Abstract
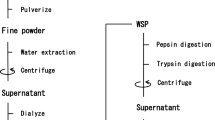
Inhibition of angiotensin I–converting enzyme (ACE) is one of the key factors to repress high blood pressure. Although many studies have been reported that seaweed protein hydrolysates showed the ACE inhibitory activity, the comprehensive understanding of the relationship was still unclear. In this study, we employed chloroplast genome for in silico analysis and compared it with in vitro experiments. We first extracted water-soluble proteins (WSP) from red alga Grateloupia asiatica, which contained mainly PE, PC, APC, and Rbc, and prepared WSP hydrolysate by thermolysin, resulting that the hydrolysate showed ACE inhibitory activity. Then, we determined the complete chloroplast genome of G. asiatica (187,518 bp: 206 protein-coding genes, 29 tRNA, and 3 rRNA) and clarified the amino acid sequences of main WSP, i.e., phycobiliproteins and Rubisco, to perform in silico analysis. Consequently, 190 potential ACE inhibitory peptides existed in the main WSP sequences, and 21 peptides were obtained by in silico thermolysin digestion. By comparing in vitro and in silico analyses, in vitro ACE inhibitory activity was correlated to the IC50 value from in silico digestion. Therefore, in silico approach provides insight into the comprehensive understanding of the potential bioactive peptides from seaweed proteins.


Similar content being viewed by others
Abbreviations
- ACE:
-
Angiotensin I–converting enzyme
- APC:
-
Allophycocyanin
- CBB:
-
Coomassie brilliant blue
- PC:
-
Phycocyanin
- PCG:
-
Protein-coding genes
- PE:
-
Phycoerythrin
- Rbc:
-
Ribulose-1, 5-bisphosphate carboxylase/oxygenase
- WSP:
-
Water-soluble proteins
References
Beaulieu L, Sirois M, Tamigneaux É (2016) Evaluation of the in vitro biological activity of protein hydrolysates of the edible red alga, Palmaria palmata (dulse) harvested from the Gaspe coast and cultivated in tanks. J Appl Phycol 28:3101–3115
Bennett A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58:419–435
Cao D, Lv X, Xu X, Yu H, Sun X, Xu N (2017) Purification and identification of a novel ACE inhibitory peptide from marine alga Gracilariopsis lemaneiformis protein hydrolysate. Eur Food Res Technol 243:1829–1837
Chen H, Yan X, Zhu P, Lin J (2006) Antioxidant activity and hepatoprotective potential of agaro-oligosaccharides in vitro and in vivo. Nutr J 5:31
Cheung HS, Cushman DW (1973) Inhibition of homogeneous angiotensin-converting enzyme of rabbit lung by synthetic venom peptides of Bothrops jararaca. Biochim Biophys Acta Enzymol 293:451–463
Cian RE, Alaiz M, Vioque J, Drago SR (2013) Enzyme proteolysis enhanced extraction of ACE inhibitory and antioxidant compounds (peptides and polyphenols) from Porphyra columbina residual cake. J Appl Phycol 25:1197–1206
Cian RE, Garzón AG, Ancona DB, Guerrero LC, Drago SR (2015) Hydrolyzates from Pyropia columbina seaweed have antiplatelet aggregation, antioxidant and ACE I inhibitory peptides which maintain bioactivity after simulated gastrointestinal digestion. LWT Food Sci Technol 64:881–888
Costa JF, Lin SM, Macaya EC, Fernandez-Garcia C, Verbruggen H (2016) Chloroplast genomes as a tool to resolve red algal phylogenies: a case study in the Nemaliales. BMC Evol Biol 16:1–13
Cota-Sánchez JH, Remarchuk K, Ubayasena K (2006) Ready-to-use DNA extracted with a CTAB method adapted for herbarium specimens and mucilaginous plant tissue. Plant Mol Biol Report 24:161–167
Daskaya-Dikmen C, Yucetepe A, Karbancioglu-Guler F, Daskaya H, Ozcelik B (2017) Angiotensin-I-converting enzyme (ACE)-inhibitory peptides from plants. Nutrients 9:316
De Ruiter GA, Rudolph B (1997) Carrageenan biotechnology. Trends Food Sci Technol 8:389–395
DePriest MS, Bhattacharya D, López-Bautista JM (2013) The plastid genome of the red macroalga Grateloupia taiwanensis (Halymeniaceae). PLoS One 8:e68246
Du Q, Bi G, Mao Y, Sui Z (2016) The complete chloroplast genome of Gracilariopsis lemaneiformis (Rhodophyta) gives new insight into the evolution of family Gracilariaceae. J Phycol 52:441–450
Fitzgerald C, Gallagher E, Tasdemir D, Hayes M (2011) Heart health peptides from macroalgae and their potential use in functional foods. J Agric Food Chem 59:6829–6836
Fitzgerald C, Aluko RE, Hossain M, Rai DK, Hayes M (2014) Potential of a renin inhibitory peptide from the red seaweed Palmaria palmata as a functional food ingredient following confirmation and characterization of a hypotensive effect in spontaneously hypertensive rats. J Agric Food Chem 62:8352–8356
Furuta T, Miyabe Y, Yasui H, Kinoshita Y, Kishimura H (2016) Angiotensin I converting enzyme inhibitory peptides derived from phycobiliproteins of dulse Palmaria palmata. Mar Drugs 14:32
Glazer AN (1985) Light harvesting by phycobilisomes. Ann Rev Biophys Chem 14:47–77
Harnedy PA, O’Keeffe MB, FitzGerald RJ (2015) Purification and identification of dipeptidyl peptidase (DPP) IV inhibitory peptides from the macroalga Palmaria palmata. Food Chem 172:400–406
He HL, Chen XL, Wu H, Sun CY, Zhang YZ, Zhou BC (2007) High throughput and rapid screening of marine protein hydrolysates enriched in peptides with angiotensin-I-converting enzyme inhibitory activity by capillary electrophoresis. Bioresour Technol 98:3499–3505
Holdt SL, Kraan S (2011) Bioactive compounds in seaweed: functional food applications and legislation. J Appl Phycol 23:543–597
Kitade Y, Miyabe Y, Yamamoto Y, Takeda H, Shimizu T, Yasui H, Kishimura H (2018) Structural characteristics of phycobiliproteins from red alga Mazzaella japonica. J Food Biochem 42:e12436
Kumagai Y, Miyabe Y, Takeda T, Adachi K, Yasui H, Kishimura H (2019a) In silico analysis of relationship between proteins from plastid genome of red alga Palmaria sp. (Japan) and angiotensin I converting enzyme inhibitory peptides. Mar Drugs 17:190
Kumagai Y, Tsubouchi R, Miyabe Y, Takeda T, Adachi K, Yasui H, Kishimura H (2019b) Complete sequence of mitochondrial DNA of red alga dulse Palmaria palmata (Linnaeus) Weber & Mohr in Japan. Mitochondrial DNA Part B 4:3177–3178
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lee SY, Hur SJ (2017) Antihypertensive peptides from animal products, marine organisms, and plants. Food Chem 228:506–517
Lee KY, Mooney DJ (2012) Alginate: properties and biomedical applications. Prog Polym Sci 37:106–126
Mai K, Mercer JP, Donlon J (1994) Comparative studies on the nutrition of two species of abalone, Haliotis tuberculata L and Haliotis discus hannai Ino: II Amino acid composition of abalone and six species of macroalgae with an assessment of their nutritional value. Aquaculture 128:115–130
Miyabe Y, Furuta T, Takeda T, Kanno G, Shimizu T, Tanaka Y, Gai Z, Yasui H, Kishimura H (2017) Structural properties of phycoerythrin from dulse Palmaria palmata. J Food Biochem 41:e12301
Ng PK, Lin SM, Lim PE, Liu LC, Chen CM, Pai TW (2017) Complete chloroplast genome of Gracilaria firma (Gracilariaceae, Rhodophyta), with discussion on the use of chloroplast phylogenomics in the subclass Rhodymeniophycidae. BCM Genomics 18:40
Sato N, Furuta T, Takeda T, Miyabe Y, Ura K, Takagi Y, Yasui H, Kumagai Y, Kishimura H (2018) Antioxidant activity of proteins extracted from red alga dulse harvested in Japan. J Food Biochem 43:e12709
Schmieder RE, Hilgers KF, Schlaich MP, Schmidt BMW (2007) Renin-angiotensin system and cardiovascular risk. Lancet 369:1208–1219
Sfriso AA, Gallo M, Baldi F (2018) Phycoerythrin productivity and diversity from five red macroalgae. J Appl Phycol 30:2523–2531
Sonani RR, Singh NK, Kumar J, Thakar D, Madamwar D (2014) Concurrent purification and antioxidant activity of phycobiliproteins from Lyngbya sp. A09DM: an antioxidant and anti-aging potential of phycoerythrin in Caenorhabditis elegans. Process Biochem 49:1757–1766
Suetsuna K (1998) Purification and identification of angiotensin I-converting enzyme inhibitors from the red alga Porphyra yezoensis. J Mar Biotechnol 6:163–167
Suetsuna K, Nakano T (2000) Identification of an antihypertensive peptide from peptic digest of wakame (Undaria pinnatifida). J Nutr Biochem 11:450–454
Watanabe K, Kishimoto T, Kumagai Y, Shimizu T, Uji T, Yasui H, Kishimura H (2019) Complete sequence of mitochondrial DNA of Gloiopeltis furcata (Postels and Ruprecht) J. Agardh. Mitochondrial DNA Part B 4:2543–2544
Yamamoto Y, Kishimura H, Kinoshita Y, Saburi W, Kumagai Y, Yasui H, Ojima T (2019) Enzymatic production of xylooligosaccharides from red alga dulse (Palmaria sp.) wasted in Japan. Process Biochem 82:117–122
Yang EC, Kim KM, Kim SY, Lee JM, Boo GH, Lee JH, Nelson WA, Yi G, Schmidt WE, Fredericq S, Boo SM, Bhattacharya D, Yoon HS (2015) Highly conserved mitochondrial genomes among multicellular red algae of the Florideophyceae. Genome Biol Evol 7:2394–2406
Yang EC, Boo SM, Bhattacharya D, Saunders GW, Knoll AH, Fredericq S, Yoon HS (2016) Divergence time estimates and the evolution of major lineages in the florideophyte red algae. Sci Rep 6:21361
Zhang J, Tang X, Chen WZ, Liu T, Li Y (2018) The complete plastid genome of Grateloupia filicina (Rhodophyta) and phylogenetic analysis. Mitochondrial DNA B 3:1172–1173
Funding
This study was partially supported by the “Regional Innovation Cluster Program (Global Type), Ministry of Education, Culture, Sports, Science and Technology, Japan” and the “Science and technology research promotion program for agriculture, forestry, fisheries and food industry (27004B).”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(XLSX 9 kb)
Rights and permissions
About this article
Cite this article
Sumikawa, K., Takei, K., Kumagai, Y. et al. In Silico Analysis of ACE Inhibitory Peptides from Chloroplast Proteins of Red Alga Grateloupia asiatica. Mar Biotechnol 22, 391–402 (2020). https://doi.org/10.1007/s10126-020-09959-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-020-09959-2