Abstract
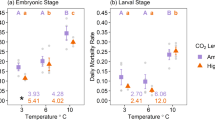
Elevated temperatures and nutrients are degrading coral reef ecosystems, but the understanding of how early life stages of reef corals respond to these stressors remains limited. Here, we test the impact of temperature (mean ~ 27 °C vs. ~ 29 °C) and nitrate and phosphate enrichment (ambient, + 5 µM nitrate, + 1 µM phosphate and combined + 5 µM nitrate with 1 µM phosphate) on coral larvae using three Hawaiian coral species with different modes of symbiont transmission and reproduction: Lobactis scutaria (horizontal, gonochoric broadcast spawner), Pocillopora acuta (vertical, hermaphroditic brooder) and Montipora capitata (vertical, hermaphroditic broadcast spawner). Temperature and nutrient effects were species specific and appear antagonistic for L. scutaria and M. capitata, but not for P. acuta. Larvae survivorship in all species was lowest under nitrate enrichment at 27 °C. M. capitata and L. scutaria survivorship increased at 29 °C. However, positive effects of warming on survivorship were lost under high nitrate, but phosphate attenuated nitrate effects when N/P ratios were balanced. P. acuta larvae exhibited high survivorship (> 91%) in all treatments and showed little change in larval size, but lower respiration rates at 29 °C. Elevated nutrients (+N+P) led to the greatest loss in larvae size for aposymbiotic L. scutaria, while positive growth in symbiotic M. capitata larvae was reduced under warming and highest in +N+P treatments. Overall, we report a greater sensitivity of broadcast spawners to warming and nutrient changes compared to a brooding coral species. These results suggest variability in biological responses to warming and nutrient enrichment is influenced by life-history traits, including the presence of symbionts (vertical transmission), in addition to nutrient type and nutrient stoichiometry.





Similar content being viewed by others
References
Baird AH, Gilmour JP, Kamiki TM, Nonaka M, Pratchett MS, Yamamoto HH, Yamasaki H (2006) Temperature tolerance of symbiotic and non-symbiotic coral larvae. In: Proceedings of the 10th international coral reef symposium, pp 38–42
Baird AH, Guest JR, Willis BL (2009) Systematic and biogeographical patterns in the reproductive biology of Scleractinian corals. Annu Rev Ecol Evol Syst 40(1):551–571
Bahr KD, Jokiel PL, Toonen RJ (2015) The unnatural history of Kāne‘ohe Bay: coral reef resilience in the face of centuries of anthropogenic impacts. PeerJ 3:e950
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: an ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80(4):435–471
Bassim K, Sammarco P (2003) Effects of temperature and ammonium on larval development and survivorship in a scleractinian coral (Diploria strigosa). Mar Biol 142(24):241–252
Coles SL, Bahr KD, Ku’ulei SR, May SL, McGowan AE, Tsang A, Bumgarner J, Han JH (2018) Evidence of acclimatization or adaptation in Hawaiian corals to higher ocean temperatures. PeerJ 6:e5347
Coles S, Jokiel PL (1977) Effects of temperature on photosynthesis and respiration in hermatypic corals. Mar Biol 43:209–216
Courtial L, Bielsa VP, Houlbrèque F, Farrier-Pagès C (2018) Effects of ultraviolet radiation and nutrient level on the physiological response and organic matter release of the scleractinian coral Pocillopora damicornis following thermal stress. PLoS ONE 13(10):e0205261
Cox EF, Ward S (2002) Impact of elevated ammonium on reproduction in two Hawaiian scleractinian corals with different life history patterns. Mar Pollut Bull 44:1230–1235
Cumbo VR, Fan TY, Edmunds PJ (2013) Effects of exposure duration on the response of Pocillopora damicornis larvae to elevated temperature and high pCO2. J Exp Mar Bio Ecol 439:100–107
Cunning R, Baker AC (2013) Excess algal symbionts increase the susceptibility of reef corals to bleaching. Nat Clim Change 3:259–262
D’Angelo C, Wiedenmann J (2014) Impacts of nutrient enrichment on coral reefs: new perspectives and implications for coastal management and reef survival. Curr Opin Environ Sustain 7:82–93
Drupp P, De Carlo EH, Mackenzie FT, Bienfang P, Sabine CL (2011) Nutrient inputs, phytoplankton response, and CO2 variations in a semi-enclosed subtropical embayment, Kāne‘ohe Bay, Hawai‘i. Aquat Geochem 17:473–498
Edmunds P, Gates R, Gleason D (2001) The biology of larvae from the reef coral Porites astreoides, and their response to temperature disturbances. Mar Biol 139(5):981–989
Edmunds PJ, Gates RD, Leggat W, Hoegh-Guldberg O, Allen-Requa L (2005) The effect of temperature on the size and population density of dinoflagellates in larvae of the reef coral Porites astreoides. Invertebr Biol 124(3):185–193
Edmunds PJ, Cumbo V, Fan TY (2011) Effects of temperature on the respiration of brooded larvae from tropical reef corals. J Exp Biol 214:2783–2790
Ezzat L, Maguer JF, Grover R, Ferrier-Pagès C (2015) New insights into carbon acquisition and exchanges within the coral–dinoflagellate symbiosis under NH4+ and NO3− supply. Proc Biol Sci 282(1812):20150610
Ezzat L, Maguer JF, Grover R, Ferrier-Pagès C (2016) Limited phosphorus availability is the Achilles heel of tropical reef corals in a warming ocean. Sci Rep 6(1):31768
Fabricius KE (2005) Effects of terrestrial runoff on the ecology of corals and coral reefs: review and synthesis. Mar Pollut Bull 50(2):125–146
Falkowski PG, Dubinsky Z, Muscatine L, McCloskey L (1993) Population control in symbiotic corals. Bioscience 43(9):606–611
Figueiredo J, Baird AH, Cohen MF, Flot JF, Kamiki T, Meziane T, Tsuchiya M, Yamasaki H (2012) Ontogenetic change in the lipid and fatty acid composition of scleractinian coral larvae. Coral Reefs 31:613–619
Figueiredo J, Baird AH, Harii S, Connolly SR (2014) Increased local retention of reef coral larvae as a result of ocean warming. Nat Clim Change 4:498–502
Fisch J, Drury C, Towle EK, Winter RN, Miller MW (2019) Physiological and reproductive repercussions of consecutive summer bleaching events of the threatened Caribbean coral Orbicella faveolata. Coral Reefs 38:863–876
Gittenberger A, Reijnen BT, Hoeksema BW (2011) A molecularly based phylogeny reconstruction of mushroom corals (Scleractinia: Fungiidae) with taxonomic consequences and evolutionary implications for life history traits. Contrib Zool 80(2):107–132
Graham NAJ, Jennings S, Macneil MA, Mouillot D, Wilson SK (2015) Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 518:94–97
Graham EM, Baird AH, Connolly SR, Sewell MA, Willis BL (2017) Uncoupling temperature-dependent mortality from lipid depletion for scleractinian coral larvae. Coral Reefs 36(1):97–104
Harii S, Nadaoka K, Yamamoto M, Iwao K (2007) Temporal changes in settlement, lipid content and lipid composition of larvae of the spawning hermatypic coral Acropora tenuis. Mar Ecol Prog Ser 346:89–96
Harii S, Yamamoto M, Hoegh-Guldberg O (2010) The relative contribution of dinoflagellate photosynthesis and stored lipids to the survivorship of symbiotic larvae of the reef-building corals. Mar Biol 157:1215–1224
Harrison P, Ward S (2001) Elevated levels of nitrogen and phosphorus reduce fertilisation success of gametes from scleractinian reef corals. Mar Biol 139(6):1057–1068
Haryanti D, Hidaka M (2015) Temperature dependence of respiration in larvae and adult colonies of the corals Acropora tenuis and Pocillopora damicornis. J Mar Sci Eng 3(3):509–519
Haryanti D, Yasuda N, Harii S, Hidaka M (2015) High tolerance of symbiotic larvae of Pocillopora damicornis to thermal stress. Zool Stud 54:52
Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford University Press, New York
Hoyos CD, Agudelo PA, Webster PJ, Curry JA (2006) Deconvolution of the factors contributing to the increase in global hurricane intensity. Science 312(5770):94–97
Hughes TP, Kerry JT, Alvarez-Noriega M, Alvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TC (2017) Global warming and recurrent mass bleaching of corals. Nature 543(7645):373
Humanes A, Ricardo GF, Willis BL, Fabricius KE, Negri AP (2017) Cumulative effects of suspended sediments, organic nutrients and temperature stress on early life history stages of the coral Acropora tenuis. Sci Rep 7(1):44101
Humphrey C, Weber M, Lott C, Cooper T, Fabricius K (2008) Effects of suspended sediments, dissolved inorganic nutrients and salinity on fertilisation and embryo development in the coral Acropora millepora (Ehrenberg, 1834). Coral Reefs 27(4):837–850
Kassambara A, Kosinski M and Biecek P (2019) Survminer: drawing survival curves using ‘ggplot2’. R package version 0.4.6. https://CRAN.R-project.org/package=survminer
LaJeunesse TC, Parkinson JE, Gabrielson PW, Jeong HJ, Reimer JD, Voolstra CR, Santos SR (2018) Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Curr Biol 28(16):2570–2580
Lam EK, Ap Chui, Kwok CK, Ip AHA, Chn SW, Leung HN, Yeung LC, Ang PO (2015) High levels of inorganic nutrients affect fertilization kinetics, early development and settlement of the scleractinian coral Platygyra acuta. Coral Reefs 34(3):837–848
Lenth R (2019) Emmeans: estimated marginal means, aka least-squares means. R package version 1.3.5. https://CRAN.R-project.org/package=emmeans
Mayfield AB, Chan PH, Putnam HM, Chen CS, Fan TY (2012) The effects of a variable temperature regime on the physiology of the reef-building coral Seriatopora hystrix: results from a laboratory-based reciprocal transplant. J Exp Biol 215:4183–4195
Nakamura M, Ohki S, Suzuki A, Sakai K (2011) Coral larvae under ocean acidification: survival, metabolism, and metamorphosis. PLoS ONE 6(1):e14521
NOAA (2019) National Data Buoy Center, Station MOKH1-1612480. Mokuoloe, HI
Nozawa Y, Okubo N (2011) Survival dynamics of reef coral larvae with special consideration of larval size and the genus Acropora. Biol Bull 220:15–22
Olsen K, Ritson-Williams R, Ochrietor JD, Paul VJ, Ross C (2013) Detecting hyperthermal stress in larvae of the hermatypic coral Porites astreoides: the suitability of using biomarkers of oxidative stress versus heat-shock protein transcriptional expression. Mar Biol 160:2609–2618
Padilla-Gamiño JL, Pochon X, Bird C, Concepcion GT, Gates RD (2012) From parent to gamete: vertical transmission of symbiodinium (Dinophyceae) ITS2 sequence assemblages in the reef building coral montipora capitata. PLoS One 7:e38440
Parsons TR, Malta Y, Lalli M (1984) A manual of chemical and biological methods for seawater analyses. Pergamon Press, New York
Putnam HM, Edmunds PJ, Fan T-Y (2010) Effect of a fluctuating thermal regime on adult and larval reef corals. Invertebr Biol 129:199–209
Putnam HM, Mayfield AB, Fan TY, Chen CS, Gates RD (2013) The physiological and molecular responses of larvae from the reef-building coral Pocillopora damicornis exposed to near-future increases in temperature and pCO2. Mar Biol 160(8):2157–2173
R Core Team (2019) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria
Rosset S, Wiedenmann J, Reed AJ, D’Angelo C (2017a) Phosphate deficiency promotes coral bleaching and is reflected by the ultrastructure of symbiotic dinoflagellates. Mar Pollut Bull 118(1):180–187
Randall CJ, Szmant AM (2009) Elevated temperature reduces survivorship and settlement of the larvae of the Caribbean scleractinian coral, Favia fragum (Esper). Coral Reefs 28(2):537–545
Richmond RH (1981) Energetic considerations in the dispersal of Pocillopora damicornis (Linnaeus) planulae. In: Proceedings of the 4th international coral reef symposium, vol 2. pp 153–156
Richmond RH (1987) Energetics, competency, and long-distance dispersal of planula larvae of the coral Pocillopora damicornis. Mar Biol 93(4):527–533
Rivest EB, Hofmann GE (2014) Responses of the metabolism of the larvae of Pocillopora damicornis to ocean acidification and warming. PLoS ONE. https://doi.org/10.1371/journal.pone.0096172
Rivest EB, Chen C-S, Fan T-Y, Li H-H, Hofmann GE (2017) Lipid consumption in coral larvae differs among sites: a consideration of environmental history in a global ocean change scenario. Proc R Soc B: Biol Sci 284:20162825
Rogers CS (1990) Responses of coral reefs and reef organisms to sedimentation. Mar Ecol Prog Ser 62(1):185–202
Ross C, Ritson-Williams R, Olsen K, Paul VJ (2012) Short-term and latent post-settlement effects associated with elevated temperature and oxidative stress on larvae from the coral Porites astreoides. Coral Reefs 32(1):71–79
Rosset S, Wiedenmann J, Reed AJ, D’Angelo C (2017b) Phosphate deficiency promotes coral bleaching and is reflected by the ultrastructure of symbiotic dinoflagellates. Mar Pollut Bull 118(1–2):180–187
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Schnitzler CE, Hollingsworh LL, Krupp DA, Weis VM (2012) Elevated temperature impairs onset of symbiosis and reduces survivorship in larvae of the Hawaiian coral, Fungia scutaria. Mar Biol 159(3):633–642
Schwarz JA, Krupp DA, Weis VM (1999) Late larval development and onset of symbiosis in the scleractinian coral Fungia scutaria. Biol Bull 196(1):70–79
Serrano XM, Miller MW, Hendee JC, Jensen BA, Gapayao JZ, Pasparakis C, Grosell M, Baker AC (2018) Effects of thermal stress and nitrate enrichment on the larval performance of two Caribbean reef corals. Coral Reefs 37(1):173–182
Shantz AA, Burkepile DE (2014) Context-dependent effects of nutrient loading on the coral-algal mutualism. Ecology 95:1995–2005
Shantz AA, Lemoine NP, Burkepile DE (2016) Nutrient loading alters the performance of key nutrient exchange mutualisms. Ecol Lett 19(1):20–28
Silbiger NJ, Nelson CE, Remple K, Sevilla JK, Quinlan ZA, Putnam HM, Fox MD, Donahue MJ (2018) Nutrient pollution disrupts key ecosystem functions on coral reefs. Proc Biol Sci 285(1880):20172718
Smale DA, Wernberg T, Oliver EC, Thomsen M, Harvey BP, Straub SC, Burrows MT, Alexander LV, Benthuysen JA, Donat MG, Feng M (2019) Marine heatwaves threaten global biodiversity and the provision of ecosystem services. Nat Clim Change 4:1
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis, 2nd edn. In: Bulletin-Fisheries Research Board of Canada, vol 167
Suzuki G, Yamashita H, Kai S, Hayashibara T, Suzuki K, Iehisa Y, Okada W, Ando W, Komori T (2013) Early uptake of specific symbionts enhances the post-settlement survival of Acropora corals. Mar Ecol Prog Ser 494:149–158
Szmant AM (2002) Nutrient enrichment on coral reefs: Is it a major cause of coral reef decline? Estuaries 25(4):743–766
Therneau T (2015) A package for survival analysis in S. version 2.38. https://CRAN.R-project.org/package=survival
Vega Thurber RL, Burkepile DE, Fuchs C, Shantz AA, McMinds R, Zaneveld JR (2014) Chronic nutrient enrichment increases prevalence and severity of coral disease and bleaching. Glob Chang Biol 20(2):544–554
Wall C (2020) cbwall/Coral-larvae-temp-and-nutrients: temperature and nutrient effects on coral larvae (Version pub.ver). Zenodo. http://doi.org/10.5281/zenodo.3674230
Wall CB, Ritson-Williams R, Popp BN, Gates RD (2019) Spatial variation in the biochemical and isotopic composition of corals during bleaching and recovery. Limnol Oceanogr 64:2011–2028
Weis VM (2008) Cellular mechanisms of Cnidarian bleaching: stress causes the collapse of symbiosis. J Exp Biol 211(19):3059–3066
Wiedenmann J, D’Angelo C, Smith EG, Hunt AN, Legiret FE, Postle AD, Achterberg EP (2013) Nutrient enrichment can increase the susceptibility of reef corals to bleaching. Nat Clim Change 3:160–164
Wooldridge SA, Done TJ (2009) Improved water quality can ameliorate effects of climate change on corals. Ecol Appl 19(6):1492–1499
Yakovleva IM, Baird AH, Yamamoto HH, Bhagooli R, Nonaka M, Hidaka M (2009) Algal symbionts increase oxidative damage and death in coral larvae at high temperatures. Mar Ecol Prog Ser 378:105–112
Acknowledgements
This study was funded by the Paul G. Allen Family Foundation, Colonel Willys E. Lord and Sandina L. Lord Endowed Scholarship, an NSF graduate research fellowship to E.A.L., a Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) fellowship to M.R.S. and an Environmental Protection Agency STAR Fellowship Assistance Agreement (FP-91779401-1) to C.B.W. The views expressed in this publication have not been reviewed or endorsed by the EPA and are solely those of the authors. We thank the SOEST Laboratory for Analytical Biogeochemistry (SLAB) at the University of Hawai‘i at Mānoa for assistance with the nutrient analysis. We would also like to thank K. Hughes, J. Davidson, C. Drury, A. Huffmyer, C. Harris and D. Chee for their support. This is HIMB contribution 1786 and SOEST contribution number 10905. We dedicate this manuscript to the life and legacy of our dear friend and mentor Dr. Ruth Gates.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Topic Editor Anastazia Banaszak
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kitchen, R.M., Piscetta, M., de Souza, M.R. et al. Symbiont transmission and reproductive mode influence responses of three Hawaiian coral larvae to elevated temperature and nutrients. Coral Reefs 39, 419–431 (2020). https://doi.org/10.1007/s00338-020-01905-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-020-01905-x