Abstract
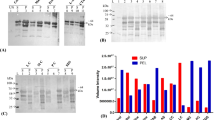
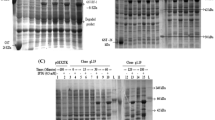
In this study, the cDNA of mature bovine interferon-α (BoIFN-α) gene was cloned and expressed in the Escherichia coli expression system. Total RNA was extracted from bovine liver cells and reverse transcribed into cDNA, and then BoIFN-α gene fragment was amplified with specific primers by PCR method before being ligated into the pET-32a (+) expression vector. Since the pET-32a (+) plasmid expresses the thioredoxin A (TrxA) tag protein, it can increase the solubility of the target protein. In order to express the target protein, the pET-32a-BoIFN-α recombinant plasmid was transformed into E. coli Rosetta (DE3) strain followed by IPTG induction. It was found that the recombinant fusion protein Trx-rBoIFN-α was finally expressed in a soluble form in the host cell, accounting for about 33.45% of the total cellular protein. The protein was purified by two-step chromatography, including Ni2+-chelating Sepharose affinity chromatography and DEAE anion exchange chromatography. The purity of the purified product reached 93.0%, and the cytopathic effects (CPE) inhibition method was adopted to determine the antiviral activity of the expressed fusion protein in vesicular stomatitis virus/ Madin-Darby bovine kidney cells titration system. The specific activity of the recombinant fusion protein Trx-rBoIFN-α was measured as (3.6 ± 0.25) × 106 U/mg. After cutting of the Trx tag protein by enterokinase digestion, the remaining solo protein was confirmed as rBoIFN-α protein by Western blot assay. These findings will enable us to produce a large number of bioactive rBoIFN-α protein for future application.





Similar content being viewed by others
REFERENCES
Tomasello, E., Pollet, E., Vu Manh, T.P., Uze, G., and Dalod, M., Front Immunol., 2014, vol. 5, p. 526.
Maher, S.G., Romero-Weaver, A.L., Scarzello, A.J., and Gamero, A.M., Curr. Med. Chem., 2007, vol. 14, no. 12, pp. 1279–1289.
Le Bon, A., Schiavoni, G., D’Agostino, G., Gresser, I., Belardelli, F., and Tough, D.F., Immunity, 2001, vol. 14, no. 4, pp. 461–470.
Sainz, B.Jr. and Halford, W.P., J. Virol., 2002, vol. 76, no. 22, pp. 11541–11550.
Yao, Q., Qian, P., Cao, Y., He, Y., Si, Y., Xu, Z., and Chen, H., Eur. Cytokine Netw., 2007, vol. 18, no. 2, pp. 71–77.
Moraes, M.P., de Los Santos, T., Koster, M., Turecek, T., Wang, H., Andreyev, V.G., and Grubman, M.J., J. Virol., 2007, vol. 81, no. 13, pp. 7124–7135.
Diaz-San Segundo, F., Weiss, M., Perez-Martin, E., Koster, M.J., Zhu, J., Grubman, M.J., and de los Santos, T., Virology, 2011, vol. 413, no. 2, pp. 283–292.
Gillespie, J., Scott, F., Geissinger, C., and Schiff, E., Zentralbl. Veterinarmed B, 1986, vol. 33, no. 10, pp. 771–776.
Gillespie, J.H., Scott, F.W., Geissinger, C.M., Czarniecki, C.W., and Scialli, V.T., J. Clin. Microbiol., 1986, vol. 24, no. 2, pp. 240–244.
Van Wyk, B., Snider, M., Scruten, E., Littel-van den Hurk, S.V., and Napper, S., Vet. Microbiol., 2016, vol. 195, pp. 104–114.
Chinsangaram, J., Piccone, M.E., and Grubman, M.J., J. Virol., 1999, vol. 73, no. 12, pp. 9891–9898.
Ohmann, H.B., Gilchrist, J.E., and Babiuk, L.A., J. Gen. Virol., 1984, vol. 65, part 9, pp. 1487–1495.
Shao, J.W., Cao, C., Bao, J., Liu, H.T., Peng, T.Q., Gao, M.C., and Wang, J.W., J. Interf. Cytok. Res., 2015, vol. 35, no. 3, pp. 168–175.
Akiyama, K., Sugii, S., and Hirota, Y., J. Vet. Med. Sci., 1993, vol. 55, no. 3, pp. 449–452.
Liu, Z.Q. and Yang, P.C., N. Am. J. Med. Sci., 2012, vol. 4, no. 12, pp. 651–655.
Mahmoudi, S., Abtahi, H., Bahador, A., Mosayebi, G., Salmanian, A.H., and Teymuri, M., Maedica (Buchar), 2012, vol. 7, pp. 241–246.
Kruger, N.J., Methods Mol. Biol., 1994,vol. 32, pp. 9–15.
Tu, Y.B., Wang, G., Wang, Y.Q., Chen, W.Y., Zhang, L., Liu, Y.G., et al., Microbiol. Res., 2016, vol. 191, pp. 12–18.
Chiba, K., Kawakami, K., Tohyama, K., Toxicol. In. Vitro., 1998, vol. 12, no.3, pp.251–258.
Liu, K., Liao, X., Zhou, B., Yao, H., Fan, S., Chen, P., and Miao, D., Res. Vet. Sci., 2013, vol. 95, no. 3, pp. 950–956.
Reed, L. and Muench, H., Am. J. Hyg., 1938, vol. 27, pp. 493–497.
Hou, F., Liu, K., Shen, T., Zhou, B., Cao, R., Li, P., and Chen, P., Res. Vet. Sci., 2011, vol. 91, no. 3, pp. e73–e79.
Ruttanapumma, R., Nakamura, M., and Takehara, K., J. Vet. Med. Sci., 2005, vol. 67, no. 1, pp. 25–28.
Marcus, P.I., van der Heide, L., and Sekellick, M.J., J. Interf. Cytok. Res., 1999, vol. 19, no. 8, pp. 881–885.
Schultz, U., Rinderle, C., Sekellick, M.J., Marcus, P.I., and Staeheli, P., Eur. J. Biochem., 1995, vol. 229, no. 1, pp. 73–76.
Chowdhury, A., Shah, S., Babu, S., Rai, R.R., Prasad, V.G., Rao, R., et al., Trop Gastroenterol., 2005, vol. 26, no. 2, pp. 70–72.
Zhou, X., Song, Z., Liu, X., Jia, F., and Wang, Y., Appl. Biochem. Biotechnol., 2011, vol. 163, no. 8, pp. 981–993.
Charlton, A. and Zachariou, M., Methods Mol. Biol., 2008, vol. 421, pp. 137–149.
Neves, F.O., Ho, P.L., Raw, I., Pereira, C.A., Moreira, C., and Nascimento, A.L., Protein Expr. Purif., 2004, vol. 35, no. 2, pp. 353–359.
Manual of Clinical Laboratory Immunology, Rose N.R, Friedman F.H., Fahey, J.L., Eds., 1986, Washington, DC: American Society for Microbiology, pp. 295–299.
Rubinstein, S., Familletti, P.C., and Pestka, S., J. Virol., 1981, vol. 37, no. 2, pp. 755–758.
Hawkins, M.J., Borden, E.C., Merritt, J.A., Edwards, B.S., Ball, L.A., Grossbard, E., and Simon, K.J., J. Clin. Oncol., 1984, vol. 2, no. 3, pp. 221–226.
McManus, N.H., Appl. Environ. Microbiol., 1976, vol. 31, no. 1, pp. 35–38.
Armstrong, J.A., Methods Enzymol., 1981, vol. 78, Pt. A, pp. 381–387.
Samuel, C.E., Clin. Microbiol. Rev., 2001, vol. 14, no. 4, pp. 778–809.
Zhao, J., Yu, H.Y., Zhao, Y., Li, F.H., Zhou, W., Xia, B.B., et al., Prep. Biochem. Biotechnol., 2019, vol. 49, no.2, pp.192–201.
ACKNOWLEDGMENTS
The authors are grateful to all the Research Staff in Wuhu Interferon Bio-products Industry Research Institute Co., Ltd (Wuhu, Anhui, P.R. China) for technical assistance.
Funding
This research was funded by the programs from the National key R&D Program of China (Grant nos. 2017YFD0500906 and 2017YFD0501000), the Natural Science Foundation of Anhui Province (Grant no. 1808085MC75), Scientific research activities of postdoctoral researchers in Anhui (Grant no. 2017B194).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Yu, HY., Liu, J., He, ZY. et al. Soluble Expression, Rapid Purification and Antiviral Activity of Recimbinant Bovine Interferon-α in Escherichia coli. Appl Biochem Microbiol 56, 154–163 (2020). https://doi.org/10.1134/S0003683820020143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683820020143