Abstract
Introduction
The production of the whiteleg shrimp Litopenaeus vannamei now accounts for approximately 75% of the total shrimp production in Indonesia. The techniques used to produce whiteleg shrimp in Indonesia are still dominated by conventional rearing strategies using open-pond systems, which often contribute to unpredictable culture performance and weak sustainability. Alternative production strategies of closed aquaculture systems, including the recirculating aquaculture system (RAS) and hybrid zero water discharge-recirculating aquaculture system (hybrid system), have been developed and implemented for higher productivity, stability and sustainability of whiteleg shrimp grow-out production in Indonesia. Despite the positive aspects of the application of closed aquaculture systems in shrimp aquaculture, the differences in the characteristics of shrimp grown in closed RAS and hybrid systems compared to open-pond systems remain unclear.
Objective
This study aims to investigate the differences in the metabolite profiles of shrimp grown in intensive closed aquaculture systems, including an RAS and hybrid system, compared to those of shrimp grown in a semi-intensive, open, earthen pond system by means of non-targeted GC–MS metabolite profiling.
Methods
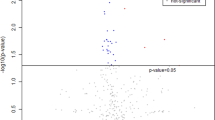
Shrimp cultured in the closed systems (RAS and hybrid system) and an open system (pond) were harvested and subjected to GC–MS non-targeted metabolomics analysis. A total of 112 metabolites were annotated from shrimp samples and subjected to principal component analysis (PCA).
Results
The metabolites annotated from GC–MS mainly included organic compounds, proteinogenic and non-proteinogenic amino acids, sugars, nucleosides and fatty acids. The results of principal component analysis showed several metabolites with high variable importance in projection (VIP) scores, including shikimic acid, β-alanine, uric acid, hypoxanthine, inosine, homocysteine, methionine, phenylalanine, tryptophan and lysine, as the main metabolites differentiating the shrimp grown in the three production systems.
Conclusion
Our findings showed that shrimp cultured in different aquaculture systems exhibited distinct metabolite profiles, and the metabolites showing high VIP scores, including shikimic acid, β-alanine, uric acid, hypoxanthine, inosine, homocysteine, methionine, phenylalanine, tryptophan and lysine, may serve as candidate markers to indicate the differences in shrimp from different production systems.




Similar content being viewed by others
References
Bai, J. R., Wu, Y. P., Liu, X. Y., Zhong, K., Huang, Y. N., & Gao, H. (2015). Antibacterial activity of shikimic acid from pine needles of Cedrus deodara against Staphylococcus aureus through damage to cell membrane. International Journal of Molecular Sciences,16, 27145–27155.
Bostock, J., Lane, A., Hough, C., & Yamamoto, K. (2016). An Assessment of the economic contribution of EU aquaculture production and the influence of policies for its sustainable development. Aquaculture International,24, 699–733.
Cao, M., Wang, X. Q., Yan, B. L., & Ma, S. (2006). Effects of stocking density, feeding frequency on survival and growth of white shrimp. Reservoir Fisheries,26(2), 34–35.
Ebeling, J. M., & Timmons, M. B. (2012). Chapter 11: Recirculating aquaculture systems, aquaculture production system (1st ed.). Hoboken: Wiley.
FAO. (2018). The state of world fisheries and aquaculture: Meeting The sustainable development goals (p, 227). Rome.
Fan, W., Ye, Y., Chen, Z., Shao, Y., Xie, X., Zhang, W., et al. (2016). Metabolic product response profiles of Cherax quadricarinatus towards white spot syndrome virus infection. Developmental & Comparative Immunology,61, 236–241.
Fumagalli, M., Lecca, D., Abbracchio, M. P., & Ceruti, S. (2017). Pathophysiological role of purines and pyrimidines in neurodevelopment: unveiling new pharmacological approaches to congenital brain diseases. Frontiers in Pharmacology,8, 941.
Gao, W., Tian, L., Huang, T., Yao, M., Hu, W., & Xu, Q. (2016). Effect of salinity on the growth performance, osmolarity and metabolism-related gene expression in white shrimp Litopenaeus vannamei. Aquaculture Report,4, 125–129.
Gao, Y., He, Z., Vector, H., Zhao, B., Li, Z., He, J., et al. (2017). Effect of stocking density on growth, oxidative stress and HSP 70 of pacific white shrimp Litopenaeus vannamei. Turkish Journal of Fisheries and Aquatic Sciences,17, 877–884.
Giovannini, L. (2008). Immunomodulatory activity of shikimic acid and quercitin in comparison with oseltamivir (Tamiflu) in an ‘in vitro’ model. Journal of Medical Virology,80, 741–745.
Kitaoka, C., Yamamoto, J., Sakamiya, A. K., Yano, T., & Yoshinaga, Y. K. (2016). The effects of baits and water temperature on taste components after storage of the Pacific oyster, Crassostrea gigas. Japanese Journal of Food Chemistry and Safety,23(3), 133–140.
Lee, W.-C., & Chen, J.-C. (2003). Hemolymph ammonia, urea and uric acid levels and nitrogenous excretion of Marsupenaeus japonicas at different salinity levels. Journal of Experimental Marine Biology and Ecology,288, 39–49.
Lin, Y.-C., Chen, C.-J., Chen, Y.-Y., Yeh, S.-T., Chen, L.-L., Huang, C. L., et al. (2015). Crowding of white shrimp Litopenaeus vananmei depresses their immunity to and resistance against Vibrio alginolyticus and white spot syndrome virus. Fish & Shellfish Immunology,45, 104–111.
Martins, C. I. M., Eding, E. H., Verdegem, M. C. J., Heinsbroek, L. T. N., Schneider, O., Blancheton, J. P., et al. (2010). New Developments in recirculating aquaculture systems in Europe: A perspective on environmental sustainability. Aquacultural Engineering,43(3), 83–93.
Muhammad, H., Situmorang, M. L., Djohan, Y. A., Aditiawati, P., & Suantika, G. (2016). Biological, technical, and financial feasibilities study of zero water discharge (ZWD) system application in low salinity white shrimp (Litopenaeus vannamei Boone) urban aquaculture, study case: Gresik District, East Java, Indonesia. Journal of Fisheries & Livestock Production,4, 197.
National Research Council. (2011). Nutrient requirements of fish and shrimp. Washington, DC: The National Academies Press.
Perez-Velazquez, M., Gonzáléz-Félix, M. L., Jaimes-Bustamente, F., Martínez-Córdova, L. R., Trujillo-Villalba, D. A., & Davis, D. A. (2007). Investigation of the effects of salinity and dietary protein level on growth and survival of pacific white shrimp Litopenaeus vannamei. Journal of the World Aquaculture Society,38, 475–485.
Putri, S. P., Nakayama, Y., Matsuda, F., Uchikata, T., Kobayashi, S., Matsubara, A., et al. (2013a). Current metabolomics: Practical applications. Journal of Bioscience and Bioengineering,115(6), 579–589.
Putri, S. P., Yamamoto, S., Tsugawa, H., & Fukusaki, E. (2013b). Current metabolomics: Technological advances. Journal of Bioscience and Bioengineering,116(1), 9–16.
Saroglia, M., & Liu, Z. (2012). Functional genomics in aquaculture (p. 397). Iowa, USA: Wiley.
Schock, T. B., Duke, J., Goodson, A., Weldon, D., Brunson, J., Leffler, J. W., et al. (2013). Evaluation of pacific white shrimp (Litopenaeus vannamei) health during a superintensive aquaculture growout using NMR-based metabolomics. PLoS ONE,8(11), 10.
Shan, H. W., Dong, Y., Ma, S., Zhou, Y. G., & Ma, Z. Y. (2018). Effects of dietary supplementation with freeze-dried powder of Ampithoe sp. on the growth performance, energy metabolism, and ammonia-nitrogen tolerance of the Pacific white shrimp Litopenaeus vannamei. Aquaculture Research,49, 2633–2643.
Shan, H., Geng, Z., Ma, S., & Wang, T. (2019). Comparative study of the key enzymes and biochemical substances involved in the energy metabolism of Pacific white shrimp, Litopenaeus vannamei, with different ammonia-N tolerances. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology,221, 73–81.
Situmorang, M. L., Suantika, G., Santoso, M., Khakim, A., Wibowo, I., Aditiawati, P., et al. (2020). Poly-β-Hydroxybutyrate (PHB) improves nursery-phase Pacific white shrimp Litopenaeus vannamei defense against vibriosis. North American Journal of Aquaculture, 82(1), 108–114.
Suantika, G., Lumbantoruan, G., Muhammad, H., Azizah, F. F. N., & Aditiawati, P. (2015). Performance of zero water discharge (ZWD) system with nitrifying bacteria and microalgae Chaetoceros calcitrans components in super intensive white shrimp (Litopenaeus vannamei) culture. Journal of Aquaculture Research and Development,6, 359.
Suantika, G., Pratiwi, M. I., Situmorang, M. L., Djohan, Y. A., Muhammad, H., & Astuti, D. I. (2016). Ammonium removal by nitrifying bacteria biofilm on limestone and bioball substrate established in freshwater trickling biofilter. Poultry, Fisheries & Wildlife Sciences, 4(2), 157.
Suantika, G., Situmorang, M. L., Nurfathurahmi, A., Taufik, I., Aditiawati, P., Yusuf, N., et al. (2018a). Application of indoor recirculation aquaculture system for white shrimp Litopenaeus vannamei grow-out super-intensive culture at low salinity condition. Journal of Aquaculture Research and Development,9, 1–6.
Suantika, G., Situmorang, M. L., Kurniawan, J. B., Pratiwi, S. A., Aditiawati, P., Astuti, D. I., et al. (2018b). Development of a zero water discharge (ZWD)—Recirculating aquaculture system (RAS) hybrid system for super-intensive white shrimp (Litopenaeus vannamei) culture under low salinity conditions and its industrial trial in commercial shrimp urban farming in Gresik, East Java, Indonesia. Aquacultural Engineering,82, 12–24.
Suantika, G., Situmorang, M. L., Aditiawati, P., Astuti, D. I., Azizah, F. F. N., & Muhammad, H. (2018c). Closed aquaculture system: Zero water discharge for shrimp and prawn farming in Indonesia. Biological Resources of Water. https://doi.org/10.5772/intechopen.69758.
Suantika, G., Turendro, O. R., & Situmorang, M. L. (2017). Use of nitrifying bacteria for promoting giant freshwater prawn (Macrobrachium rosenbergii de Man) nursery phase in indoor system. Journal of Fisheries & Livestock Production,5(2), 228.
Sun, J.-Y., You, C.-Y., Dong, K., You, H.-S., & Xing, J.-F. (2016). Anti-inflammatory, analgesic and antioxidant activities of 3,4-oxoisopropylidene-shikimic acid. Pharmaceutical Biology,54, 2282–2287.
Tacon, A. G. J., Jory, D. E., & Nunes, A. J. P. (2013). Shrimp feed management: issues and perspectives. In: M.R. Hasan and M.B. New, eds. On-farm feeding and feed management in aquaculture. FAO Fisheries and Aquaculture Technical Paper No. 583 (pp. 481–488). Rome: FAO.
Wang, Z., Qu, Y., Yan, M., Li, J., Zou, J., & Fan, L. (2019). Physiological Responses Of Pacific White Shrimp Litopenaeus vannamei to temperature fluctuation in low-salinity water. Frontiers in Physiology,10, 1025.
Xing, J. F., Sun, J. Y., You, H. S., Lv, J., Sun, J. N., & dan Dong, Y. L. (2012). Anti-inflammatory effect of 3,4-oxo-isopropylidene-shikimic acid on acetic acid-induced colitis in rats. Inflammation,35, 1872–1879.
Zhang, W., Yang, Q., Tan, B., Wang, F., Dong, X., Chi, S., et al. (2019). Study of the requirements of dietary cholesterol at two different growth stages of Pacific white shrimps Litopenaeus vannamei. Aquaculture International,27(6), 1583–1597.
Acknowledgements
The authors would like to thank PT. Gapura Akua Kultiva, Bandung, Indonesia, for the supply of the nitrification bacteria culture for the RAS, as well as PT. Suri Tani Pemuka, Indramayu, Indonesia, for the supply of shrimp post-larvae used for shrimp grow-out using a closed RAS and hybrid system.
Funding
This work was supported by the Ministry of Research, Technology, and Higher Education of the Republic of Indonesia, under the World Class Professor Program 2019.
Author information
Authors and Affiliations
Contributions
Both GS and MLS have contributed equally to the work. GS and MLS conceived and designed the study. FIS and SLEP performed experiments and analysis. FIS contributed to the manuscript writing process. SPP, PA and EF analysed the data and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
All procedures involving animals were performed in accordance with the guidelines for the care and use of animals at which the studies were conducted. This article does not involve human participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Suantika, G., Situmorang, M.L., Saputra, F.I. et al. Metabolite profiling of whiteleg shrimp Litopenaeus vannamei from super-intensive culture in closed aquaculture systems: a recirculating aquaculture system and a hybrid zero water discharge–recirculating aquaculture system. Metabolomics 16, 49 (2020). https://doi.org/10.1007/s11306-020-01675-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01675-1