Abstract
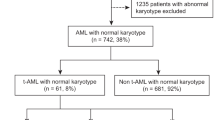
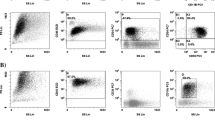
Terminal deoxynucleotidyl transferase (TdT) is expressed on precursor lymphoblastic neoplasms and some acute myeloid leukemia (AML) cells. The clinical impact of TdT expression on AML outcomes remains unclear. Here, we conducted a retrospective analysis to identify prognostic implications of TdT expression in AML with an intermediate-risk karyotype. Forty-eight cases of intermediate-risk AML were enrolled. TdT positivity was defined as expression on ≥ 10% of the gated cells. Of 48 cases, 12 (25%) were positive for TdT [median expression rate of TdT 0.9% (range 0–86.9%)]. No significant differences in patient characteristics or complete remission rate were observed between TdT-positive and TdT-negative cases. The probability of overall survival (OS) and event-free survival (EFS) at 1 year was not significantly different between TdT-positive and TdT-negative cases (OS: 58.3% vs. 65.2%, p = 0.32; EFS: 33.3% vs. 57.1%, p = 0.06). Relapse-free survival (RFS) probability at 1 year was significantly lower for TdT-positive than TdT-negative cases (10% vs. 71.3%, p = 0.002). Multivariate analyses revealed that TdT positivity was an independent significant adverse factor for RFS [hazard ratio: 3.309, 95% confidence interval: 1.334–8.209, p = 0.009]. Our results suggest that TdT expression is associated with increased risk of relapse in patients with intermediate-risk AML.




Similar content being viewed by others
References
Silverstone AE, Cantor H, Goldstein G, Baltimore D. Terminal deoxynucleotidyl transferase is found in prothymocytes. J Exp Med. 1976;144:543–8.
McGraw TP, Folds JD, Bollum FJ, Stass SA. Terminal deoxynucleotidyl transferase-positive acute myeloblastic leukemia. Am J Hematol. 1981;10:251–8.
Schachner J, Kantarjian H, Dalton W, McCredie K, Keating M, Freireich EJ. Cytogenetic association and prognostic significance of bone marrow blast cell terminal transferase in patients with acute myeloblastic leukemia. Leukemia. 1988;2:667–71.
Lee EJ, Yang J, Leavitt RD, Testa JR, Civin CI, Forrest A, et al. The significance of CD34 and TdT determinations in patients with untreated de novo acute myeloid leukemia. Leukemia. 1992;6:1203–9.
Huh YO, Smith TL, Collins P, Bueso-Ramos C, Albitar M, Kantarjian HM, et al. Terminal deoxynucleotidyl transferase expression in acute myelogenous leukemia and myelodysplasia as determined by flow cytometry. Leuk Lymphoma. 2000;37:319–31.
Chang H, Salma F, Yi QL, Patterson B, Brien B, Minden MD. Prognostic relevance of immunophenotyping in 379 patients with acute myeloid leukemia. Leuk Res. 2004;28:43–8.
Slovak ML, Kopecky KJ, Cassileth PA, Harrington DH, Theil KS, Mohamed A, et al. Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: a Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood. 2000;96:4075–83.
Koreth J, Schlenk R, Kopecky KJ, Honda S, Sierra J, Djulbegovic BJ, et al. Allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission: systematic review and meta-analysis of prospective clinical trials. JAMA. 2009;301:2349–61.
Takami A. Hematopoietic stem cell transplantation for acute myeloid leukemia. Int J Hematol. 2018;107:513–8.
Silva FP, Almeida I, Morolli B, Brouwer-Mandema G, Wessels H, Vossen R, et al. Genome wide molecular analysis of minimally differentiated acute myeloid leukemia. Haematologica. 2009;94:1546–54.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391–405.
O'Donnell MR, Tallman MS, Abboud CN, Altman JK, Appelbaum FR, Arber DA, et al. Acute myeloid leukemia, version 3 2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw. 2017;15:926–57.
Kanda Y. Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Silva FP, Swagemakers SM, Erpelinck-Verschueren C, Wouters BJ, Delwel R, Vrieling H, et al. Gene expression profiling of minimally differentiated acute myeloid leukemia: M0 is a distinct entity subdivided by RUNX1 mutation status. Blood. 2009;114:3001–7.
Greif PA, Konstandin NP, Metzeler KH, Herold T, Pasalic Z, Ksienzyk B, et al. RUNX1 mutations in cytogenetically normal acute myeloid leukemia are associated with a poor prognosis and up-regulation of lymphoid genes. Haematologica. 2012;97:1909–15.
Schnittger S, Dicker F, Kern W, Wendland N, Sundermann J, Alpermann T, et al. RUNX1 mutations are frequent in de novo AML with noncomplex karyotype and confer an unfavorable prognosis. Blood. 2011;117:2348–57.
Borrow J, Dyer SA, Akiki S, Griffiths MJ. Terminal deoxynucleotidyl transferase promotes acute myeloid leukemia by priming FLT3-ITD replication slippage. Blood. 2019;134:2281–90.
Borrow J, Dyer SA, Akiki S, Griffiths MJ. Molecular roulette: nucleophosmin mutations in AML are orchestrated through N-nucleotide addition by TdT. Blood. 2019;134:2291–303.
Author information
Authors and Affiliations
Contributions
MS and MO were responsible for the design of the study, interpretation of the data, and writing the manuscript. KI, KK, and TN treated the patients, collected the samples, and provided patient data. All other authors have contributed to data collection and interpretation, and critically reviewed the manuscript. All authors contributed to the revision of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Saburi, M., Ogata, M., Satou, T. et al. Prognostic implications of TdT expression in acute myeloid leukemia with an intermediate-risk karyotype. Int J Hematol 112, 17–23 (2020). https://doi.org/10.1007/s12185-020-02871-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-020-02871-4