Abstract
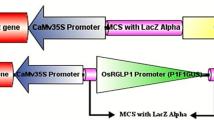
The expression analysis of GhNAC4, a NAC (NAM, ATAF1-2, and CUC2) domain-containing transcription factor of the upland cotton, Gossypium hirsutum was carried out. Its expression was up-regulated by abscisic acid, cytokinin, methyl jasmonic acid, gibberellic acid, auxin, and ethylene. Its expression was also highly induced by drought, osmotic, oxidative, salinity, high and low-temperature stresses, and wounding. To corroborate these observations, we cloned the promoter of GhNAC4 and fused it transcriptionally with uidA (GUS) gene for studies in transgenic tobacco. Fluorometric GUS analysis of the transgenic plants revealed that it is also induced by various phytohormones and environmental stresses. The spatio-temporal analysis of the GhNAC4 promoter revealed that GUS expression was active in all stages of plant development including the reproductive organs. In the mature plant, the GhNAC4 is expressed at a higher level in vascular bundles and guard cells. We also observed intense expression in other cells upon wounding. These observations were supported by a detailed bioinformatic analysis of the GhNAC4 promoter for identifying the cis-acting elements that are associated with the regulation of gene expression in a tissue-specific and induced manner.
Key message
GhNAC4 gene is induced by phytohormones and environmental stress treatments in cotton. It is expressed throughout a mature plant, including the reproductive organs, vascular bundles, and guard cells.






Similar content being viewed by others
Data availability
All the data presented in the present manuscript are freely available to the interested researchers and would be made available upon request to the Corresponding author-VST (email- strishlajain@gmail.com).
Abbreviations
- 4-MU:
-
4-Methylumbelliferone
- 4-MUG/MUG:
-
4-Methyl-umbelliferyl-β-d-glucuronide
- ABA:
-
Abscisic acid
- BAP:
-
6-Benzyl aminopurine
- GA:
-
Gibberellic acid
- GUS:
-
Β-glucuronidase
- IAA:
-
Indole-3-acetic acid
- JA:
-
Jasmonic acid
- MeJA:
-
Methyl jasmonic acid
- MV:
-
Methyl viologen
- NAC:
-
NAM ATAF CUC
- PEG:
-
Polyethylene glycol
- SA:
-
Salicylic acid
- TF:
-
Transcription factor
References
Abe H, Urao T, Ito T, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 15:63–78. https://doi.org/10.1105/tpc.006130
Al-Abdallat AM, Ali-Sheikh-Omar MA, Alnemer LM (2015) Overexpression of two ATNAC3-related genes improves drought and salt tolerance in tomato (Solanum lycopersicum L.). Plant Cell Tiss Organ Cult 120:989–1001. https://doi.org/10.1007/s11240-014-0652-8
Baniwal SK, Bharti K, Chan KY, Fauth M, Ganguli A, Kotak S et al (2004) Heat stress response in plants: a complex game with chaperones and more than twenty heat stress transcription factors. J Biosciences 29:471–487. https://doi.org/10.1007/BF02712120
Bate N, Twell D (1998) Functional architecture of a late pollen promoter: pollen-specific transcription is developmentally regulated by multiple stage-specific and co-dependent activator elements. Plant Mol Biol 37:859–869. https://doi.org/10.1023/A:1006095023050
Bhattacharjee S (2005) Reactive oxygen species and oxidative burst: roles in stress, senescence and signal transduction in plants. Curr Sci 89:1113–1121
Bielach A, Hrtyan M, Tognetti VB (2017) Plants under stress: involvement of auxin and cytokinin. Int J Mol Sci 18:1427. https://doi.org/10.3390/ijms18071427
Boter M, Ruíz-Rivero O, Abdeen A, Prat S (2004) Conserved MYC transcription factors play a key role in jasmonate signaling both in tomato and Arabidopsis. Genes Dev 18:1577–1591. https://doi.org/10.1101/gad.297704
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bu Q, Jiang H, Li CB, Zhai Q, Zhang J, Wu X et al (2008) Role of the Arabidopsis thaliana NAC transcription factors ANAC019 and ANAC055 in regulating jasmonic acid-signaled defense responses. Cell Res 18:756–767. https://doi.org/10.1038/cr.2008.53
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116. https://doi.org/10.1007/BF02670468
Chang WC, Lee TY, Huang HD, Huang HY, Pan RL (2008) PlantPAN: plant promoter analysis navigator, for identifying combinatorial cis-regulatory elements with distance constraint in plant gene groups. BMC Genomics 9:561. https://doi.org/10.1186/1471-2164-9-561
Elmayan T, Tepfer M (1995) Evaluation in tobacco of the organ specificity and strength of the rolD promoter, domain A of the 35S promoter and the 35S2 promoter. Transgenic Res 4:388–396. https://doi.org/10.1007/BF01973757
Ernst HA, Olsen AN, Skriver K, Larsen S, Lo Leggio L (2004) Structure of the conserved domain of ANAC, a member of the NAC family of transcription factors. EMBO Rep 5:297–303. https://doi.org/10.1038/sj.embor.7400093
Erpen L, Devi HS, Grosser JW, Dutt M (2018) Potential use of the DREB/ERF, MYB, NAC and WRKY transcription factors to improve abiotic and biotic stress in transgenic plants. Plant Cell Tiss Organ Cult 132:1–125. https://doi.org/10.1007/s11240-017-1320-6
Fang Y, You J, Xie K, Xie W, Xiong L (2008) Systematic sequence analysis and identification of tissue-specific or stress-responsive genes of NAC transcription factor family in rice. Mol Genet Genomics 280:547–563. https://doi.org/10.1007/s00438-008-0386-6
Fehlberg V, Vieweg MF, Dohmann EMN, Hohnjec N, Pühler A, Perlick AM, Küster H (2005) The promoter of the leghaemoglobin gene VfLb29: functional analysis and identification of modules necessary for its activation in the infected cells of root nodules and in the arbuscule-containing cells of mycorrhizal roots. J Exp Bot 413:799–806. https://doi.org/10.1093/jxb/eri074
Fusada N, Masuda T, Kuroda H, Shimada H, Ohta H, Takamiya KI (2005) Identification of a novel cis-element exhibiting cytokinin-dependent protein binding in vitro in the 5′-region of NADPH-protochlorophyllide oxidoreductase gene in cucumber. Plant Mol Biol 59:631–645. https://doi.org/10.1007/s11103-005-0579-x
Gowik U, Burscheidt J, Akyildiz M, Schlue U, Koczor M, Streubel M, Westhoff P (2004) cis-regulatory elements for mesophyll-specific gene expression in the C 4 plant Flaveria trinervia, the promoter of the C 4 phosphoenolpyruvate carboxylase gene. Plant Cell 16:1077–1090. https://doi.org/10.1105/tpc.019729
Guo Y, Gan S (2006) AtNAP, a NAC family transcription factor, has an important role in leaf senescence. Plant J 46:601–612. https://doi.org/10.1111/j.1365-313X.2006.02723.x
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res 27:297–300. https://doi.org/10.1093/nar/27.1.297
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231. https://doi.org/10.1126/science.227.4691.1229
Hu H, Dai M, Yao J, Xiao B, Li X, Zhang Q, Xiong L (2006) Overexpressing a NAM, ATAF, and CUC (NAC) transcription factor enhances drought resistance and salt tolerance in rice. Proc Natl Acad Sci USA 103:12987–12992. https://doi.org/10.1073/pnas.0604882103
Hu R, Qi G, Kong Y, Kong D, Gao Q, Zhou G (2010) Comprehensive analysis of NAC domain transcription factor gene family in Populus trichocarpa. BMC Plant Biol 10:145. https://doi.org/10.1186/1471-2229-10-145
Jefferson RA (1989) The GUS reporter gene system. Nature 342:837–838. https://doi.org/10.1038/342837a0
Joshi CP (1987) An inspection of the domain between putative TATA box and translation start site in 79 plant genes. Nucleic Acids Res 15:6643–6653. https://doi.org/10.1093/nar/15.16.6643
Kiełbowicz-Matuk A (2012) Involvement of plant C2H2-type zinc finger transcription factors in stress responses. Plant Sci 185–186:78–85. https://doi.org/10.1016/j.plantsci.2011.11.015
Ko JH, Yang SH, Park AH, Lerouxel O, Han KH (2007) ANAC012, a member of the plant-specific NAC transcription factor family, negatively regulates xylary fiber development in Arabidopsis thaliana. Plant J 50:1035–1048. https://doi.org/10.1111/j.1365-313X.2007.03109.x
Kuppu S, Mishra N, Hu R, Sun L, Zhu X, Shen G et al (2013) Water-deficit inducible expression of a cytokinin biosynthetic gene IPT improves drought tolerance in cotton. PLoS ONE 8:e64190. https://doi.org/10.1371/journal.pone.0064190
Le DT, Nishiyama R, Watanabe Y, Mochida K, Yamaguchi-Shinozaki K, Shinozaki K, Tran LSP (2011) Genome-wide survey and expression analysis of the plant-specific NAC transcription factor family in soybean during development and dehydration stress. DNA Res 18:263–276. https://doi.org/10.1093/dnares/dsr015
Le Hir R, Bellini C (2013) The plant-specific Dof transcription factors family: new players involved in vascular system development and functioning in arabidopsis. Front Plant Sci 4:164. https://doi.org/10.3389/fpls.2013.00164
Lescot M, Dehais P, Thijs G, Marchal K, Van de Peer Y, Rouze P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Lessard PA, Allen RD, Bernier F, Crispino JD, Fujiwara T, Beachy RN (1991) Multiple nuclear factors interact with upstream sequences of differentially regulated beta-conglycinin genes. Plant Mol Biol 16:397–413. https://doi.org/10.1007/BF00023991
Livak K, Schmittgen T (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(T)(-Delta Delta C) method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lu M, Zhang DF, Shi YS, Song YC, Wang TY, Li Y (2013) Expression of SbSNAC1, a NAC transcription factor from sorghum, confers drought tolerance to transgenic Arabidopsis. Plant Cell Tiss Organ Cult 115:443–455. https://doi.org/10.1007/s11240-013-0375-2
Meng C, Cai C, Zhang T, Guo W (2009) Characterization of six novel NAC genes and their responses to abiotic stresses in Gossypium hirsutum L. Plant Sci 176:352–359. https://doi.org/10.1016/j.plantsci.2008.12.003
Mitsuda N, Seki M, Shinozaki K, Ohme-Takagi M (2005) The NAC transcription factors NST1 and NST2 of arabidopsis regulate secondary wall thickenings and are required for anther dehiscence. Plant Cell 17:2993–3006. https://doi.org/10.1105/tpc.105.036004
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nakashima K, Tran LSP, Van Nguyen D, Fujita M, Maruyama K, Todaka D et al (2007) Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and *biotic stress-responsive gene expression in rice. Plant J 51:617–630. https://doi.org/10.1111/j.1365-313X.2007.03168.x
Negi S, Tak H, Ganapathi TR (2016) Expression analysis of MusaNAC68 transcription factor and its functional analysis by overexpression in transgenic banana plants. Plant Cell Tiss Organ Cult 125:59–70. https://doi.org/10.1007/s11240-015-0929-6
Nishiuchi T, Shinshi H, Suzuki K (2004) Rapid and transient activation of transcription of the ERF3 gene by wounding in tobacco leaves: possible involvement of NtWRKYs and autorepression. J Biol Chem 279:55355–55361. https://doi.org/10.1074/jbc.M409674200
Nuruzzaman M, Manimekalai R, Sharoni AM, Satoh K, Kondoh H, Ooka H, Kikuchi S (2010) Genome-wide analysis of NAC transcription factor family in rice. Gene 456:30–44. https://doi.org/10.1016/j.gene.2010.06.008
Ohnishi T, Sugahara S, Yamada T, Kikuchi K, Yoshiba Y, Hirano HY, Tsutsumi N (2005) OsNAC6, a member of the NAC gene family, is induced by various stresses in rice. Genes Genet Syst 80:135–139. https://doi.org/10.1266/ggs.80.135
Olsen AN, Ernst HA, Leggio LL, Skriver K (2005) NAC transcription factors: structurally distinct, functionally diverse. Trends Plant Sci 10:79–87. https://doi.org/10.1016/j.tplants.2004.12.010
Park J, Kim YS, Kim SG, Jung JH, Woo JC, Park CM (2011) Integration of auxin and salt signals by the NAC transcription factor NTM2 during seed germination in arabidopsis. Plant Physiol 156:537–549. https://doi.org/10.1104/pp.111.177071
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14:290–295. https://doi.org/10.1016/j.pbi.2011.02.001
Plesch G, Ehrhardt T, Mueller-Roeber B (2001) Involvement of TAAAG elements suggests a role for Dof transcription factors in guard cell-specific gene expression. Plant J 28:455–464. https://doi.org/10.1046/j.1365-313X.2001.01166.x
Puranik S, Bahadur RP, Srivastava PS, Prasad M (2011) Molecular cloning and characterization of a membrane associated NAC family gene, SiNAC from foxtail millet [Setaria italica (L.) P. Beauv]. Mol Biotechnol 49:138–150. https://doi.org/10.1007/s12033-011-9385-7
Puranik S, Sahu PP, Mandal SN, Parida SK, Prasad M (2013) Comprehensive genome-wide survey, genomic constitution and expression profiling of the NAC transcription factor family in foxtail millet (Setaria italica L.). PLoS ONE 8(5):64594. https://doi.org/10.1371/journal.pone.0064594
Rogers HJ, Bate N, Combe J, Sullivan J, Sweetman J, Swan C et al (2001) Functional analysis of cis-regulatory elements within the promoter of the tobacco late pollen gene g10. Plant Mol Biol 45:577–585. https://doi.org/10.1023/A:1010695226241
Rosa YBCJ, Aizza LCB, Bello CCM, Dornelas MC (2013) The PmNAC1 gene is induced by auxin and expressed in differentiating vascular cells in callus cultures of Passiflora. Plant Cell Tiss Organ Cult 115:275–283. https://doi.org/10.1007/s11240-013-0360-9
Sablowski RWM, Meyerowitz EM (1998) A homolog of NO APICAL MERISTEM is an immediate target of the floral homeotic genes APETALA3/PISTILLATA. Cell 531:220–234. https://doi.org/10.1016/j.gene.2013.09.007
Shah ST, Pang C, Fan S, Song M, Arain S, Yu S (2013) Isolation and expression profiling of GhNAC transcription factor genes in cotton (Gossypium hirsutum L.) during leaf senescence and in response to stresses. Gene 20:403–423. https://doi.org/10.1093/dnares/dst019
Shahmuradov IA, Umarov RK, Solovyev VV (2017) TSSPlant: a new tool for prediction of plant Pol II promoters. Nucleic Acids Res USA 88:7266–7270. https://doi.org/10.1073/pnas.88.16.7266
Skriver K, Olsen FL, Rogers JC, Mundy J (1991) cis-acting DNA elements responsive to gibberellin and its antagonist abscisic acid. Proc Natl Acad Sci USA 88:7266–7270. https://doi.org/10.1073/pnas.88.16.7266
Souer E, Van Houwelingen A, Kloos D, Mol J, Koes R (1996) The no apical Meristem gene of petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell 85:159–170. https://doi.org/10.1016/S0092-8674(00)81093-4
Sperotto RA, Ricachenevsky FK, Duarte GL, Boff T, Lopes KL, Sperb ER et al (2009) Identification of up-regulated genes in flag leaves during rice grain filling and characterization of OsNAC5, a new ABA-dependent transcription factor. Planta 230:985–1002. https://doi.org/10.1007/s00425-009-1000-9
Stender EG, O’Shea C, Skriver K (2015) Subgroup-specific intrinsic disorder profiles of arabidopsis NAC transcription factors: Identification of functional hotspots. Plant Signal Behav 10:6. https://doi.org/10.1080/15592324.2015.1010967
Stockinger EJ, Gilmour SJ, Thomashow MF (1997) Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat. Proc Natl Acad Sci USA 94:1035–1040. https://doi.org/10.1073/pnas.94.3.1035
Sun H, Hu M, Li J, Chen L, Li M, Zhang S et al (2018) Comprehensive analysis of NAC transcription factors uncovers their roles during fiber development and stress response in cotton. BMC Plant Biol 18:1–15. https://doi.org/10.1186/s12870-018-1367-5
Takasaki H, Maruyama K, Kidokoro S, Ito Y, Fujita Y, Shinozaki K et al (2010) The abiotic stress-responsive NAC-type transcription factor OsNAC5 regulates stress-inducible genes and stress tolerance in rice. Mol Genet Genomics 284:173–183. https://doi.org/10.1007/s00438-010-0557-0
Wang NN, Shin MC, Li N (2005) The GUS reporter-aided analysis of the promoter activities of Arabidopsis ACC synthase genes AtACS4, AtACS5, and AtACS7 induced by hormones and stresses. J Exp Bot 56:909–920. https://doi.org/10.1093/jxb/eri083
Wang L, Hu Z, Zhu M, Zhu Z, Hu J, Qanmber G, Chen G (2017) The abiotic stress-responsive NAC transcription factor SlNAC11 is involved in drought and salt response in tomato (Solanum lycopersicum L.). Plant Cell Tiss Organ Cult 129:161–174. https://doi.org/10.1007/s11240-017-1167-x
Xia N, Zhang G, Liu XY, Deng L, Cai GL, Zhang Y et al (2010) Characterization of a novel wheat NAC transcription factor gene involved in defense response against stripe rust pathogen infection and abiotic stresses. Mol Biol Rep 37:3703–3712. https://doi.org/10.1007/s11033-010-0023-4
Xie Q, Frugis G, Colgan D, Chua NH (2000) Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes Dev 14:3024–3036. https://doi.org/10.1101/gad.852200
Xue GP (2002) An AP2 domain transcription factor HvCBF1 activates expression of cold-responsive genes in barley through interaction with a (G/a)(C/t)CGAC motif. BBA-Gene Regul Mech 1577:63–72. https://doi.org/10.1016/S0167-4781(02)00410-4
Yin Y, Chen L, Beachy R (1997) Promoter elements required for phloem-specific gene expression from the RTBV promoter in rice. Plant J 12:1179–1188. https://doi.org/10.1046/j.1365-313X.1997.12051179.x
Yokoyama A, Yamashino T, Amano YI, Tajima Y, Imamura A, Sakakibara H, Mizuno T (2007) Type-B ARR transcription factors, ARR10 and ARR12, are implicated in cytokinin-mediated regulation of protoxylem differentiation in roots of Arabidopsis thaliana. Plant Cell Physiol 48:84–96. https://doi.org/10.1093/pcp/pcl040
Zhan PL, Ke SW, Zhang PY, Zhou CC, Fu BL, Zhang XQ, Zhong TX, Chen S, Xie XM (2018) Overexpression of two cold-responsive ATAF-like NAC transcription factors from fine-stem stylo (Stylosanthes guianensis var. intermedia) enhances cold tolerance in tobacco plants. Plant Cell Tiss Organ 135:545–558. https://doi.org/10.1007/s11240-018-1486-6
Acknowledgements
VST would like to acknowledge Vikas Kumar Jain and Manimaran Panneer Selvam for the critical reading of the manuscript. The authors are grateful to the Head, Department of Plant Sciences, University of Hyderabad for the facilities under various umbrella programs like DST-FIST and UGC-SAP-DRS.
Funding
VST would like to acknowledge the Council of scientific and industrial research (CSIR), New Delhi for research fellowship (20-6/2009(i)EU-IV).
Author information
Authors and Affiliations
Contributions
VST and PBK conceived and designed the experiments. VST, SM, and PB performed the experiments. VST analyzed the data. VST and PBK wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
This investigation does not necessitate the use of animals and hence, does not need the approval from the Animal Ethics Committee.
Consent to participate
The present manuscript is the result of the participation of all the authors listed in it.
Consent for publication
The authors of the manuscript hereby give their consent for publication of the manuscript in PCTOC.
Additional information
Communicated by Barbara Mary Doyle Prestwich.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Trishla, V.S., Marriboina, S., Boyidi, P. et al. GUS-reporter based analysis of the promoter activity of Gossypium hirsutum NAC transcription factor, GhNAC4 that is induced by phytohormones and environmental stresses. Plant Cell Tiss Organ Cult 141, 643–654 (2020). https://doi.org/10.1007/s11240-020-01825-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01825-2