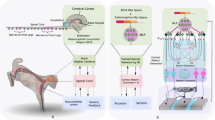
Abstract
Study design
Prospective, quasi-experimental study, pre- and post-design, single arm study.
Objectives
Investigate whether persons affected by SCI can safely experience walking function using Robotic Exoskeletons and Functional Electrical Stimulation (FES).
Setting
Inpatient
Methods
52 persons with SCI were recruited (36 completed the protocol) and assigned to one of two groups based on their Lower Limb Motor Scores (LEMS): Group A: LEMS ≥ 10 and Group B: LEMS < 10. Participants in Group A (n = 19) underwent 20 sessions of Robot-Assisted Gait Training (RAGT) on a treadmill followed by 20 sessions of FES during Overground Gait (FES-OG). Participants in Group B (n = 17) received 20 sessions of FES-cycling followed by 20 sessions of overground RAGT. The main outcome measures were: WISCI-II, 10MWT, 6MWT, TUG and SCIM-II.
Results
36 persons completed the study with no complications; only 4 of the 16 dropped out because of mild complications during the RAGT. Participants in Group A exhibited significant improvements in WISCI-II, 10MWT, 6MWT and TUG (p < 0.05), while those in Group B did not significantly improve their gait function but their walking velocity and resistance with the assistance of the robotic exoskeleton increased. SCIM-II scores increased followed therapy only in Group A.
Conclusions
Persons affected by SCI can safely experience their walking function with RAGT and FES therapy; only few mild complications were observed. Our data provides initial evidence of the potential value of these technologies, especially in persons with SCI having LEMS > 10.
Similar content being viewed by others
Introduction
Walking functions are frequently lost or impaired in persons affected by spinal cord injury (SCI) and SCI-related motor impairments are associated with general dissatisfaction and reduced quality of life [1]. The recovery of locomotor function is thus considered an important rehabilitation goal both by patients, their relatives and friends, physicians, physiotherapists and nurses [2].
Gait recovery is related to neurological function. The correlation between the American Spinal Injury Association Impairment Scale (AIS) [3] and the prognosis of gait recovery is important to identify the correct rehabilitation programme in order to obtain the best results [4].
For instance, persons with AIS A (complete sensory and motor lesion) have a 2.5% probability of neurological recovery, whereas the probability of regaining the ability to walk is 33% in persons with AIS B (complete motor, incomplete sensory lesion), and 75% for AIS C (incomplete lesion with <50% Motor Scores ≥3) and 100% in AIS D (incomplete lesion with ≥50% Motor Scores ≥3), respectively [5]. Higher motor strength and “pinprick” sensory integrity are associated with a strong recovery [4].
The introduction of Body Weight Support (BWS) during walking on treadmill has increased the clinical application of gait training in persons with a SCI. It permits a more normal walking pattern compared with the gait practice on treadmill without BWS, as BWS has been shown to reduce the muscle spasticity [6]. Robotic devices for treadmill training with BWS have been developed to improve gait recovery; these technologies facilitate a high quality of gait training, which maintains very similar joint ranges of motion during the different gait phases. In particular, Robot-Assisted Gait Training (RAGT) on a treadmill activates central pattern generator using a robotic exoskeleton in addition to BWS [7]. This training modality induces plastic changes in muscle recruitment and in sensory-motor cortex in patients with incomplete lesions [8,9,10].
Nowadays adjustable exoskeletons for overground RAGT are available for walking rehabilitation [11]. People with complete spinal lesions can walk with the assistance of robotic exoskeletons [12], thereby increasing their daily physical activity and improving their body composition [13].
Functional Electrical Stimulation (FES) involves the electrical activation of muscles by means of stimulation in order to reproduce functional activities. FES for Overground Gait (FES-OG) could be used in persons with incomplete SCI to rehabilitate different phases of the gait cycle [14]. FES-Cycling (FES-C) can be also used to reproduce a cycling motion in persons with SCI to induce an improvement in body structures and quality of life [15,16,17,18].
At present many robots for walking are available [19, 20]; the BWS treadmill-based robotic training (BWSTRT) has been used to recover walking ability in incomplete SCI persons [21]. The combination of this training with a following FES-OG is not proposed till now, therefore its effects on gait rehabilitation of incomplete SCI persons should be investigated.
More recently robotic exoskeletons for overground walking have been proposed: they can be used in persons with both incomplete and complete SCI [19, 20]. The rationale of using an overground robotic system in persons with complete SCI has twofold aims: (1) to verify if the application of this technology produces any relevant collateral effects; (2) to analyse the possibility to increase the person’s ability of using an overground robot with the objective of walking in the daily time with a personalised exoskeleton.
In addition, FES-C training can be used to increase muscle, joint and bone tropisms, which are usually compromised in persons with complete SCI lesions [15,16,17,18].
The aim of this study was to investigate whether persons affected by SCI can safely experience their walking function via RAGT and FES therapy.
Methods
Study design
Two-hundred-forty-nine SCI inpatients were assessed for inclusion in this study between January 2015 and January 2018, according to the eligibility criteria listed in Table 1. After a neurological evaluation, participants were allocated to one of two rehabilitation groups, according to their Lower Limb Motor Scores (LEMS): those with an LEMS ≥ 10 were assigned to Group A, and those with LEMS Scores < 10 were assigned to Group B.
The following evaluations were administered at the beginning and at the end of the training protocol: (i) clinical neurological evaluation using the ASIA Impairment Scale (AIS) [3]; (ii) general function using the Spinal Cord Independence Measure (SCIM-II) [22]; (iii) locomotor functional evaluation using the Walking Index for SCI scale (WISCI-II) [23].
The safety of the proposed rehabilitation treatments was assessed via the number and severity of possible complications.
Group A
The first group (Group A) underwent 20 sessions of BWSTRT followed by 20 sessions of lower limb FES-OG. The BWSTRT was carried out by means of a bilateral robotic exoskeleton system (Lokomat system, Hocoma, Volketswil, Switzeraland) with two degrees of freedom per leg for sagittal knee and hip rotation under BWS [7]. The FES device for FES-OG (Microstim, Medel, Hamburg, Germany) provided two channels stimulation of the hip and/or ankle flexor muscles, with gait phase synchronisation enabled by pressure sensors located on the sole of each foot. After an initial session of 30 min for subject familiarisation with the use of the exoskeleton on a treadmill, the other sessions of BWSTRT had a duration ranging from 40 to 60 min. The participants used FES-OG device on only one leg in order to stimulate hip and ankle flexors, or on both legs, to stimulate only the hip flexors or the ankle flexors bilaterally; the choice was centred on patients’ specific needs.
Stimulation parameters: FES-OG was delivered using a biphasic square pulse, with amplitude of 30–70 mA, frequency of 35 Hz, and pulse width of 250 μs. Subjects completed three training sessions per week, with each session lasting ~30 min. The whole rehabilitation programme lasted 14 weeks.
The gait assessment of Group A participants was performed, before and after training protocol, without the exoskeleton, but with the use of any additional assistive devices (cane, etc.) or orthoses routinely used by the patient and needed for safety. Outcome measures assessed included the 10-Metres Walk Test (10MWT), 6-Minutes Walk Test (6MWT) and Timed Up and Go test (TUG) [24].
Group B
Participants of Group B underwent 20 sessions of FES-C training for reconditioning the joints and the muscles because most had previously experienced long periods of prior immobilisation. After one session of familiarisation, they were trained with 20 sessions of walking by using the Overground Robotic Exoskeleton. FES-C is a FES system, which permits to integrate a cyclo-ergometer system with an electronic interface (Pegaso, Biotech Srl, Bologna, Italy). The quadriceps and the femoral biceps of both legs were stimulated using an alternating biphasic square alternated pulse, amplitude from 35 to 75 mA for quadriceps and from 25 to 50 mA for biceps, frequency 50 Hz, pulse width 300 μs. Each FES-C session lasted about 30 min.
To evaluate the modifications of thigh volume after FES-C training, on the two legs, four thighs circumferences were measured in the first and last sessions at four different distances (5, 10, 15 and 20 cm, respectively) proximal to the superior margin of the patella, towards the hip.
An Overground Robotic Exoskeleton Training (ORET) was performed by using a powered exoskeleton for overground walking (Ekso, Ekso Bionics, Richmond, CA, USA) [11].
Each ORET session lasted from 50 to 68 min, and participants completed three sessions per week, for a total rehabilitation programme of 14 weeks.
The gait assessment of Group B participants was performed by using the overground robotic exoskeleton, with the following outcome measures: 10MWT-exo, 6MWT-exo, TUG-exo and Endurance Test (Endurance-exo) at the first and last session of training; during Endurance-exo each participant was asked to walk with the assistance of the robotic exoskeleton as long as possible without any break (the score was expressed in seconds). During the “6MWT-exo” patients performed from 1 to 3 turns.
Statistical analysis
Statistical analysis was performed using Sigma Stat v3.5 statistical package (System Software Inc., San Jose, CA, USA). The results on thigh circumferences, walking ability tests and Endurance-exo are expressed as Mean ± Standard Deviation (M ± SD), those on SCIM-II and WISCI-II as median and interquartile range (median; IQR). The differences between Pre- and Post-treatment scores were analysed by means of the Student t test and the Wilcoxon–Mann–Whitney test. Significance has been set at p < 0.05.
Results
Out of two-hundred and forty-nine inpatients with SCI evaluated in three years, 52 persons (21%) were recruited: 38 males, 14 females, ratio men/women: 2,7:1, mean age (SD): 49.0 (15.3) years, range: 22–75 years (Table 2).
Different distributions of participants with Traumatic (T) vs. Non-Traumatic (NT) and complete vs. incomplete lesions were observed between the two groups: Group A included a greater number of NT and incomplete motor injures, whereas in the Group B a greater number of T and complete motor injuries were observed. The distribution of time since injury between the two groups is different with a predominance of <1 year in the Group A. Even the age was different in the two groups: the persons of Group A were older on average than those in Group B, in line with epidemiologic literature reporting a higher mean age in persons with NT SC lesions [25, 26].
Sixteen participants (eight from Group A and eight from Group B) did not complete the treatment but only four from Group B dropped out because of complications during the ORET: one presented a shoulder pain during the ambulation, one had muscular haemorrhage and two developed skin lesions. Six persons stopped the training due to SCI comorbidity and six abandoned the training for personal reasons. Except in two cases, showing a shift respectively from AIS B to C and AIS C to D, AIS did not change after the rehabilitation treatment.
Functional electrical stimulation
Thirteen participants of group A used FES-OG on only one limb. Among them FES-OG was applied on the ankle dorsal flexors (n = 8) or on both hip and ankle flexors (n = 5). Bilateral FES application was used in six persons on ankle dorsal flexors. Participants of Group B (n = 17) underwent 20 sessions of FES-C in order to improve the bone, muscle and joint reconditioning. Thigh circumferences increased significantly after the 20 FES-C sessions (Fig. 1) (Table 3).
Pre- (grey) and post- (black) FES-C treatment bilateral thigh circumferences (expressed as mean ± standard deviation) recorded at distance (d) of 5 cm (top row, left plot), 10 cm (top row, right plot), 15 cm (bottom row, left plot) and 20 cm (bottom row, right plot) from the knee cap (*p < 0.05) (colour figure online).
Group A
In Group A, seven participants, whose SCI occurred recently (i.e., <12 months), were unable to walk (WISCI-II score = 0) before gait training; after the BWSTRT, WISCI-II score increased. No further score increase were observed after the FES-OG (Table 4); on the other hand, a significant difference of WISCI-II score was observed between pre- and post-treatment (i.e., BWSTRT plus FES-OG) (Table 4).
Twelve participants were able to walk and a significant increase of the WISCI-II scores after the BWSTRT (Table 4), but no further increase after the FES-OG, was observed (Table 4).
It was possible to perform the walking tests (before and after the training) only in seven participants among them with an initial WISCI-II score ≥6: 10MWT, 6MWT and TUG improved after the BWSTRT (Table 4); no further improvement was observed after the FES-OG; the difference of 10MWT, 6MWT and TUG score between pre- and post- treatment (BWSTST plus FES-OG) was significant (Fig. 2) (Table 4).
Median SCIM-II scores of persons in Group A was 46 (IQR 25.2–67.0) before treatment and 58 (IQR 40.5–85.7) after the training (BWSTRT plus FES-OG) with the difference registering as significant (p = 0.001).
Group B
All participants of Group B (n = 17) were unable to walk at baseline (WISCI-II score = 0) and exhibited no change in WISCI-II score at the end of the training. During the training with the overground exoskeleton, the walking time and the number of steps increased from 16.9 (5.6) min and 282 (129) steps in the first session to 30.5 (16.4) min and 906 (489) steps in the last session, respectively.
For the Group B, walking tests were carried out using the overground robotic exoskeleton: 10MWT-exo, 6MWT-exo, TUG-exo and Endurance-exo score improved significantly from the first session and the last session of ORET (Fig. 3, Table 5).
10MWT-exo (seconds) (top row, left plot), 6MWT-exo (metres) (top row, right plot), TUG-exo (seconds) (bottom row, left plot), Endurance-exo (seconds) (bottom row, right plot) at pre- (grey) and post- (black) treatment in n = 17 persons of Group B. Note: the tests were carried out with the assistance of the overground robotic exoskeleton; *p < 0.05 (colour figure online).
In Group B, no statistically significant difference was observed (p = 0.59) in the SCIM-II scores before (median 66, IQR 60.0–72.0) vs. after (median 67, IQR 61.0–72.2) FES-C plus ORET training.
Discussion
This study confirms that when using robotic exoskeletons for walking, careful monitoring is recommended to avoid skin lesions [11]. Specifically, 8% of persons of Group B exhibited skin redness at the end of one of ORET sessions, and further adverse health effects (e.g., bedsores) were avoided via the prompt interruption of the training. Bone density should also be examined before training because bone fractures have been reported in persons with SCI undergoing overground robotic exoskeleton gait training [27].
In the clinical spinal cord unit where study participants were recruited, an osteoporosis evaluation and a treatment protocol were applied as standard care, thus reducing the risk of bone fractures. During the ORET, a walker or two crutches are held by the upper limbs; this can lead to a musculoskeletal pain, in particular localised in the shoulders [27]. Muscle haemorrhage lesions are another adverse event that can arise in persons with SCI during robotic gait training, and the anticoagulant drugs often used in these persons could be, partially responsible.
Enroled participants accounted for just 21% of the examined persons with SCI lesions. This low enrolment percentage may be attributed to several factors, including (a) the time-intensiveness of the prescribed rehabilitation programme (requiring from two to four months of uninterrupted participation); (b) more than one instrument with different eligibility criteria (Table 1) was used to complete the training; (c) persons with SCI have a delicate state of health with numerous concurrent comorbidity, often requiring interruption of training. Among these factors, this last reason accounted for a high percentage (38%) of study withdrawals, with a comparable number of participants citing personal reasons relating to their difficulty maintaining participation for the long duration of the rehabilitation programme (38%).
Only 24% of study withdrawals were attributed to complications related to the robotic training, including skin lesions, pain and muscle haemorrhagic lesions.
The safety of the proposed rehabilitation programme as regards Group A (i.e., higher functioning group) was demonstrated by the absence of any such complication. On the other hand, some mild complications affected participants in Group B.
The selected study population comprises a range severity of functional deficits, including persons with paraplegia (44%), tetraplegia in high cervical injury (11%), and tetraplegia in low cervical injury (10%), as well as a large portion of participants (35%) with AIS D lesions and a clinical prognosis of a strong possibility of complete gait recovery.
Group A
Gait recovery was evaluated using validated and widely accepted clinical assessment scales, including the 10MWT, 6MWT and TUG [24]. The results of the present study suggest that the application of a rehabilitation protocol featuring combined BWSTRT plus FES-OG allows many persons affected by incomplete SCI to regain locomotor function.
Participants in Group A increased their mean walking speed by 30% (0.7 ± 0.4 m/s), reaching a value similar to uninjured individuals (0.8 m/s). This increased walking velocity and resistance was observed at the end of the treatment. In addition, an improvement of 58 and 41% was observed in resistance and transfer ability, respectively (Table 4).
On the other hand, persons who were not able to walk before the treatment regained their walking ability after the BWSTRT plus FES-OG. The use of robotic exoskeletons facilitates the walking recovery in persons with SCI and LEMS ≥ 10, probably by activating a neural plasticity: in fact, all persons with SCI recruited recovered their walking capacity [8, 9].
Group B
Persons in Group B did not regain significant walking function on the clinical scales employed in this study. In fact, the increase observed in the walking ability (10MWT-exo, 6MWT-exo and TUG-exo) and endurance (Endurance-exo) means that a better skill in the use of the overground robotic exoskeleton for walking was reached, thus employing the most of the device’s possibilities.
The gait speed during the ORET increased significantly from 0.11 (0.04) m/s to 0.15 (0.04) m/s, which remains clearly slower than the above-mentioned normal gait speed for healthy individuals.
Other factors which may influence the gait speed achieved in overground exoskeletons are highlighted in the medical literature. For instance, Louie and colleagues found significant correlations between walking velocity and increasing age, injury level and number of training sessions [28]. According to the literature, the mean number of rehabilitation sessions should be around 20 sessions; there are published experiences of persons who practised exoskeleton training for longer period obtaining better results [29].
At present, persons with SCI and poor lower limb motor activity can experience walking using overground robotic exoskeletons, with better health feeling and an increased ability to use the device. The use of robotic exoskeletons for walking may increase functionality in activities of daily living, thus contributing to improve the body composition [12, 13].
Integration of gait training with exoskeleton and FES
The primary innovation of this study is represented by the integration of multiple advanced technologies in the functional rehabilitation of persons with SCI, including robotic exoskeletons, FES-C and FES-OG, with results supporting their combined use in multimodal rehabilitative programmes. In accordance with this opinion, a consensus among different clinical centres has been reported, applying across a variety of rehabilitation devices used by different rehabilitation hospitals, which develop and implement their own protocols [30].
In the present study, persons with SCI and LEMS < 10 underwent a joint, bone and neuromuscular reconditioning using FES-C before starting the robotic training. The results confirm that the thigh circumferences significantly increased after FES-C, in accordance with higher muscle volumes observed elsewhere [31]. The introduction of FES-C training before the robotic training requires participants to wait for a longer period before obtaining the physical and psychological benefits of the exoskeleton training, but has been reported to improve the safety of its use [32]. Persons with SCI, who have less compromised joint, bone and muscle composition thanks to residual motor voluntary activity at sublesional levels (LEMS ≥ 10), can start directly the robotic walking training. In this subgroup, a significant increase in the SCIM-II scores was observed after the treatment.
The 6-month and 12-month follow-up data, currently under investigation, may demonstrate a long-term improvement in terms of walking function and independence.
Study limitations
The design of the current study is not sufficient to prove a cause-effect relationship due to the lack of a control group, a randomisation procedure, a sample size computation for statistical power and blinded assessors. These limitations should be carefully considered when weighing the strength of the results. Moreover, the rehabilitation protocol was challenging due to the required participant engagement of 8–12 weeks, and not all persons with SCI were eligible due to the instrumentation-specific requirements.
These limitations may have introduced a subject selection bias of the studied population, wherein only persons who have not contraindications regarding the use of the devices utilised in this study were included in the training. On the other hand, participants with different levels of functional impairment (complete, incomplete; AIS A, B, C and D) different spinal lesion levels of functional impairment (from C4 to L3), and different lesion onset have been included.
This subject heterogeneity could be a limitation of the study, as it resulted in a small sample size for the subgroups analysed.
Conclusions
Although the present study design was not sufficient to prove a direct cause-effect relationship, the main findings of this study provide some encouraging evidence for the positive response of persons with SCI to multimodal rehabilitation paradigms using advanced robotic gait assistance and FES technologies. Persons affected by SCI, who have no contraindications to the use of robotic exoskeletons or FES, can safely experience walking function using these technologies, the use of which can contribute to improve the gait abilities in persons with SCI having LEMS > 10. In particular, BWSTRT followed by FES-OG training in persons with a relevant motor strength preservation improved not only the gait velocity and resistance, but also their functional walking abilities as assessed using clinical scales. On the other hand, persons with no or low motor strength preservation may experience walking function by means of the assistance of the overground robotic exoskeleton.
An ORET carried out after body reconditioning with FES-C is likely to be able to increase their abilities to interact with the device.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Stevens S, Caputo J, Fuller D, Morgan DW. Physical activity and quality of life in adults with spinal cord injury. J Spinal Cord Med. 2008;31:373–8.
Simpson LA, Eng JJ, Hsieh JT, Wolfe DL, and the SCIRE Research Team. The health and life priorities of individuals with spinal cord injury: a systematic review. J Neurotrauma. 2012;29:1548–55.
Kirshblum SC, Burns SP, Biering-Sorensen F, Donovan W, Graves DE, Jha A, et al. International standards for neurological classification of spinal cord injury. J Spinal Cord Med. 2011;34:535–46.
Moon J, Yu J, Choi J, Kim MY, Min K. Degree of contributions of motor sensory scores to predict gait ability in patients with incomplete spinal cord injury. Rehabil Med. 2017;41:969–78.
Van Middendorp JJ, Hosman AJ, Pouw MH, EM-SCI Study Group, Van de Meent H. ASIA impairment scale conversion in traumatic SCI: is it related with the ability to walk? A descriptive comparison with functional ambulation outcome measures in 273 patients. Spinal Cord. 2009;47:555–60.
Visintin M, Barbeau H. The effect of body weight support on the locomotor pattern of spastic paretic patients. Can J Neurol Sci. 1989;16:315–25.
Colombo G, Joerg M, Schreier R, Dietz V. Treadmill training of paraplegic patients using a robotic orthosis. J Rehabil Res Dev. 2000;37:693–700.
Mazzoleni S, Boldrini E, Laschi C, Carrozza MC, Stampacchia G, Rossi B. Change on EMG activation in healthy subjects and incomplete SCI patients following a robot-assisted locomotor training, in Proc. International Conference on Rehabilitation Robotics 2011, Zurich, Switzerland, pp. 864–9.
Winchester P, McColl R, Querry R. Changes in supraspinal activation patterns following robotic locomotor therapy in motor-incomplete spinal cord injury. NeuroRehab Neural Repair. 2005;19:313–24.
Hubli M, Dietz V. The physiological basis of neurorehabilitation–locomotor training after spinal cord injury. J Neuroeng Rehabil. 2013;10:5. https://doi.org/10.1186/1743-0003-10-5.
Baunsgaard CB, Nissen UV, Brust AK, Frotzler A, Ribeill C, Kalke YB, et al. Gait training after spinal cord injury: safety, feasibility and gait function following 8 weeks of training with the exoskeletons from Ekso Bionics. Spinal Cord. 2018;56:106–16.
Stampacchia G, Rustici A, Bigazzi S, Gerini A, Tombini T, Mazzoleni S. Walking with a powered robotic exoskeleton: subjective experience, spasticity and pain in spinal cord injured persons. NeuroRehab. 2016;39:277–83.
Kressler J, Thomas CK, Field-Fote EC, Sanchez J, Widerström-Noga E, Cilien DC, et al. Understanding therapeutic benefits of overground bionic ambulation: exploratory case series in persons with chronic, complete spinal cord injury. Arch Phys Med Rehabil. 2015;21:122–32.
Kapandia N, Masani K, Craven BC, Giangregorio ML, Hitzig SL, Richards K, et al. A randomized trial of functional electrical stimulation for walking in incomplete spinal cord injury: effects on walking competency. J Spinal Cord Med. 2014;37:511–24.
Frotzler A, Coupaud S, Perrett C, Kakebeeke TH, Hunt KJ, Donaldson NN, et al. High-Volume FES-Cycling partially reverses bone loss in people with chronic spinal cord injury. Bone. 2008;43:169–76.
Yasar E, Yilmaz B, Goktepe S, Kesikburun S. The effect of functional electrical stimulation cycling on late functional improvement in patients with chronic incomplete spinal cord injury. Spinal Cord. 2015;53:866–9.
Dolbow DR, Gorgey AS, Ketchum JM, Gater DR. Home-based functional electrical stimulation cycling enhance quality of life in individuals with spinal cord injury. Top Spinal Cord Inj Rehabil. 2013;19:324–9.
Mazzoleni S, Stampacchia G, Gerini A, Tombini T, Carrozza MC. FES-cycling training in spinal cord injured patients. Conf Proc IEEE Eng Med Biol Soc. 2013;2013:5339–41.
Fisahn C, Aach M, Jansen O, Moisi M, Mayadev A, Pagarigan KT, et al. The effectiveness and safety of exoskeletons as assistive and rehabilitation devices in the treatment of neurologic gait disorders in patients with spinal cord injury: a systematic review. Glob Spine J. 2016;6:822–41.
Shackleton C, Evans R, Shamley D, West S, Albertus Y. Effectiveness of over-ground robotic locomotor training in improving walking performance, cardiovascular demands, secondary complications and user-satisfaction in individuals with spinal cord injuries: a systematic review. J Rehabil Med. 2019;51:723–33.
Morawietz C, Moffat F. Effects of locomotor training after incomplete spinal cord injury: a systematic review. Arch Phys Med Rehabil. 2013;94:2297–308.
Catz A, Itzkovich M, Steinberg F, Philo O, Ring H, Ronen J, et al. The Catz-Itzkovich SCIM: a revised version of the spinal cord independence measure. Disabil Rehabil. 2001;23:263–8.
Ditunno P, Ditunno J. Walking index for spinal cord injury (WISCI II): scale revision. Spinal Cord. 2001;39:654–6.
Van Hedel HJ, Wirz M, Dietz V. Assessing walking ability in subjects with spinal cord injury: validity and reliability of 3 walking tests. Arch Phys Med Rehabil. 2005;86:190–6.
Ferro S, Cecconi L, Bonavita J, Pagliacci MC, Biggeri A, Franceschini M. Incidence of traumatic spinal cord injury in Italy during 2013-2014: a popoulation-based study. Spinal Cord. 2017;55:1103–7.
Citterio A, Franceschini M, Spizzichino L, Reggio A, Rossi B, Stampacchia G, for the Gruppo Italiano Studio Epidemiologico Mielolesioni. Nontraumatic spinal cord injury: an Italian survey. Arch Phys Med Rehabil. 2004;85:1483–7.
Benson I, Hart K, Tussler D, Van Middendorp JJ. Lower-limb exoskeletons for individuals with chronic spinal cord injury: findings from a feasibility study. Clin Rehabil. 2016;30:73–84.
Louie DR, Eng JJ, Lam T, and Spinal Cord Injury Research Evidence (SCIRE) Research Team. Gait speed using powered robotic exoskeletons after spinal cord injury: a systematic review and correlational study. J NeuroEng Rehab. 2015;12:1–10.
Yang A, Asselin P, Knezevic S, Kornfeld S, Spungen AM. Assessment of in-hospital walking velocity and level of assistance in powered exoskeletons in persons with spinal cord injury. Top Spinal Cord Inj Rehabil. 2015;21:100–9.
Heinemann AW, Jayaraman A, Mummydisetty CK, Spraggins J, Pinto D, Charlifue S, et al. Experience of robotic exoskeleton use at four spinal cord injury model systems centers. JNPT. 2018;42:256–67.
Fornusek C, Davis GM, Russold MF. Pilot study of the effect of low-cadence functional electrical stimulation cycling after spinal cord injury on thigh girth and strength. Arch Phys Med Rehab. 2013;94:990–3.
Andrews B, Shippen J, Armegol M, Gibbons R, Holderbaum W, Harwin W. A design method for FES bone health therapy in SCI. Eur J Transl Myol. 2016;26:297–300.
Acknowledgements
The authors would like to thank Zach McKinney for his linguistic expertise in reviewing this paper.
Funding
Italian Health Ministry founded the project: “Clinical and healthcare strategies for improving quality of life in persons affected by SCI: Tuscany regional network and use of innovative technologies devices”. Code Project: RF-2011-02346770.
Author information
Authors and Affiliations
Contributions
GS was responsible of the study protocol, carried out patients’ selection, supervised clinical evaluations and treatments, supervised dataset production and analysed data, interpreted results, and drafted the paper. MO was responsible of dataset production, contributed to statistical analysis and contributed to drafted the paper. AR carried out patients’ physiotherapy evaluation and treatments, contributed to dataset production. CDA contributed to clinical evaluations. AG carried out clinical evaluations. SM contributed to design the study protocol, supervised statistical analysis, analysed the data, interpreted results and drafted the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study followed the guidelines of the Declaration of Helsinki. All applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research. All the necessary approvals were obtained, including local ethic committee (protocol number 410699).
Informed consent
All participants received oral, as well as written information about the study, before informed consent was obtained.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Stampacchia, G., Olivieri, M., Rustici, A. et al. Gait rehabilitation in persons with spinal cord injury using innovative technologies: an observational study. Spinal Cord 58, 988–997 (2020). https://doi.org/10.1038/s41393-020-0454-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41393-020-0454-2
This article is cited by
-
Functional electrical stimulation cycling exercise after spinal cord injury: a systematic review of health and fitness-related outcomes
Journal of NeuroEngineering and Rehabilitation (2021)