. https://doi.org/10.1023/B:RESB.0000040465.45300.97
Mehta D, Mazumdar S, Singh SK (2015) Magnetic adsorbents for the treatment of water/wastewater—a review. J Water Process Eng 7:244–265. https://doi.org/10.1016/j.jwpe.2015.07.001
Morin-Crini N, Lichtfouse E, Torri G, Crini G (2019) Applications of chitosan in food, pharmaceuticals, medicine, cosmetics, agriculture, textiles, pulp and paper, biotechnology, and environmental chemistry. Environ Chem Lett 17:1667–1692. https://doi.org/10.1007/s10311-019-00904-x
Morsi RE, Al-Sabagh AM, Moustafa YM et al (2018) Polythiophene modified chitosan/magnetite nanocomposites for heavy metals and selective mercury removal. Egypt J Pet 27:1077–1085. https://doi.org/10.1016/j.ejpe.2018.03.004
Motornov M, Roiter Y, Tokarev I, Minko S (2008) Colloidal systems on the nanometer length scale. In: Birdi KS (ed) Handbook of surface and colloid chemistry, 3rd edn. CRC Press, Boca Raton, p 24
Nasirimoghaddam S, Zeinali S, Sabbaghi S (2015) Chitosan coated magnetic nanoparticles as nano-adsorbent for efficient removal of mercury contents from industrial aqueous and oily samples. J Ind Eng Chem 27:79–87. https://doi.org/10.1016/j.jiec.2014.12.020
Nordberg GF, Fowler BA, Nordberg M, Friberg LT (2007) Introduction-general considerations and international perspectives, 3rd edn. Elsevier Inc, Amsterdam
Nordberg GF, Nogawa K, Nordberg M (2015) Chapter 32 – Cadmium. In: Nordberg GF, Fowler BA, Nordberg M (eds) Handbook on the toxicology of metals, 4th edn. Elsevier, pp 667–716
Ojima Y, Kosako S, Kihara M et al (2019) Recovering metals from aqueous solutions by biosorption onto phosphorylated dry baker’s yeast. Sci Rep 9:1–9. https://doi.org/10.1038/s41598-018-36306-2
OSHA (2019) Toxic metals. In: United States Dep. Labor. https://www.osha.gov/SLTC/metalsheavy/. Accessed 21 Jan 2020
Putro JN, Kurniawan A, Ismadji S, Ju Y-H (2017) Nanocellulose based biosorbents for wastewater treatment: study of isotherm, kinetic, thermodynamic and reusability. Environ Nanotechnol Monit Manag 8:134–149. https://doi.org/10.1016/J.ENMM.2017.07.002
Rahmi Fathurrahmi, Lelifajri PurnamaWati F (2019) Preparation of magnetic chitosan using local iron sand for mercury removal. Heliyon 5:e01731. https://doi.org/10.1016/j.heliyon.2019.e01731
Ramesh AV, Rama Devi D, Mohan Botsa S, Basavaiah K (2018) Facile green synthesis of Fe 3 O 4 nanoparticles using aqueous leaf extract of Zanthoxylum armatum DC. for efficient adsorption of methylene blue. J Asian Ceram Soc 6:145–155. https://doi.org/10.1080/21870764.2018.1459335
Reddy DHK, Lee SM (2013) Application of magnetic chitosan composites for the removal of toxic metal and dyes from aqueous solutions. Adv Colloid Interface Sci 201–202:68–93. https://doi.org/10.1016/j.cis.2013.10.002
Ren Y, Abbood HA, He F et al (2013) Magnetic EDTA-modified chitosan/SiO2/Fe3O4 adsorbent: preparation, characterization, and application in heavy metal adsorption. Chem Eng J 226:300–311. https://doi.org/10.1016/j.cej.2013.04.059
Renu Agarwal M, Singh K (2017) Heavy metal removal from wastewater using various adsorbents: a review. J Water Reuse Desalin 7:387–419. https://doi.org/10.2166/wrd.2016.104
Rouquerol F, Rouquerol J, Sing KSW et al (2012) Adsorption by powders and porous solids, 2nd edn. Elsevier, San Diego
Ruthven DM (1984) Principals of adsorption and adsorption processes. Wiley, New York
Sari IP, Yulizar Y (2017) Green synthesis of magnetite (Fe3O4) nanoparticles using Graptophyllum pictum leaf aqueous extract. IOP Conf Ser Mater Sci Eng. https://doi.org/10.1088/1757-899X/191/1/012014
Satarug S, Garrett SH, Sens MA, Sens DA (2010) Cadmium, environmental exposure, and health outcomes. Environ Health Perspect 118:182–190. https://doi.org/10.1289/ehp.0901234
Schoolmeester L, White D (1980) Arsenic poisoning. South Med J 73:198–208
Schützendübel A, Nikolova P, Rudolf C, Polle A (2002) Cadmium and H2O2-induced oxidative stress in Populus x canescens roots. Plant Physiol Biochem 40:577–584. https://doi.org/10.1016/S0981-9428(02)01411-0
Seraj F, Rahman T (2018) Heavy metals, metalloids, their toxic effect and living systems. Am J Plant Sci 09:2626–2643. https://doi.org/10.4236/ajps.2018.913191
Shafaei F, Babaei SE, Shahvelayati AS, Honarmand Janatabadi F (2019) Biosynthesis of Fe3O4-magnetic nanoparticles using clover leaf aqueous extract: green synthesis of 1,3-benzoxazole derivatives. J Chin Chem Soc. https://doi.org/10.1002/jccs.201800489
Sharififard H, Ghorbanpour M, Hosseinirad S (2018) Cadmium removal from wastewater using nano-clay/TiO2 composite: kinetics, equilibrium and thermodynamic study. Adv Environ Technol 4:203–209. https://doi.org/10.22104/aet.2019.3029.1149
Sherlala AIA, Raman AAA, Bello MM, Buthiyappan A (2019) Adsorption of arsenic using chitosan magnetic graphene oxide nanocomposite. J Environ Manag 246:547–556. https://doi.org/10.1016/j.jenvman.2019.05.117
Singh V, Ram C (2016) Physico-chemical characterization of electroplating industrial effluents of Chandigarh and Haryana region. J Civ Environ Eng. https://doi.org/10.4172/2165-784x.1000237
Snodgrass WR (2015) Heavy metal toxicology. Encycl Food Heal. https://doi.org/10.1016/B978-0-12-384947-2.00373-1
Sun M, Cheng G, Ge X et al (2018) Aqueous Hg(II) immobilization by chitosan stabilized magnetic iron sulfide nanoparticles. Sci Total Environ 621:1074–1083. https://doi.org/10.1016/j.scitotenv.2017.10.119
Taib NI, Latif FA, Mohamed Z, Zambri NDS (2018) Green synthesis of iron oxide nanoparticles (Fe3O4-NPs) using azadirachta indica aqueous leaf extract. Int J Eng Technol 7:9–13
Tchobanoglous G, Burton FL, Stensel HD et al (2003) Wastewater engineering: treatment and reuse. McGraw-Hill Education, New York
Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ (2012) Molecular, clinical and environmental toxicology. Springer, Basel
U.S. EPA (2010) Acute exposure guideline levels (AEGLs) for mercury vapor (Hgo). In: NAC/Interim: 09/2010. https://www.epa.gov/aegl/mercury-vapor-results-aegl-program. Accessed 3 Feb 2020
Vassallo DV, Simões MR, Furieri LB et al (2011) Toxic effects of mercury, lead and gadolinium on vascular reactivity. Braz J Med Biol Res 44:939–946. https://doi.org/10.1590/S0100-879X2011007500098
Venkateswarlu S, Yoon M (2015) Rapid removal of cadmium ions using green-synthesized Fe3O4 nanoparticles capped with diethyl-4-(4 amino-5-mercapto-4H-1,2,4-triazol-3-yl)phenyl phosphonate. RSC Adv 5:65444–65453. https://doi.org/10.1039/c5ra10628a
Vianna AS, de Matos EP, de Jesus IM et al (2019) Human exposure to mercury and its hematological effects: a systematic revie. Cad Saude Publica 35:1–22. https://doi.org/10.1590/0102-311x00091618
Viju Kumar VG, Prem AA (2018) Green synthesis and characterization of iron oxide nanoparticles using Phyllanthus niruri extract. Orient J Chem 34:2583–2589. https://doi.org/10.13005/ojc/340547
Virgen M del RM, Vázquez OFG, Montoya VH, Gómez RT (2018) Removal of heavy metals using adsorption processes subject to an external magnetic field. In: Saleh HE-DM, Aglan RF (eds) Heavy metals. InTech, p 13
Vu D, Li X, Wang C (2013) Adsorption of As(III) from aqueous solution based on porous magnetic/chitosan/ferric hydroxide microspheres prepared via electrospraying. Sci China Chem 56:678–684. https://doi.org/10.1007/s11426-012-4817-1
Wang J, Xu W, Chen L et al (2014) Preparation and evaluation of magnetic nanoparticles impregnated chitosan beads for arsenic removal from water. Chem Eng J 251:25–34. https://doi.org/10.1016/j.cej.2014.04.061
Wang Y, Wu D, Wei Q et al (2017) Rapid removal of Pb(II) from aqueous solution using branched polyethylenimine enhanced magnetic carboxymethyl chitosan optimized with response surface methodology. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-09700-5
Wani AL, Ara A, Usmani JA (2015) Lead toxicity: a review. Interdiscip Toxicol 8:55–64. https://doi.org/10.1515/intox-2015-0009
WHO (2010) Ten chemicals of major public health concern. WHO, Geneva, pp 1–4
WHO (2018) Arsenic. In: World heal. Organ. https://www.who.int/en/news-room/fact-sheets/detail/arsenic. Accessed 20 Dec 2019
Worch E (2012) Adsorption technology in water treatment: fundamentals, processes, and modeling. Walter de Gruyter, Berlin
Wu X, Cobbina SJ, Mao G et al (2016) A review of toxicity and mechanisms of individual and mixtures of heavy metals in the environment. Environ Sci Pollut Res 23:8244–8259. https://doi.org/10.1007/s11356-016-6333-x
Yang P-F, Lee C-K (2007) Hyaluronic acid interaction with chitosan-conjugated magnetite particles and its purification. Biochem Eng J 33:284–289. https://doi.org/10.1016/j.bej.2006.11.010
Yang G, Tang L, Lei X et al (2014) Cd(II) removal from aqueous solution by adsorption on α-ketoglutaric acid-modified magnetic chitosan. Appl Surf Sci 292:710–716. https://doi.org/10.1016/j.apsusc.2013.12.038
Yew YP, Shameli K, Miyake M et al (2018) Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: a review. Arab J Chem. https://doi.org/10.1016/j.arabjc.2018.04.013
Ynalvez R, Gutierrez J, Gonzalez-Cantu H (2016) Mini-review: toxicity of mercury as a consequence of enzyme alteration. Biometals 29:781–788. https://doi.org/10.1007/s10534-016-9967-8
Yu JG, Yue BY, Wu XW et al (2016) Removal of mercury by adsorption: a review. Environ Sci Pollut Res 23:5056–5076. https://doi.org/10.1007/s11356-015-5880-x
Zhang L, Zeng Y, Cheng Z (2016) Removal of heavy metal ions using chitosan and modified chitosan: a review. J Mol Liq 214:175–191. https://doi.org/10.1016/j.molliq.2015.12.013
Zhao M, Xu Y, Zhang C et al (2016) New trends in removing heavy metals from wastewater. Appl Microbiol Biotechnol 100:6509–6518. https://doi.org/10.1007/s00253-016-7646-x
Zhu W, Wang J, Wu D et al (2017) Investigating the heavy metal adsorption of mesoporous silica materials prepared by microwave synthesis. Nanoscale Res Lett. https://doi.org/10.1186/s11671-017-2070-4
Zou W, Geng H, Lin M, Xiong X (2012) Facile one-pot preparation of superparamagnetic chitosan sphere and its derived hollow sphere. J Appl Polym Sci 123:3587–3594. https://doi.org/10.1002/app.34887
Acknowledgements
The authors are grateful for the financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) (Proc. 2017/18236-1), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (406193/2018-5) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brião, G.d., de Andrade, J.R., da Silva, M.G.C. et al. Removal of toxic metals from water using chitosan-based magnetic adsorbents. A review. Environ Chem Lett 18, 1145–1168 (2020). https://doi.org/10.1007/s10311-020-01003-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-020-01003-y