Abstract
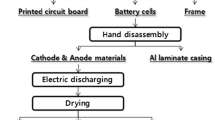
During the mass recycling of spent lithium-ion battery (LIB) packs, the packs that have not been disassembled are heat-treated to remove organic substances; further, the valuable metals obtained in the burnt product are recovered in the leaching process. Numerous methods have been reported for the efficient recovery of valuable metals in the leaching process; however, the material balance of the constituents of LIB during heat treatment has not been elucidated yet. In this study, the spent pack of lithium-ion secondary battery was employed wherein the positive electrode was essentially Li(Ni1/3Mn1/3Co1/3)O2 coated on an Al foil and the negative electrode was graphite coated on a Cu foil. Additionally, LiPF6 dissolved in propylene carbonate was used as the electrolyte, and the outer package was a cylindrical can composed of steel. After heat treatment of the spent LIB packs, Al2O3, Li2O-Al2O3-based compounds, MnO, Co, Ni, and Cu were found to be present in the cylindrical steel can of the burned product. Although high concentrations of the electrolyte elements of F and P were also present in the burned product, the amount recovered as dust from the exhaust gas treatment equipment was found to be higher as compared to that of the other elements; this amount was observed to reach or exceed 30 pct. Moreover, the constituent elements of the electrolyte such as Li, F, and P were converted to Li2O, LiF, and P2O5, leading to erosion of refractories.










Similar content being viewed by others
References
1. B.A. Johnson and R.E. White: Journal of Power Sources, 1998, vol. 70, pp. 48-54.
2. M. Wakihara: Mater. Sci. Eng. R, 2001, vol. 33, pp. 109-134.
3. R. Moshtev and B. Johnson: Journal of Power Sources, 2000, vol. 91, pp. 86-91.
Fuji Keizai Co Ltd (2016) Overall Status Survey of Battery Related Market, 2016, vol I. Fuji Keizai Co. Ltd, Tokyo (in Japanese)
5. A. Yoshino: Bunseki, 2013, vol. 10, pp. 580-584
N. Nakatani, K. Moriwaki, and N. Yoshinaga: IEICE Technical Report. CPM, Electronic Components/Materials, 97(221), 15-22, 1997-08-05 (in Japanese).
7. Y. Kim, M. Matsuda, A. Shibayama, T. Miyazaki, and T. Hujita: Shigen-to-Sozai, 2002, vol. 118, pp. 687-693.
8. J. Xu, H.R. Thomas, R.W. Francis, K.R. Lum, J. Wang, and B. Liang: Journal of Power Sources, 2008, vol. 177, pp. 512–527.
9. T.G. Maschler, B. Friedrich, R. Weyhe, H. Heegn, and M. Rutz: Journal of Power Sources, 2012, vol. 207, pp. 173-182.
10. S. Koyanaka, K. Horai, H. Kawai, S. Kato, J. Shibata, N. Murayama, T. Ohki, and M. Masuda: J. MMIJ, 2012, vol. 128, pp. 232-240.
11. K. Horai, J. Shibata, N. Murayama, S. Koyanaka and M. Niinae: J. Japan Inst. Met. Mater., 2014, vol. 78, pp. 250-257.
12. K. Horiuchi, M. Matsuoka, C. Tokoro, S. Owada, and S. Usui: Kagaku Ronbunshu, 2017, vol. 43, pp. 213-218.
Nie X, Xi X, Yang Y, Ning Q, Guo J, Wang M, Gu Z, Wu X (2019) Electrochim Acta. https://doi.org/10.1016/j.electacta.2019.134625
14. S. Junjie, P. Chao, C. Min, L. Yun, E. Hurman, K. Lassi, L. Mari, T. Pekka, and J.Ari: JOM, 2019, vol. 71, No. 12, pp. 4473-4482.
15. L. Li, P. Zheng, T. Yang, R. Sturges, M. Ellis, W. Michael, and Z. Li: JOM, 2019, vol. 71, pp. 4457-4464.
16. W. Wang, Y. Han, T. Zhang, L. Zhang, and S. Xu: ACS Sustainable Chem. Eng., 2019, vol. 7, No. 19, pp. 16729-16737.
17. C. Peng, C. Chang, Z. Wang, B.P. Wilson, F. Liu, and M. Lundström: JOM, 2019, https://doi.org/10.1007/s11837-019-03832-x
18. J. Lin, C. Liu, H. Cao, Y. Yang, R. Chen, L. Li, and Z. Sun: Green Chemistry, 2019, vol. 21, pp. 5904-5913.
Murakami Y, Matsuzaki Y, Kamimura T, Nishiura T, Masuda K, Shibayama A, Inoue R (2019) Ceram Int. https://doi.org/10.1016/j.ceramint.2019.12.182
Chemical Society of Japan, ed.: Handbook of Chemistry: Pure Chemistry, 5th revised edition, 2004 (in Japanese).
Cook LP, Plante ER (1992) Ceram Trans Fabr Prop Lithium Ceram 3 27:193-222
22. K.S. Gavrichev, G.A. Sharpataya, L.N. Golushina, V.N. Plakhotnik, I.V. Goncharova: Russian J. Inorg. Chem., 2002, vol. 47, pp. 940-944.
23. E. Aukrust and A. Muan: Trans. Metall. Soc. AIME, 1964, vol. 230, pp. 378-382.
24. A. Rahmel and P.J. Spencer: Oxid. Met., 1991, vol. 35, pp. 53-68.
25. W.C. Hahn, Jr. and A. Muan: Am. J. Sci., 1960, vol. 258, pp. 66-78.
26. J. Hu, J. Zhang, H. Li, Y. Chen, and C. Wang: J. Power Sources, 2017, vol. 351, pp. 192-199.
27. O. Knacke, O. Kubaschewski and K. Hesselmann: Thermochemical Properties of Inorganic Substances, 2nd ed., 1991, Springer-Verlag, Berlin.
28. A. Khorassani, G. Izquierdo and A.R. West: Mater. Res. Bull., 1981, vol.16, pp. 1561-1567.
29. V.P. Kochergin, L.V. Paderova, E.G. Viskova, S.S. Nokhrin, and N.N. Anikina: Rasplavy, 1987, vol. 1, pp. 102-109.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted August 8, 2019.
Rights and permissions
About this article
Cite this article
Murakami, Y., Matsuzaki, Y., Murakami, K. et al. Recovery Rates of Used Rechargeable Lithium-Ion Battery Constituent Elements in Heat Treatment. Metall Mater Trans B 51, 1355–1362 (2020). https://doi.org/10.1007/s11663-020-01834-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-020-01834-8