Abstract
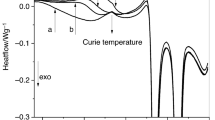
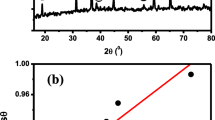
Molybdenum trioxide (MoO3) has been studied by four thermal analysis techniques. The as-synthesized material has hexagonal structure (h-MoO3). The phase change in the material studied using four thermal analysis techniques: thermogravimetric analysis, differential scanning calorimetry (DSC), thermomechanical analysis, and dynamic mechanical analysis. Crystallography phase transformation of h-MoO3 was found between 675 and 701 K by these techniques. DSC technique provided the highest detection sensitivity at about 690 K since the metastable monoclinic phase (β-MoO3) transformation could be observed before reaching the stable orthorhombic (α-MoO3) phase. The crystallographic phases of particles and morphologies were confirmed by X-ray diffraction and scanning electron microscopy techniques. The h-MoO3 was proposed as a standard sample to validate the operations of the thermal analysis instruments.






Similar content being viewed by others
References
Chiang TH, Yeh HC. The synthesis of α-MoO3 by ethylene glycol. Materials. 2013;6(10):4609–25.
Phuc NHH, et al. Simple method to prepare new structure of metastable molybdenum (VI) oxide. Mater Lett. 2012;76:173–6.
Li T, et al. Nanobelt-assembled nest-like MoO3 hierarchical structure: hydrothermal synthesis and gas-sensing properties. Mater Lett. 2015;160:476–9.
Jiao L, et al. A novel method for synthesis of microstructure MoO3. Mater Lett. 2005;59(24–25):3112–4.
Niederberger M, et al. Synthesis and characterization of novel nanoscopic molybdenum oxide fibers. J Mater Chem. 2001;11(7):1941–5.
Liu T, Xie Y, Chu B. Use of block copolymer micelles on the formation of hollow MoO3 nanospheres. Langmuir. 2000;16(23):9015–22.
Antonelli DM, Trudeau M. Phase changes and electronic properties in toroidal mesoporous molybdenum oxides. Angew Chem Int Ed. 1999;38(10):1471–5.
Sui L, et al. Construction of three-dimensional flower-like α-MoO3 with hierarchical structure for highly selective triethylamine sensor. Sens Actuators B Chem. 2015;208:406–14.
Hartl M, Daemen L, Lunk JH, Hartl H, Frisk AT, Shendervich I, Eckelt R. The extended family of hexagonal molybdenum oxide (No. LA-UR-09-01729; LA-UR-09-1729). Los Alamos National Lab. (LANL), Los Alamos, NM (United States); 2009.
Prakash NG, et al. High-performance one dimensional α-MoO3 nanorods for supercapacitor applications. Ceram Int. 2018;44:9967–75.
Zheng L, et al. Novel metastable hexagonal MoO3 nanobelts: synthesis, photochromic, and electrochromic properties. Chem Mater. 2009;21(23):5681–90.
Thangasmy P, Shanmugapriya V, Sathish M. The one-dimensional growth of hexagonal rods of metastable h-MoO3 using one-pot, rapid and environmentally benign supercritical fluid processing. Phys E. 2018;99:189–93.
Lunk H-J, et al. “Hexagonal molybdenum trioxide” known for 100 years and still a fount of new discoveries. Inorg Chem. 2010;49(20):9400–8.
Paraguay-Delgado F, et al. Optimization of the synthesis of α-MoO3 nanoribbons and hydrodesulfurization (HDS) catalyst test. J Nanosci Nanotechnol. 2007;7(10):3677–83.
Dhage SR, Hassan MS, Yang O-B. Low-temperature fabrication of hexagon shaped h-MoO3 nanorods and its phase transformation. Mater Chem Phys. 2009;114(2–3):511–4.
Irmawati R, Shafizah M. The production of high purity hexagonal MoO3 through the acid washing of as-prepared solids. Int J Basic Appl Sci. 2009;9:241–4.
Dastan D. Effect of preparation methods on the properties of titania nanoparticles: solvothermal versus sol–gel. Appl Phys A. 2017;123(11):699.
Dastan D, Chaure N, Kartha M. Surfactants assisted solvothermal derived titania nanoparticles: synthesis and simulation. J Mater Sci Mater Electron. 2017;28(11):7784–96.
Felder JB. Hydrothermal synthesis: a gateway to metastable crystals with usual properties. Ph.D. Thesis. University of South Carolina; 2018.
Wang S, et al. Shape tunable synthesis of perovskite structured rare-earth chromites RECrO3 via a mild hydrothermal method. Cryst Eng Commun. 2017;19:6436–42.
Chang KB, et al. Hydrothermal crystal growth, piezoelectricity and triboluminescence of KNaNbOF5. J Solid State Chem. 2016;236:78–82.
Santos-Beltran A, et al. Heat treatment effect of MoO3 on the MB removal and its reuse. J Phys Chem Solids. 2018;121:266–75.
Chithambararaj A, Rajeswari Yogamalar N, Bose AC. Hydrothermally synthesized h-MoO3 and α-MoO3 nanocrystals: new findings on crystal-structure-dependent charge transport. Cryst Growth Des. 2016;16(4):1984–95.
Hanafi ZM, Khilla MA, Askar MH. The thermal decomposition of ammonium heptamolybdate. Thermochim Acta. 1981;45(3):221–32.
Chithambararaj A, Bose AC. Hydrothermal synthesis of hexagonal and orthorhombic MoO3 nanoparticles. J Alloys Compd. 2011;509(31):8105–10.
Khalameida SV, et al. Chemical and phase transformation in the V2O5–(NH4)2 Mo2O7 system during the mechanochemical treatment in various media. J Therm Anal Calorim. 2010;101(3):823–32.
Khalameida S, et al. Physical–chemical transformations in the system V2O5/(NH4)2Mo2O7 under hydrothermal conditions. Open Chem. 2014;12(2):140–52.
Ramana CV, et al. Low-temperature synthesis of morphology-controlled metastable hexagonal molybdenum trioxide (MoO3). Solid State Commun. 2009;149(1–2):6–9.
Wu Z, et al. Ultrasonic-assisted preparation of metastable hexagonal MoO3 nanorods and their transformation to microbelts. Ultrason Sonochem. 2011;18(1):288–92.
Song Y, et al. Aqueous synthesis of molybdenum trioxide (h-MoO3, α-MoO3·H2O, and h-/α-MoO3 composites) and their photochromic properties study. J Alloys Compd. 2017;693:1290–6.
Mizushima T, et al. Soft chemical transformation of α-MoO3 to β-MoO3 as a catalyst for vapor-phase oxidation of methanol. Catal Commun. 2011;13(1):10–3.
Pham TTP, et al. Facile method for synthesis of nanosized β-MoO3 and their catalytic behavior for selective oxidation of methanol to formaldehyde. Adv Nat Sci Nanosci Nanotechnol. 2015;6(4):045010.
Peng Z, Redfern SAT. Mechanical properties of quartz at the α-β phase transition: implications for tectonic and seismic anomalies. Geochem Geophys Geosyst. 2013;14(1):18–28.
Dong Y, et al. Studies on glass transition temperature of chitosan with four techniques. J Appl Polym Sci. 2004;93(4):1553–8.
Acknowledgements
The authors wish to thank and acknowledge L. de La Torre for his technical analysis. Thanks to W. Antunez, K. Campos, I. Estrada G. and E. Lestarjet for their help by SEM and XRD data acquisition, and Nanotech Lab, CIMAV, Chihuahua, Mexico.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paraguay-Delgado, F., Mendoza Duarte, M.E., Kalu, O. et al. h-MoO3 phase transformation by four thermal analysis techniques. J Therm Anal Calorim 140, 735–741 (2020). https://doi.org/10.1007/s10973-019-08842-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-019-08842-0