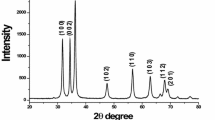
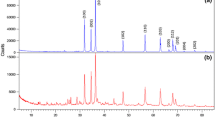
Abstract
The effects of ammonium polycarboxylate (AMP) on the dispersion of zinc oxide nanopowders were investigated in this study. Good dispersibility and stability in ZnO suspension with a dispersant content of 0.5 wt.% at pH 10 was observed by field emission scanning electron microscopy, and was confirmed by sedimentation experiments. The adsorption state of AMP on the surface of nano-sized ZnO powder was examined by Fourier transform infrared spectroscopy. For the ZnO nanopowders with various AMP amounts in the alkaline region, a maximum absolute zeta potential of 55.8 mV was measured at pH 10, which correlated well with the saturation amount of adsorbed AMP. Compacted varistor discs were prepared from the nano-origin ZnO powders before and after AMP absorption, showing sintered densities of 97.6% and 98.4%, respectively. The advantages of AMP were clearly seen in grain structuring, in addition to the enhanced densification and homogeneous microstructures for obtaining a high nonlinear coefficient.
Similar content being viewed by others
References
M. Zhang, H. Zhang, and L. Li, J. Alloys Compd. 744, 364 (2018).
M.H. Kathawala, K.W. Ng, and S.C.J. Loo, J. Nanopart. Res. 17, 263 (2015).
J. Song, L. Liu, and X.F. Wang, J. Mater. Chem. A 5, 13439 (2017).
J. He, X. Li, and Q. Zhu, Ceram. Int. 42, 9737 (2016).
YuG Trifonov, A.D. Shlyapin, AYu Omarov, and V.P. Tarasovskii, Refract. Ind. Ceram 56, 201 (2015).
X.L. Fu, H. Feng, and Z.J. Peng, Key Eng. Mater. 633, 308 (2014).
H.S. Woo, C.W. Na, and I.D. Kim, Nanotechnology 23, 245501 (2012).
S. Balachandran and M. Swaminathan, J. Phys. Chem. C 116, 26306 (2012).
M. Dorraj, A. Zakaria, and Y. Abdollahi, Sci. World J. 2014, 741034 (2014).
R.K. Sendi, A. Munshi, and S. Mahmud, Superlattice. Microst. 69, 212 (2014).
A.C. Malcolm, J.M. Parnis, and A.J. Vreugdenhil, J. Non-Cryst. Solids. 357, 1203 (2011).
S. Liufu, H. Xiao, and Y. Li, J. Colloid Interface Sci. 281, 155 (2005).
K. Gan, J. Xu, and Y. Lu, J. Eur. Ceram. Soc. 37, 891 (2017).
J. Zhigang, Z. Le, and Z. Fangzheng, Ceram. Int. 44, 16859 (2018).
J. Liu, X. Wu, and M. Wang, J. Electron. Mater. 48, 8024 (2019).
H. Guanghua, P. Zhidong, and W. Yanmin, Colloid Surface A 58, 591 (2018).
Y.W. Zhang, M. Tang, and X. Jin, Solid-State Sci. 5, 435 (2003).
Z. Guo, J. Xiong, and M. Yang, J. Alloys Compd. 493, 362 (2010).
T.T. Liu, M.H. Wang, and H. Su, J. Electron. Mater. 44, 10 (2015).
H. Foratirad, H.R. Baharvandi, and M.G. Maragheh, Int. J. Refract. Met. H. 56, 96 (2016).
T.H. Ho, S.J. Chang, and C.C. Li, Mater. Chem. Phys. 172, 1 (2016).
Y. Xiong, J. Hu, and Z. Shen, J. Eur. Ceram. Soc. 33, 2087 (2013).
Acknowledgments
This work was financially supported by Changzhou Science of Changzhou University Institute of Huaide, Nature Science Foundation of China (CE20180033).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, J., Wang, M., Wu, X. et al. Dispersion of ZnO Nanocrystals Using Ammonium Polycarboxylate (AMP) for the Fabrication of ZnO-Based Ceramics. J. Electron. Mater. 49, 3216–3224 (2020). https://doi.org/10.1007/s11664-020-08000-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-020-08000-5