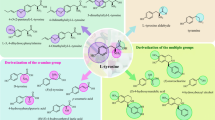
Abstract
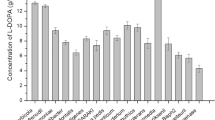
Tyrosine phenol-lyase (TPL) is a valuable and cost-effective biocatalyst for the biosynthesis of L-tyrosine and its derivatives, which are valuable intermediates in the pharmaceutical industry. A TPL from Morganella morganii (Mm-TPL) was overexpressed in Escherichia coli and characterized. Mm-TPL was determined as a homotetramer with molecular weight of 52 kDa per subunit. Its optimal temperature and pH for β-elimination of L-tyrosine were 45 °C and pH 8.5, respectively. Mm-TPL manifested strict substrate specificity for the reverse reaction of β-elimination and ortho- and meta-substituted phenols with small steric size were preferred substrates. The enzyme showed excellent catalytic performance for synthesis of L-tyrosine, 3-fluoro-L-tyrosine, and L-DOPA with a yield of 98.1%, 95.1%, and 87.2%, respectively. Furthermore, the fed-batch bioprocess displayed space-time yields of 9.6 g L−1 h−1 for L-tyrosine and 4.2 g L−1 h−1 for 3-fluoro-L-tyrosine with a yield of 67.4 g L−1 and 29.5 g L−1, respectively. These results demonstrated the great potential of Mm-TPL for industrial application.







Similar content being viewed by others
References
Milic, D., Demidkina, T. V., Faleev, N. G., Phillips, R. S., Matkovic-Calogovic, D., & Antson, A. A. (2011). Crystallographic snapshots of tyrosine phenol-lyase show that substrate strain plays a role in C-C bond cleavage. Journal of the American Chemical Society, 133(41), 16468–16476.
Yamada, H., & Kumagai, H. (1975). Synthesis of L-tyrosine-related amino acids by beta-tyrosinase. Advances in Applied Microbiology, 19, 249–288.
Phillips, R. S., Chen, H. Y., & Faleev, N. G. (2006). Aminoacrylate intermediates in the reaction of Citrobacter freundii tyrosine phenol-lyase. Biochemistry, 45(31), 9575–9583.
Santos, C. N. S., & Stephanopoulos, G. (2007). Melanin-based high-throughput screen for L-tyrosine production in Escherichia coli. Applied and Environmental Microbiology, 74(4), 1190–1197.
Santos, C. N. S., Xiao, W. H., & Stephanopoulos, G. (2012). Rational, combinatorial, and genomic approaches for engineering L-tyrosine production in Escherichia coli. Proceedings of the National Academy of Sciences, 109(34), 13538–13543.
Patil, S. A., Apine, O. A., Surwase, S. N., & Jadhav, J. P. (2013). Biological sources of L-DOPA: an alternative approach. Advances in Parkinson's Disease, 02(03), 81–87.
Monclus, M., Masson, C., & Luxen, A. (1995). Asymmetric synthesis of fluorinated L-tyrosine and meta-L-tyrosines. Journal of Fluorine Chemistry, 70, 39–43.
Reid, P. J., Loftus, C., Beeson, C. C., & Study, U. V. r. R. (2003). Evaluating the potential of fluorinated tyrosines as spectroscopic probes of local protein environments: a UV resonance Raman study. Biochemistry, 42(8), 2441–2448.
Ikeda, M. (2002). Amino acid production processes. Advances in Biochemical Engineering/Biotechnology, 79, 1–35.
Noisier, A. F., Harris, C. S., & Brimble, M. A. (2013). Novel preparation of chiral alpha-amino acids using the Mitsunobu-Tsunoda reaction. Chemical Communications, 49(70), 7744–7746.
Juminaga, D., Baidoo, E. E. K., Redding-Johanson, A. M., Batth, T. S., Burd, H., Mukhopadhyay, A., Petzold, C. J., & Keasling, J. D. (2012). Modular engineering of L-tyrosine production in Escherichia coli. Applied and Environmental Microbiology, 78(1), 89–98.
Xue, Y. P., Cao, C. H., & Zheng, Y. G. (2018). Enzymatic asymmetric synthesis of chiral amino acids. Chemical Society Reviews, 47(4), 1516–1561.
Milic, D., Matkovic-Calogovic, D., Demidkina, T. V., Kulikova, V. V., Sinitzina, N. I., & Antson, A. A. (2006). Structures of apo- and holo-tyrosine phenol-lyase reveal a catalytically critical closed conformation and suggest a mechanism for activation by K+ ions. Biochemistry, 45(24), 7544–7552.
Ogawa, J., & Shimizu, S. (2002). Industrial microbial enzymes: their discovery by screening and use in large-scale production of useful chemicals in Japan. Current Opinion in Biotechnology, 13(4), 367–375.
Tang, X. L., Suo, H., Wang, Z. C., Zheng, R. C., & Zheng, Y. G. (2018). Process development for efficient biosynthesis of L-DOPA with recombinant Escherichia coli harboring tyrosine phenol lyase from Fusobacterium nucleatum. Bioprocess and Biosystems Engineering, 41(9), 1347–1354.
Kim, D. Y., Rha, E., Choi, S. L., Song, J. J., Hong, S. P., Sung, M. H., & Lee, S. G. (2007). Development of bioreactor system for L-tyrosine synthesis using thermostable tyrosine phenol-lyase. Journal of Microbiology and Biotechnology, 17(1), 116–122.
Lozinsky, V. I., Faleev, N. G., Zubov, A. L., Ruvinov, S. B., Antonova, T. V., Vainerman, E. S., Belikov, V. M., & Rogozhin, S. V. (1989). Use of PVA-cryogel entrapped Citrobacter intermedius cells for continuous production of 3-fluoro-L-tyrosine. Biotechnology Letters, 11(1), 43–48.
Zhang, H. J., Lu, Y., Wu, S. P., Wei, Y., Liu, Q., Liu, J. Z., & Jiao, Q. C. (2016). Two-step enzymatic synthesis of tyramine from raw pyruvate fermentation broth. Journal of Molecular Catalysis B: Enzymatic, 124, 38–44.
Kumagai, H., Kashima, N., & Yamada, H. (1970). Racemization of D- or L-alanine by crystalline tyrosine phenol-lyase from Eshherichia intermedia. Biochemical and Biophysical Research Communications, 39(5), 796–801.
Chandel, M., & Azmi, W. (2013). Purification and characterization of tyrosine phenol lyase from Citrobacter freundii. Applied Biochemistry and Biotechnology, 171(8), 2040–2052.
Kumagai, H., Kashima, N., Torii, H., Yamada, H., Enei, H., & Okumuea, S. (1971). Purification, crystallization and properties of tyrosine phenol lyase from Erwinia herbicola. Agricultural and Biological Chemistry, 36(3), 472–482.
Rhee, S. K., Lee, S. G., Hong, S. P., Choi, Y. H., Park, J. H., Kim, C. J., & Sung, M. H. (2000). A novel microbial interaction: obligate commensalism between a new gram-negative thermophile and a thermophilic Bacillus strain. Extremophiles, 4(3), 131–136.
Zheng, R. C., Tang, X. L., Suo, H., Feng, L. L., Liu, X., Yang, J., & Zheng, Y. G. (2018). Biochemical characterization of a novel tyrosine phenol-lyase from Fusobacterium nucleatum for highly efficient biosynthesis of l-DOPA. Enzyme and Microbial Technology, 112, 88–93.
Wynands, B., Lenzen, C., Otto, M., Koch, F., Blank, L. M., & Wierckx, N. (2018). Metabolic engineering of Pseudomonas taiwanensis VLB120 with minimal genomic modifications for high-yield phenol production. Metabolic Engineering, 47, 121–133.
Carman, G. M., & Levin, R. E. (1977). Partial purification and some properties of tyrosine phenol-lyase from Aeromonas phenologenes ATCC 29063. Applied and Environmental Microbiology, 33(1), 192–198.
Brot, N., Smit, Z., & Weissbach, H. (1965). Conversion of l-tyrosine to phenol by Clostridium tetanomorphum. Archives of Biochemistry and Biophysics, 112(1), 1–6.
Enei, H., Nakazawa, H., Okumura, S., & Yamada, H. (1973). Synthesis of L-tyrosine or 3, 4-dihydroxyphenyl-L-alanine from pyruvic acid, ammonia and phenol or pyrocatechol. Agricultural and Biological Chemistry, 37(4), 725–735.
Milic, D., Demidkina, T. V., Faleev, N. G., Matkovic-Calogovic, D., & Antson, A. A. (2008). Insights into the catalytic mechanism of tyrosine phenol-lyase from X-ray structures of quinonoid intermediates. The Journal of Biological Chemistry, 283(43), 29206–29214.
Faleev, N. G., Axenova, O. V., Demidkina, T. V., & Phillips, R. S. (2003). The role of acidic dissociation of substrate’s phenol group in the mechanism of tyrosine phenol-lyase. Biochimica et Biophysica Acta, 1647(1-2), 260–265.
Lee, S. G., Hong, S. P., Kwak, M. S., Esaki, N., & Sung, M. H. (1999). Characterization of thermostable tyrosine phenol-lyase from an obligatory symbiotic thermophile, Symbiobacterium sp. SC-1. Journal of Biochemistry and Molecular Biology, 32(5), 480–485.
Kumagai, H., Yamada, H., Matsui, H., Ohkishi, H., & Ogata, K. (1970). Tyrosine phenol lyase. I. Purification, crystallization, and properties. The Journal of Biological Chemistry, 245(7), 1767–1772.
Miagkikh, I. V., & Demidkina, T. V. (1985). Effect of monovalent cations on the catalytic and spectral properties of tyrosine-phenol-lyase from Citrobacter intermedius. Molecular Biology, 19(3), 671–678.
Sundararaju, B., Chen, H., Shilcutt, S., & Phillips, R. S. (2000). The role of glutamic acid-69 in the activation of Citrobacter freundii tyrosine phenol-lyase by monovalent cations. Biochemistry, 39(29), 8546–8555.
Nagasawa, T., Utagawa, T., Goto, J., Kim, C. J., Tani, Y., Kumagai, H., & Yamada, H. (1981). Syntheses of L-tyrosine-related amino acids by tyrosine phenol-lyase of Citrobacter intermedius. European Journal of Biochemistry, 117(1), 33–40.
Hay, P. J., & Shavitt, I. (1974). Ab initioconfiguration interaction studies of the π-electron states of benzene. The Journal of Chemical Physics, 60(7), 2865–2877.
Skalický, T., Chollet, C., Pasquier, N., & Allan, M. (2002). Properties of the π* and σ* states of the chlorobenzene anion determined by electron impact spectroscopy. Physical Chemistry Chemical Physics, 4(15), 3583–3590.
Phillips, R. S., Von Tersch, R. L., & Secundo, F. (1997). Effects of tyrosine ring fluorination on rates and equilibria of formation of intermediates in the reactions of carbon-carbon lyases. European Journal of Biochemistry, 244(2), 658–663.
Tina, L. E., Santos, C. N. S., & Stephanopoulos, G. (2007). Perspectives of biotechnological production of L-tyrosine and its applications. Applied Microbiology and Biotechnology, 77(4), 751–762.
Funding
This study was financially supported by the National Natural Science Foundation of China (No. 31900912) and Natural Science Foundation of Zhejiang Province (LQ17B06004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhu, HQ., Tang, XL., Zheng, RC. et al. Purification and Biochemical Characterization of a Tyrosine Phenol-lyase from Morganella morganii. Appl Biochem Biotechnol 192, 71–84 (2020). https://doi.org/10.1007/s12010-020-03301-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-020-03301-1