Abstract
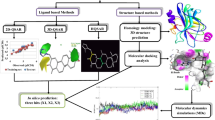
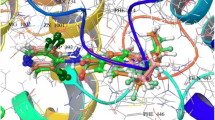
Valosine containing protein (VCP/p97), which involves in several important cellular functions and plays an important role in ubiquitin-mediated misfolding protein degradation, has been found to be a novel target for the treatment of a range of cancers, such as lung cancer and breast cancer. In this study, the atom-based three-dimensional quantitative structure–activity relationship (3D-QSAR) and docking models were developed using PHASE and GLIDE modules of Schrödinger software, respectively. The theoretical models were generated from 38 N-benzylpyrimidin-4-amine inhibitors of p97. An AADRRR model consisting of two hydrogen bond acceptors (A), one hydrogen bond donor (D), and three aromatic rings (R) was obtained. Thirty eight derivatives were divided into a training set with 27 molecules to generate 3D-QSAR models and a test set with 11 molecules to validate 3D-QSAR model. A robust QSAR model with good prediction in internal and external verification was constructed, where R2, Q2, and Pearson-R were 0.924, 0.701, and 0.8783, respectively. QSAR model showed the hydrogen bond donor, electron-withdrawing group, and hydrophobic characteristics affecting the p97 activity. Molecular docking studies indicated that the H-bond and hydrophobic interactions existed between the inhibitors and p97, which was consistent with the results of 3D-QSAR. These results provided some useful information for designing new and effective p97 inhibitors.






Similar content being viewed by others
References
Alverez C, Bulfer SL, Chakrasali R, Chimenti MS, Deshaies RJ, Green N, Kelly M, LaPorte MG, Lewis TS, Liang M, Moore WJ, Neitz RJ, Peshkov VA, Walters MA, Zhang F, Arkin MR, Wipf P, Huryn DM (2015) Allosteric indole amide inhibitors of p97: Identification of a novel probe of the ubiquitin pathway. ACS Med Chem Lett 7:182–187
Bursavich MG, Parker DP, Willardsen JA, Gao ZH, Davis T, Ostanin K, Robinson R, Peterson A, Cimbora DM, Zhu JF, Richards B (2010) 2-Anilino-4-aryl-1,3-thiazole inhibitors of valosin-containing protein (VCP or p97). Bioorg Med Chem Lett 20:1677–1679
Chapman E, Fry AN, Kang MJ (2011) The complexities of p97 function in health and disease. Mol Biosyst 7:700–710
Chapman E, Maksim N, de la Cruz F, La Clair JJ (2015) Inhibitors of the AAA+ chaperone p97. Molecules 20:3027–3049
Chou TF, Brown SJ, Minond D, Nordin BE, Li K, Jones AC, Chase P, Porubsky PR, Stoltz BM, Schoenen FJ, Patricelli MP, Hodder P, Rosen H, Deshaies RJ (2011) Reversible inhibitor of p97, DBeQ, impairs both ubiquitin-dependent and autophagic protein clearance pathways. Proc Natl Acad Sci USA 108:4834–4839
Chou TF, Deshaies RJ (2011) Development of p97 AAA ATPase inhibitors. Autophagy 7:1091–1092
Ding R, Zhang T, Wilson DJ, Xie JS, Williams J, Xu Y, Ye YH, Chen LQ (2019) Discovery of irreversible p97 inhibitors. J Med Chem 62:2814–2829
Dixon SL, Smondyrev AM, Knoll EH, Rao SN, Shaw DE, Friesner RA(2006) PHASE: a new engine for pharmacophore perception 3D QSAR model Dev, 3D database Screen: 1 Methodol preliminary results. J Comput Aid Mol Des 20:647–671
Evans DA, Doman TN, Thorner DA, Bodkin MJ (2007) 3D QSAR methods: phase and catalyst compared. J Chem Inf Model 47:1248–1257
Gao L, Zu M, Wu S, Liu AL, Du GH (2011) 3D QSAR and docking study of flavone derivatives as potent inhibitors of influenza H1N1 virus neuraminidase. Bioorg Med Chem Lett 21:5964–5970
GLIDE, version 6.7 (2015) Schrödinger, LLC, New York
Le Moigne R, Aftab BT, Djakovic S, Dhimolea E, Valle E, Murnane M, King EM, Soriano F, Menon MK, Wu ZY, Wong ST, Lee GJ, Yao B, Wiita AP, Lam C, Rice J, Wang J, Chesi M, Bergsagel PL, Kraus M, Driessen C, Kiss von Soly S, Yakes FM, Wustrow D, Shawver L, Zhou HJ, Martin TG, Wolf JL, Mitsiades CS, Anderson DJ, Rolfe M (2017) The p97 Inhibitor CB-5083 is a unique disrupter of protein homeostasis in models of multiple myeloma. Mol Cancer Ther 16:2375–2386
LigPrep, version 3.4 (2015) Schrödinger, LLC, New York
Maestro, version 10.2 (2015) Schrödinger, LLC, New York
Meyer H, Bug M, Bremer S (2012) Emerging functions of the VCP/p97 AAA-ATPase in the ubiquitin system. Nat Cell Biol 14:117–123
Meyer H, Weihl CC (2014) The VCP/p97 system at a Slance: connecting cellular function to disease pathogenesis. J Cell Sci 127:3877–3883
PHASE, version 4.3 (2015) Schrödinger, LLC, New York
Qiu Y, Tomita Y, Zhang B, Nakamichi I, Morii E, Aozasa K (2007) Pre-B-cell leukemia transcription factor 1 regulates expression of valosin-containing protein, a gene involved in cancer growth. Am J Pathol 170:152–159
Segura-Cabrera A, Tripathi R, Zhang XY, Gui L, Chou TF, Komurov K (2017) A structure- and chemical genomics-based approach for repositioning of drugs against VCP/p97 ATPase. Sci Rep 7:1–10
Smith MH, Ploegh HL, Weissman JS (2011) Road to ruin: targeting proteins for degradation in the endoplasmic reticulum. Science 334:1086–1090
Song CC, Wang Q, Li CC (2003) ATPase activity of p97-valosin-containing protein (VCP). D2 mediates the major enzyme activity, and D1 contributes to the heat-induced activity. J Biol Chem 278:3648–3655
Suryanarayanan V, Sudha A, Rajamanikandan S, Vanajothi R, Srinivasan P (2012) Atom-based 3D QSAR studies on novel N-β-d-xylosylindole derivatives as SGLT2 inhibitors. Med Chem Res 22:615–624
Tang WK, Odzorig T, Jin W, Xia D (2019) Structural basis of p97 inhibition by the site-selective anticancer compound CB-5083. Mol Pharm 95:286–293
Tropsha A (2010) Best practices for QSAR model development, validation, and exploitation. Mol. Inform 29:476–488
van Vlijmen H, Desjarlais RL, Mirzadegan TJ (2017) Computational chemistry at Janssen. J Comput Aid Mol Des 31:267–273
Wang Q, Song CC, Li CC (2004) Molecular perspectives on p97-VCP: progress in understanding its structure and diverse biological functions. J Struct Biol 146:44–57
Wang XY, Bai EH, Zhou H, Sha SJ, Miao H, Qin YR, Liu ZG, Wang J, Zhang HY, Lei M, Liu J, Hai O, Zhu YQ (2019) Discovery of a new class of valosine containing protein (VCP/P97) inhibitors for the treatment of non-small cell lung cancer. Bioorg Med Chem 27:533–544
Zhang T, Midshra P, Hay BA, Chan D, Guo M (2017) Valosin-containing protein (VCP/p97) inhibitors relieve Mitofusin-dependent mitochondrial defects due to VCP disease mutants. eLife 6:1–28
Zhou HJ, Wang JH, Yao B, Wong S, Djakovic S, Kumar B, Rice J, Valle E, Soriano F, Menon MK, Madriaga A, Kiss von Soly S, Kumar A, Parlati F, Yakes FM, Shawver L, Moigne RL, Anderson DJ, Rolfe M, Wustrow D (2015) Discovery of a first-in-class, potent, selective, and orally bioavailable inhibitor of the p97 AAA ATPase (CB-5083). J Med Chem 58:9480–9497
Acknowledgements
This research work was supported by the Technology Support Program of Science and Technology Department of Jiangsu Province (BE2015703) and the Major Program for the Natural Science Fundamental Research of the Higher Education Institutions of Jiangsu Province (15KJA350002). We thank Yadong Chen’s group (College of Science, China Pharmaceutical University) for providing the computational facilities, Schröedinger, for this research work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gu, C., Zhang, Y., Xie, X. et al. Pharmacophore modeling, atom-based 3D-QSAR and molecular docking studies on N-benzylpyrimidin-4-amine derivatives as VCP/p97 inhibitors. Med Chem Res 29, 727–737 (2020). https://doi.org/10.1007/s00044-020-02517-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02517-4