Abstract
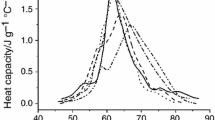
In this work, we examined by DSC protein denaturation heat capacity profiles for two body fluids, cerebrospinal fluid (CSF) and blood plasma obtained from brain tumor (mainly glioblastoma) patients and healthy volunteers. We observed large distinctions between the heat capacity profiles of CSF and blood plasma, although their protein compositions are believed to have much in common. A prominent, previously unreported CSF feature was the existence of a pre-denaturation exothermic transition peaking at ~ 50–52 °C, recorded for both control and brain tumor CSF. This appears to be the first observation of a pre-denaturation exotherm in a human body fluid. In all studied samples, the exotherms deconvoluted with high precision into a sum of two Gaussian peaks. These exotherms are apparently specific, originating from brain tissue-soluble proteins in the CSF not present in blood plasma. Malignant brain tumors (glioblastoma multiforme, Grade IV, and low-grade glioma, Grade II) reduced twofold the enthalpy of the exotherms relative to the control. These results suggest that the amount and/or conformational state of the CSF proteins (e.g., intrinsic disorder) giving rise to pre-denaturation exothermic events substantially changed upon brain tumor progression. Concomitantly, the enthalpy of the CSF endothermic peaks was partially redistributed from a lower-temperature (main) transition to a higher-temperature transition. The presented data demonstrated that the heat capacity profiles of intrinsic CSF proteins constitute a sensitive biomarker of glioblastoma and other brain malignancies.




Similar content being viewed by others
References
Abarova S, Koynova R, Tancheva L, Tenchov B (2017) A novel DSC approach for evaluating protectant drugs efficacy against dementia. Biochimica Biophysica Acta (BBA) 1863:2934–2941. https://doi.org/10.1016/j.bbadis.2017.07.033
Banks RE, Stanley AJ, Cairns DA, Barrett JH, Clarke P, Thompson D, Selby PJ (2005) Influences of blood sample processing on low-molecular-weight proteome identified by surface-enhanced laser desorption/ionization mass spectrometry. Clin Chem 51:1637–1649. https://doi.org/10.1373/clinchem.2005.051417
Brudar S, Cernigoj U, Podgornik H, Krzan M, Prislan I (2017) Use of differential scanning calorimetry and immunoaffinity chromatography to identify disease induced changes in human blood plasma proteome. Acta Chim Slov 64:564–570. https://doi.org/10.17344/acsi.2016.2970
Chagovetz AA, Jensen RL, Recht L, Glantz M, Chagovetz AM (2011) Preliminary use of differential scanning calorimetry of cerebrospinal fluid for the diagnosis of glioblastoma multiforme. J Neurooncol 105:499–506. https://doi.org/10.1007/s11060-011-0630-5
Ferencz A, Zapf I, Lorinczy D (2016) Harmful effect of neoadjuvant chemotherapy monitoring by DSC on breast cancer patients' blood plasma. J Therm Anal Calorim 126:55–59. https://doi.org/10.1007/s10973-016-5291-3
Garbett NC, Miller JJ, Jenson AB, Chaires JB (2008) Calorimetry outside the box: a new window into the plasma proteome. Biophys J 94:1377–1383. https://doi.org/10.1529/biophysj.107.119453
Garbett NC, Mekmaysy CS, Helm CW, Jenson AB, Chaires JB (2009) Differential scanning calorimetry of blood plasma for clinical diagnosis and monitoring. Exp Mol Pathol 86:186–191. https://doi.org/10.1016/j.yexmp.2008.12.001
Garbett NC, Merchant ML, Helm CW, Jenson AB, Klein JB, Chaires JB (2014) Detection of cervical cancer biomarker patterns in blood plasma and urine by differential scanning calorimetry and mass spectrometry. PLoS ONE. https://doi.org/10.1371/journal.pone.0084710
Huhmer AF, Biringer RG, Amato H, Fonteh AN, Harrington MG (2006) Protein analysis in human cerebrospinal fluid: physiological aspects, future challenges. Dis Markers 22:3–26
Iakoucheva LM, Brown CJ, Lawson JD, Obradovic Z, Dunker AK (2002) Intrinsic disorder in cell-signaling and cancer-associated proteins. J Mol Biol 323:573–584. https://doi.org/10.1016/s0022-2836(02)00969-5
Jakob U, Kriwacki R, Uversky VN (2014) Conditionally and transiently disordered proteins: awakening cryptic disorder to regulate protein function. Chem Rev 114:6779–6805. https://doi.org/10.1021/cr400459c
Khalil AA (2007) Biomarker discovery: a proteomic approach for brain cancer profiling. Cancer Sci 98:201–213. https://doi.org/10.1111/j.1349-7006.2007.00374.x
Kielbon A, Michnik A, Grelik KP, Duch K, Sadowska-Krepa E (2019) Differential scanning calorimetry of human blood serum exposed in vitro to X-ray radiation. Thermochim Acta. https://doi.org/10.1016/j.tca.2019.178358
Koynova R, Antonova B, Sezanova B, Tenchov B (2018) Beneficial effect of sequential chemotherapy treatments of lung cancer patients revealed by calorimetric monitoring of blood plasma proteome denaturation. Thermochim Acta 659:1–7. https://doi.org/10.1016/j.tca.2017.11.001
Lohner K, Esser AF (1991) Thermal unfolding and aggregation of human-complement protein-C9: a differential scanning calorimetry study. Biochemistry 30:6620–6625
Lorinczy D (2005) The nature of biological systems as revealed by thermal methods. Hot topics in thermal analysis and calorimetry. Springer, Netherlands
Meng QM et al (2014) Bex2 controls proliferation of human glioblastoma cells through nf-kappa b signaling pathway. J Mol Neurosci 53:262–270. https://doi.org/10.1007/s12031-013-0215-1
Michnik A, Drzazga Z (2010) Thermal denaturation of mixtures of human serum proteins. J Therm Anal Calorim 101:513–518. https://doi.org/10.1007/s10973-010-0826-5
Michnik A, Drzazga Z, Michalik K, Barczyk A, Santura I, Sozanska E, Pierzchala W (2010) Differential scanning calorimetry study of blood serum in chronic obstructive pulmonary disease. J Therm Anal Calorim 102:57–60. https://doi.org/10.1007/s10973-009-0602-6
Michnik A, Polaczek-Grelik K, Stas M, Sadowska-Krepa E, Gibinska J, Drzazga Z (2016) Delayed effects of neutron radiation on human serum. J Therm Anal Calorim 126:37–45. https://doi.org/10.1007/s10973-016-5255-7
Ostrom QT et al (2014) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol 16:1–63. https://doi.org/10.1093/neuonc/nou223
Privalov PL, Khechinashvili NN (1974) Thermodynamic approach to problem of stabilization of globular protein structure: calorimetric study. J Mol Biol 86:665–684. https://doi.org/10.1016/0022-2836(74)90188-0
Prochownik EV (2004) c-Myc as a therapeutic target in cancer. Expert Rev Anticancer Ther 4:289–302. https://doi.org/10.1586/14737140.4.2.289
Rao JS (2003) Molecular mechanisms of glioma invasiveness: the role of proteases. Nat Rev Cancer 3:489–501. https://doi.org/10.1038/nrc1121
Roche S, Gabelle A, Lehmann S (2008) Clinical proteomics of the cerebrospinal fluid: towards the discovery off now biomarkers. Proteom Clin Appl 2:428–436. https://doi.org/10.1002/prca.200780040
Sasahara K, Yagi H, Naiki H, Goto Y (2009) Thermal response with exothermic effects of beta(2)-microglobulin amyloid fibrils and fibrillation. J Mol Biol 389:584–594. https://doi.org/10.1016/j.jmb.2009.04.026
Schain RJ (1964) Cerebrospinal fluid and serum cation levels. Arch Neurol 11:330–333. https://doi.org/10.1001/archneur.1964.00460210108012
Schutzer SE et al (2010) Establishing the proteome of normal human cerebrospinal fluid. PLoS ONE. https://doi.org/10.1371/journal.pone.0010980
Szalai Z, Molnar TF, Lorinczy D (2017) Role of differential scanning calorimetry (DSC) in the staging of COPD. J Therm Anal Calorim 127:1231–1238. https://doi.org/10.1007/s10973-016-5495-6
Takahashi K, Sturtevant JM (1981) Thermal-denaturation of Streptomyces subtilisin inhibitor, subtilisin bpn', and the inhibitor-subtilisin complex. Biochemistry 20:6185–6190. https://doi.org/10.1021/bi00524a042
Tenchov B, Abarova S, Koynova R, Traikov L, Dragomanova S, Tancheva L (2017a) A new approach for investigating neurodegenerative disorders in mice based on DSC. J Therm Anal Calorim 127:483–486. https://doi.org/10.1007/s10973-016-5749-3
Tenchov B, Abarova S, Koynova R, Traikov L, Tancheva L (2017b) Low-temperature exothermic transitions in brain proteome of mice, effect of scopolamine. Thermochim Acta. https://doi.org/10.1016/j.tca.2017.01.012
Todinova S, Krumova S, Kurtev P, Dimitrov V, Djongov L, Dudunkov Z, Taneva SG (2012) Calorimetry-based profiling of blood plasma from colorectal cancer patients. Biochimica Biophys Acta 1820:1879–1885. https://doi.org/10.1016/j.bbagen.2012.08.001
Todinova S, Krumova S, Radoeva R, Gartcheva L, Taneva SG (2014) Calorimetric markers of bence jones and nonsecretory multiple myeloma serum proteome. Anal Chem 86:12355–12361. https://doi.org/10.1021/ac503677d
Tompa P, Szasz C, Buday L (2005) Structural disorder throws new light on moonlighting. Trends Biochem Sci 30:484–489. https://doi.org/10.1016/j.tibs.2005.07.008
Tsvetkov PO et al (2018) Differential scanning calorimetry of plasma in glioblastoma: toward a new prognostic / monitoring tool. Oncotarget 9:9391–9399. https://doi.org/10.18632/oncotarget.24317
Uversky VN (2009) Intrinsically disordered proteins and their environment: effects of strong denaturants, temperature, pH, counter ions, membranes, binding partners, osmolytes, and macromolecular crowding. Protein J 28:305–325. https://doi.org/10.1007/s10930-009-9201-4
Uversky VN (2015) Intrinsically disordered proteins and their (disordered) proteomes in neurodegenerative disorders. Front Aging Neurosci. https://doi.org/10.3389/fnagi.2015.00018
Uversky VN, Oldfield CJ, Dunker AK (2008) Intrinsically disordered proteins in human diseases: introducing the D(2) concept. Ann Rev Biophys 37:215–246. https://doi.org/10.1146/annurev.biophys.37.032807.125924
Zapf I, Moezzi M, Fekecs T, Nedvig K, Lorinczy D, Ferencz A (2016) Influence of oxidative injury and monitoring of blood plasma by DSC on breast cancer patients. J Therm Anal Calorim 123:2029–2035. https://doi.org/10.1007/s10973-015-4642-9
Zougman A, Pilch B, Podtelejnikov A, Kiehntopf M, Schnabel C, Kurnar C, Mann M (2008) Integrated analysis of the cerebrospinal fluid peptidome and proteome. J Proteom Res 7:386–399. https://doi.org/10.1021/pr070501k
Acknowledgements
We thank Dr. Dimiter Monov for help with the control CSF isolation and the staff of the Central Clinical Laboratory of “St. Ivan Rilski” University Hospital for the protein content determinations. This work was supported by the Bulgarian National Science Research Fund (grant DN03/13/2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Research Ethics Committee of Medical University-Sofia (protocol reference No. 24/19.11.2015) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Antonova, B., Naydenov, E., Koynova, R. et al. Exothermic transitions in the heat capacity profiles of human cerebrospinal fluid. Eur Biophys J 49, 231–238 (2020). https://doi.org/10.1007/s00249-020-01429-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-020-01429-w