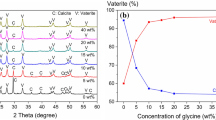
Abstract
Offsetting global warming requires rapid methods to sequester atmospheric carbon dioxide (CO2), a major greenhouse gas. For instance, mineral carbonation of gypsum from flue gas desulfurization (FGD) is a potential technology for CO2 sequestration, yet reaction conditions are not actually optimized to produce a recyclable product. Here, we transformed gypsum, CaSO4·2H2O, into vaterite, CaCO3, by sonication. We studied conversion, phase compositions and morphologies of carbonation products, and sulfate (SO42−) concentrations during carbonation. Results show that the conversion of CaSO4·2H2O increased from 60 to 98% with ultrasonic amplitude. Pure vaterite was obtained at 50% ultrasonic amplitude in 30 min. Low Ca2+/CO32− ratio, high concentration of SO42− and their synergistic effects appear as key factors to form vaterite. Overall, findings reveal the feasibility of gypsum carbonation to produce high-value vaterite by sonication.




Similar content being viewed by others
References
Alexopoulou M, Mystiridou E, Mouzakis D, Zaoutsos S, Fatouros DG, Bouropoulos N (2016) Preparation, characterization and in vitro assessment of ibuprofen loaded calcium phosphate/gypsum bone cements. Cryst Res Technol 51:41–48. https://doi.org/10.1002/crat.201500143
Bai H, Yeh AC (1997) Removal of CO2 greenhouse gas by ammonia scrubbing. Ind Eng Chem Res 36:2490–2493. https://doi.org/10.1021/ie960748j
Beuvier T, Calvignac B, Delcroix GJ-R, Tran MK, Kodjikian S, Delorme N, Bardeau J-F, Gibaud A, Boury F (2011) Synthesis of hollow vaterite CaCO3 microspheres in supercritical carbon dioxide medium. J Mater Chem 21:9757–9761. https://doi.org/10.1039/C1JM10770D
Böke H, Akkurt S, Özdemir S, Göktürk EH, Saltik ENC (2004) Quantification of CaCO3–CaSO3·0.5 H2O–CaSO4·2H2O mixtures by FTIR analysis and its ANN model. Mater Lett 58:723–726. https://doi.org/10.1016/j.matlet.2003.07.008
Bots P, Benning LG, Rodriguez-Blanco JD, Roncal-Herrero T, Shaw S (2012) Mechanistic insights into the crystallization of amorphous calcium carbonate (ACC). Cryst Growth Des 12:3806–3814. https://doi.org/10.1021/cg300676b
Chen H, Dou B, Song Y, Xu Y, Wang X, Zhang Y, Du X, Wang C, Zhang X, Tan C (2012) Studies on absorption and regeneration for CO2 capture by aqueous ammonia. Int J Greenh Gas Control 6:171–178. https://doi.org/10.1016/j.ijggc.2011.11.017
Cheng H, Wang X, Wang B, Zhao J, Liu Y, Cheng F (2017) Effect of ultrasound on the morphology of the CaCO3 precipitated from CaSO4–NH3–CO2–H2O system. J Cryst Growth 469:97–105. https://doi.org/10.1016/j.jcrysgro.2016.10.017
Choi K-M, Kuroda K (2012) Polymorph control of calcium carbonate on the surface of mesoporous silica. Cryst Growth Des 12:887–893. https://doi.org/10.1021/cg201314k
Costa G, Polettini A, Pomi R, Stramazzo A, Zingaretti D (2017) Energetic assessment of CO2 sequestration through slurry carbonation of steel slag: a factorial study. Greenh Gases 7:530–541. https://doi.org/10.1002/ghg.1659
Cuesta Mayorga I, Astilleros JM, Fernández-Díaz L (2019) Precipitation of CaCO3 polymorphs from aqueous solutions: the role of pH and sulphate groups. Minerals 9:178. https://doi.org/10.3390/min9030178
Dalvi SV, Dave RN (2009) Controlling particle size of a poorly water-soluble drug using ultrasound and stabilizers in antisolvent precipitation. Ind Eng Chem Res 48:7581–7593. https://doi.org/10.1021/ie900248f
Fernández-Díaz L, Fernández-González Á, Prieto M (2010) The role of sulfate groups in controlling CaCO3 polymorphism. Geochim Cosmochim Acta 74:6064–6076. https://doi.org/10.1016/j.gca.2010.08.010
Fuchigami K, Taguchi Y, Tanaka M (2009) Synthesis of calcium carbonate vaterite crystals and their effect on stabilization of suspension polymerization of MMA. Adv Powder Technol 20:74–79. https://doi.org/10.1016/j.apt.2008.10.003
Gopi S, Subramanian VK, Palanisamy K (2013) Aragonite-calcite-vaterite: a temperature influenced sequential polymorphic transformation of CaCO3 in the presence of DTPA. Mater Res Bull 48:1906–1912. https://doi.org/10.1016/j.materresbull.2013.01.048
Green DW, Bolland BJ, Kanczler JM, Lanham SA, Walsh D, Mann S, Oreffo RO (2009) Augmentation of skeletal tissue formation in impaction bone grafting using vaterite microsphere biocomposites. Biomaterials 30:1918–1927. https://doi.org/10.1016/j.biomaterials.2008.12.052
Guo B, Zhao T, Sha F, Zhang F, Li Q, Zhao J, Zhang J (2017) Synthesis of vaterite CaCO3 micro-spheres by carbide slag and a novel CO2-storage material. J CO2 Util 18:23–29. https://doi.org/10.1016/j.jcou.2017.01.004
Han YS, Hadiko G, Fuji M, Takahashi M (2006a) Crystallization and transformation of vaterite at controlled pH. J Cryst Growth 289:269–274. https://doi.org/10.1016/j.jcrysgro.2005.11.011
Han YS, Hadiko G, Fuji M, Takahashi M (2006b) Factors affecting the phase and morphology of CaCO3 prepared by a bubbling method. J Eur Ceram Soc 26:843–847. https://doi.org/10.1016/j.jeurceramsoc.2005.07.050
Hepburn C, Adlen E, Beddington J, Carter EA, Fuss S, Mac Dowell N, Minx JC, Smith P, Williams CK (2019) The technological and economic prospects for CO2 utilization and removal. Nature 575:87–97. https://doi.org/10.1038/s41586-019-1681-6
Hu YL, Zhang RL, Fang D (2019) Quaternary phosphonium cationic ionic liquid/porous metal–organic framework as an efficient catalytic system for cycloaddition of carbon dioxide into cyclic carbonates. Environ Chem Lett 17:501–508. https://doi.org/10.1007/s10311-018-0793-9
Huijgen WJJ, Comans RNJ, Witkamp GJ (2007) Cost evaluation of CO2 sequestration by aqueous mineral carbonation. Energy Convers Manag 48:1923–1935. https://doi.org/10.1016/j.enconman.2007.01.035
Kirchofer A, Becker A, Brandt A, Wilcox J (2013) CO2 mitigation potential of mineral carbonation with industrial alkalinity sources in the united states. Environ Sci Technol 47:7548–7554. https://doi.org/10.1021/es4003982
Kitamura M (2002) Controlling factor of polymorphism in crystallization process. J Cryst Growth 237:2205–2214
Kralj D, Brečević L, Kontrec J (1997) Vaterite growth and dissolution in aqueous solution III. Kinetics of transformation. J Cryst Growth 177:248–257. https://doi.org/10.1016/S0022-0248(96)01128-1
Lai Y, Chen L, Bao W, Ren Y, Gao Y, Yin Y, Zhao Y (2015) Glycine-mediated, selective preparation of monodisperse spherical vaterite calcium carbonate in various reaction systems. Cryst Growth Des 15:1194–1200. https://doi.org/10.1021/cg5015847
Lee MG, Jangwon RyuKimBang YNKWJH (2012) Mineral carbonation of flue gas desulfurization gypsum for CO2 sequestration. Energy 47:370–377. https://doi.org/10.1016/j.energy.2012.09.009
Lichtfouse E, Lichtfouse M, Jaffrezic A (2003) δ13C values of grasses as a novel indicator of pollution by fossil-fuel-derived greenhouse gas CO2 in urban areas. Environ Sci Technol 37:87–89. https://doi.org/10.1021/es025979y
Lichtfouse E, Lichtfouse M, Kashgarian M, Bol R (2005) 14C of grasses as an indicator of fossil fuel CO2 pollution. Environ Chem Lett 3:78–81. https://doi.org/10.1007/s10311-005-0100-4
Lu SQ, Lan PQ, Wu SF (2016) Preparation of nano-CaCO3 from phosphogypsum by gas–liquid–solid reaction for CO2 sorption. Ind Eng Chem Res 55:10172–10177. https://doi.org/10.1021/acs.iecr.6b02551
Lu JG, Li X, Zhao YX, Ma HL, Wang LF, Wang XY, Yu YF, Shen TY, Xu H, Zhang YT (2019) CO2 capture by ionic liquid membrane absorption for reduction of emissions of greenhouse gas. Environ Chem Lett 17:1031–1038. https://doi.org/10.1007/s10311-018-00822-4
Mantilaka M, Rajapakse R, Karunaratne D, Pitawala H (2014) Preparation of amorphous calcium carbonate nanoparticles from impure dolomitic marble with the aid of poly (acrylic acid) as a stabilizer. Adv Powder Technol 25:591–598. https://doi.org/10.1016/j.apt.2013.09.008
Olajire AA (2013) A review of mineral carbonation technology in sequestration of CO2. J Petrol Sci Eng 109:364–392. https://doi.org/10.1016/j.petrol.2013.03.013
Parakhonskiy BV, Haase A, Antolini R (2012) Sub-micrometer vaterite containers: synthesis, substance loading, and release. Angew Chem Int Edit 51:1195–1197. https://doi.org/10.1002/anie.201104316
Parakhonskiy BV, Foss C, Carletti E, Fedel M, Haase A, Motta A, Migliaresi C, Antolini R (2013) Tailored intracellular delivery via a crystal phase transition in 400 nm vaterite particles. Biomater Sci 1:1273–1281. https://doi.org/10.1039/C3BM60141B
Rodriguez-Blanco JD, Shaw S, Benning LG (2011) The kinetics and mechanisms of amorphous calcium carbonate (ACC) crystallization to calcite, via vaterite. Nanoscale 3:265–271. https://doi.org/10.1039/C0NR00589D
Sanna A, Uibu M, Caramanna G, Kuusik R, Maroto-Valer M (2014) A review of mineral carbonation technologies to sequester CO2. Chem Soc Rev 43:8049–8080. https://doi.org/10.1039/C4CS00035H
Shirsath S, Sonawane S, Saini D, Pandit A (2015) Continuous precipitation of calcium carbonate using sonochemical reactor. Ultrason Sonochem 24:132–139. https://doi.org/10.1016/j.ultsonch.2014.12.003
Song K, Jang YN, Kim W, Lee MG, Shin D, Bang JH, Jeon CW, Chae SC (2012) Precipitation of calcium carbonate during direct aqueous carbonation of flue gas desulfurization gypsum. Chem Eng J 213:251–258. https://doi.org/10.1016/j.cej.2012.10.010
Song K, Kim W, Bang JH, Park S, Jeon CW (2015) Polymorphs of pure calcium carbonate prepared by the mineral carbonation of flue gas desulfurization gypsum. Mater Des 83:308–313. https://doi.org/10.1016/j.matdes.2015.06.051
Spanos N, Koutsoukos PG (1998) The transformation of vaterite to calcite: effect of the conditions of the solutions in contact with the mineral phase. J Cryst Growth 191:783–790. https://doi.org/10.1016/S0022-0248(98)00385-6
Tamilselvi Dananjayan RR, Kandasamy P, Andimuthu R (2016) Direct mineral carbonation of coal fly ash for CO2 sequestration. J Clean Prod 112:4173–4182. https://doi.org/10.1016/j.jclepro.2015.05.145
Tan Y, Nookuea W, Li H, Thorin E, Yan J (2016) Property impacts on carbon capture and storage (CCS) processes: a review. Energy Convers Manag 118:204–222. https://doi.org/10.1016/j.enconman.2016.03.079
Tan W, Zhang Z, Li H, Li Y, Shen Z (2017) Carbonation of gypsum from wet flue gas desulfurization process: experiments and modeling. Environ Sci Pollut R 24:8602–8608. https://doi.org/10.1007/s11356-017-8480-0
Tolba E, Müller WE, El-Hady BMA, Neufurth M, Wurm F, Wang S, Schröder HC, Wang X (2016) High biocompatibility and improved osteogenic potential of amorphous calcium carbonate/vaterite. J Mater Chem B 4:376–386. https://doi.org/10.1039/C5TB02228B
Trushina DB, Bukreeva TV, Kovalchuk MV, Antipina MN (2014) CaCO3 vaterite microparticles for biomedical and personal care applications. Mater Sci Eng C 45:644–658. https://doi.org/10.1016/j.msec.2014.04.050
Wang A, Yang Y, Zhang X, Liu X, Cui W, Li J (2016) Gelatin-assisted synthesis of vaterite nanoparticles with higher surface area and porosity as anticancer drug containers in vitro. ChemPlusChem 81:194–201. https://doi.org/10.1002/cplu.201500515
Wang B, Pan Z, Cheng H, Chen Z, Cheng F (2018) High-yield synthesis of vaterite microparticles in gypsum suspension system via ultrasonic probe vibration/magnetic stirring. J Cryst Growth 492:122–131. https://doi.org/10.1016/j.jcrysgro.2018.02.021
Xu Y, Ma G, Wang M (2014) Fabrication and growth mechanism of pumpkin-shaped vaterite hierarchical structures. Cryst Growth Des 14:6166–6171. https://doi.org/10.1021/cg501477u
Zhang Z, Li Y, Zhang W, Wang J, Soltanian MR, Olabi AG (2018) Effectiveness of amino acid salt solutions in capturing CO2: a review. Renew Sustain Energy Rev 98:179–188. https://doi.org/10.1016/j.rser.2018.09.019
Zhao H, Li H, Bao W, Wang C, Li S, Lin W (2015) Experimental study of enhanced phosphogypsum carbonation with ammonia under increased CO2 pressure. J CO2 Util 11:10–19. https://doi.org/10.1016/j.jcou.2014.11.004
Acknowledgements
This work was supported by the National Key R&D Program of China (2018YFB0605804).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, B., Pan, Z., Cheng, H. et al. CO2 sequestration: high conversion of gypsum into CaCO3 by ultrasonic carbonation. Environ Chem Lett 18, 1369–1377 (2020). https://doi.org/10.1007/s10311-020-00997-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-020-00997-9