Abstract
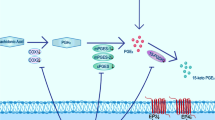
Lupeol has been reported to exhibit anti-inflammatory and anti-tumor activities in many diseases, but its potential effects in cerebral ischemia injury have not been studied to date. In this work we present evidence for a beneficial effect of lupeol in a rat model of middle cerebral artery occlusion (MCAO) followed by reperfusion (MCAO/R) injury and provide some histological and biochemical evidence for its mechanism of action. A cerebral MCAO rat model was established by vascular occlusion for 2 h, followed by 24 h reperfusion period. The infarct volume, neurological deficits, and brain water content were compared with animals treated during reperfusion with different concentrations of lupeol. Macroscopic parameters, cell viability, pro-inflammatory factors generation, as well as oxidative stress parameters and associated apoptotic signaling cascades were evaluated. Treatment with lupeol significantly reduced the cerebral infarct volume and water content and recovered neuro behavioral functions in affected rats. Lupeol treatment down-regulated the expression of oxidative stress and inflammation factors. In addition, lupeol activated Nrf2, suppressed caspase-3 activity, reduced BAX/Bcl-2 ratio and inhibited phosphorylation of p38 MAPK. The data suggest that lupeol may exert protective effects against cerebral ischemia by suppressing oxidative stress and reduction of inflammation factors possible via activation of nuclear transcription factors and inhibition of cell death pathways.






Similar content being viewed by others
References
Badshah H, Ali T, Rehman S-U, Amin F-U, Ullah F, Kim TH, Kim MO (2016) Protective effect of lupeol against lipopolysaccharide-induced neuroinflammation via the p38/c-Jun N-terminal kinase pathway in the adult mouse brain. J NeuroImmune Pharmacol 11:48–60
Cai Z et al (2003) Hearts from rodents exposed to intermittent hypoxia or erythropoietin are protected against ischemia-reperfusion injury. Circulation 108:79–85. https://doi.org/10.1161/01.CIR.0000078635.89229.8A
Cao C-X, Yang Q-W, Lv F-L, Cui J, Fu H-B, Wang J-Z (2007) Reduced cerebral ischemia-reperfusion injury in Toll-like receptor 4 deficient mice. Biochem Biophys Res Commun 353:509–514
Cheng C-Y, Su S-Y, Tang N-Y, Ho T-Y, Chiang S-Y, Hsieh C-L (2008) Ferulic acid provides neuroprotection against oxidative stress-related apoptosis after cerebral ischemia/reperfusion injury by inhibiting ICAM-1 mRNA expression in rats. Brain Res 1209:136–150
Fox C et al (2005) Minocycline confers early but transient protection in the immature brain following focal cerebral ischemia—reperfusion. J Cereb Blood Flow Metab 25:1138–1149
Garcia JH, Wagner S, Liu K-F, Hu X-J (1995) Neurological deficit and extent of neuronal necrosis attributable to middle cerebral artery occlusion in rats: statistical validation. Stroke 26:627–635
Geetha T, Varalakshmi P (2001) Anti-inflammatory activity of lupeol and lupeol linoleate in rats. J Ethnopharmacol 76:77–80
Guevara AP, Amor E, Russell G (1996) Antimutagens from Plumeria acuminata ait. Mutation Research/Environmental mutagenesis and related subjects 361:67–72
Guo M-B, Wang D-C, Liu H-F, Chen L-W, Wei J-W, Lin Y, Xue H (2018) Lupeol against high-glucose-induced apoptosis via enhancing the anti-oxidative stress in rabbit nucleus pulposus cells. Eur Spine J 27:2609–2620
Hashmi WJ, Ismail H, Mehmood F, Mirza B (2018) Neuroprotective, antidiabetic and antioxidant effect of Hedera nepalensis and lupeol against STZ+ AlCl 3 induced rats model. DARU journal of pharmaceutical sciences 26:179–190
He Y, Wan H, Du Y, Bie X, Zhao T, Fu W, Xing P (2012) Protective effect of Danhong injection on cerebral ischemia–reperfusion injury in rats. J Ethnopharmacol 144:387–394
Herlaar E, Brown Z (1999) p38 MAPK signalling cascades in inflammatory disease. Molecular medicine today 5:439–447
Ishii T, Itoh K, Takahashi S, Sato H, Yanagawa T, Katoh Y, Bannai S, Yamamoto M (2000) Transcription factor Nrf2 coordinately regulates a group of oxidative stress-inducible genes in macrophages. J Biol Chem 275:16023–16029
Jean WC, Spellman SR, Nussbaum ES, Low WC (1998) Reperfusion Injury after Focal Cerebral Ischemia: The Role Inflammation and the The rapeutic Horizon. Neurosurgery 43:1382–1396
Kohmoto J, Nakao A, Stolz DB, Kaizu T, Tsung A, Ikeda A, Shimizu H, Takahashi T, Tomiyama K, Sugimoto R, Choi AM, Billiar TR, Murase N, McCurry K (2007) Carbon monoxide protects rat lung transplants from ischemia-reperfusion injury via a mechanism involving p38 MAPK pathway. Am J Transplant 7:2279–2290
Kumari A, Kakkar P (2012) Lupeol protects against acetaminophen-induced oxidative stress and cell death in rat primary hepatocytes. Food Chem Toxicol 50:1781–1789
Leonard MO et al (2006) Reoxygenation-specific activation of the antioxidant transcription factor Nrf2 mediates cytoprotective gene expression in ischemia-reperfusion injury. FASEB J 20:2624–2626. https://doi.org/10.1096/fj.06-5097fje
Liu Y, Bi T, Shen G, Li Z, Wu G, Wang Z, Qian L, Gao Q (2016) Lupeol induces apoptosis and inhibits invasion in gallbladder carcinoma GBC-SD cells by suppression of EGFR/MMP-9 signaling pathway. Cytotechnology 68:123–133
Mdzinarishvili A, Kiewert C, Kumar V, Hillert M, Klein J (2007) Bilobalide prevents ischemia-induced edema formation in vitro and in vivo. Neuroscience 144:217–222
Nagaraj M, Sunitha S, Varalakshmi P (2000) Effect of lupeol, a pentacyclic triterpene, on the lipid peroxidation and antioxidant status in rat kidney after chronic cadmium exposure. Journal of applied toxicology: an international journal 20:413–417
Nelson CW, Wei EP, Povlishock JT, Kontos HA, Moskowitz MA (1992) Oxygen radicals in cerebral ischemia. Am J Phys Heart Circ Phys 263:H1356–H1362
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295
Pan J, Konstas A-A, Bateman B, Ortolano GA, Pile-Spellman J (2007) Reperfusion injury following cerebral ischemia: pathophysiology, MR imaging, and potential therapies. Neuroradiology 49:93–102
Prabhu B, Balakrishnan D, Sundaresan S (2016) Antiproliferative and anti-inflammatory properties of diindolylmethane and lupeol against N-butyl-N-(4-hydroxybutyl) nitrosamine induced bladder carcinogenesis in experimental rats. Hum Exp Toxicol 35:685–692
Prasad S, Kalra N, Singh M, Shukla Y (2008) Protective effects of lupeol and mango extract against androgen induced oxidative stress in Swiss albino mice. Asian journal of andrology 10:313–318
Prasad S, Madan E, Nigam N, Roy P, George J, Shukla Y (2009) Induction of apoptosis by lupeol in human epidermoid carcinoma A431 cells through regulation of mitochondrial, Akt/PKB and NF-kappaB signaling pathways. Cancer biology & therapy 8:1632–1639
Preetha S, Kanniappan M, Selvakumar E, Nagaraj M, Varalakshmi P (2006) Lupeol ameliorates aflatoxin B1-induced peroxidative hepatic damage in rats. Comparative biochemistry and physiology part C: Toxicology & Pharmacology 143:333–339
Ragasa CY, Javier ESC, Tan IG (2003) Antimutagenic terpenes and sterols from Vitex parviflora. Philipp J Sci 132:21–26
Saleem M (2009) Lupeol, a novel anti-inflammatory and anti-cancer dietary triterpene. Cancer Lett 285:109–115
Siripong P, Kanokmedakul K, Piyaviriyagul S, Yahuafai J, Chanpai R, Ruchirawat S, OKU N (2006) Antiproliferative naphthoquinone esters from Rhinacanthus nasutus Kurz. Roots on various cancer cells. journal of traditional medicines 23:166–172
Sudhahar V, Kumar SA, Varalakshmi P (2006) Role of lupeol and lupeol linoleate on lipemic–oxidative stress in experimental hypercholesterolemia Life. Sciences 78:1329–1335
Todd NV, Picozzi P, Crockard HA, Russell RR (1986) Reperfusion after cerebral ischemia: influence of duration of ischemia. Stroke 17:460–466
Traystman RJ, Kirsch JR, Koehler RC (1991) Oxygen radical mechanisms of brain injury following ischemia and reperfusion. J Appl Physiol 71:1185–1195
van Bruggen N et al (1999) VEGF antagonism reduces edema formation and tissue damage after ischemia/reperfusion injury in the mouse brain. J Clin Invest 104:1613–1620
Wahl F, Allix M, Plotkine M, Boulu R (1992) Neurological and behavioral outcomes of focal cerebral ischemia in rats. Stroke 23:267–272
Warach S, Latour LL (2004) Evidence of reperfusion injury, exacerbated by thrombolytic therapy, in human focal brain ischemia using a novel imaging marker of early blood–brain barrier disruption. Stroke 35:2659–2661
Wei Y, Gong J, Yoshida T, Eberhart CG, Xu Z, Kombairaju P, Sporn MB, Handa JT, Duh EJ (2011) Nrf2 has a protective role against neuronal and capillary degeneration in retinal ischemia–reperfusion injury. Free Radic Biol Med 51:216–224
Xiao YQ, Malcolm K, Worthen GS, Gardai S, Schiemann WP, Fadok VA, Bratton DL, Henson PM (2002) Cross-talk between ERK and p38 MAPK mediates selective suppression of pro-inflammatory cytokines by transforming growth factor-β. J Biol Chem 277:14884–14893
Xing B, Chen H, Zhang M, Zhao D, Jiang R, Liu X, Zhang S (2008) Ischemic postconditioning inhibits apoptosis after focal cerebral ischemia/reperfusion injury in the rat. Stroke 39:2362–2369
Yoon HY, Kang NI, Lee HK, Jang KY, Park JW, Park BH (2008) Sulforaphane protects kidneys against ischemia-reperfusion injury through induction of the Nrf2-dependent phase 2 enzyme. Biochem Pharmacol 75:2214–2223. https://doi.org/10.1016/j.bcp.2008.02.029
Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, Vinten-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285:H579–H588. https://doi.org/10.1152/ajpheart.01064.2002
Author information
Authors and Affiliations
Contributions
ZYZ, CFX, JHH, HYT and LYZ conceived and designed research. ZYZ, GFX, MZ, ZDW, TKY, KL, WDL and QLJ conducted experiments. ZCL, DW and ZQM contributed new reagents or analytical tools. ZYZ, CFX, JHH, HYT, TKY and KL analyzed data. ZYZ, CFX, JHH, HYT and LYZ wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Animal Ethics Committee of Liaocheng people’s hospital. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Informed consent
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Z., Xu, C., Hao, J. et al. Beneficial consequences of Lupeol on middle cerebral artery-induced cerebral ischemia in the rat involves Nrf2 and P38 MAPK modulation. Metab Brain Dis 35, 841–848 (2020). https://doi.org/10.1007/s11011-020-00565-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00565-8