Key message
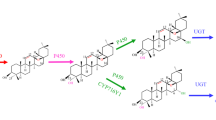
We employed both metabolomic and transcriptomic approaches to explore the accumulation patterns of physalins, flavonoids and chlorogenic acid in Physalis angulata and revealed the genes associated with the biosynthesis of bioactive compounds under methyl-jasmonate (MeJA) treatment.
Abstract
Physalis angulata L. is an annual Solanaceae plant with a number of medicinally active compounds. Despite the potential pharmacological benefits of P. angulata, the scarce genomic information regarding this plant has limited the studies on the mechanisms of bioactive compound biosynthesis. To facilitate the basic understanding of the main chemical constituent biosynthesis pathways, we performed both metabolomic and transcriptomic approaches to reveal the genes associated with the biosynthesis of bioactive compounds under methyl-jasmonate (MeJA) treatment. Untargeted metabolome analysis showed that most physalins, flavonoids and chlorogenic acid were significantly upregulated. Targeted HPLC–MS/MS analysis confirmed variations in the contents of two important representative steroid derivatives (physalins B and G), total flavonoids, neochlorogenic acid, and chlorogenic acid between MeJA-treated plants and controls. Transcript levels of a few steroid biosynthesis-, flavonoid biosynthesis-, and chlorogenic acid biosynthesis-related genes were upregulated, providing a potential explanation for MeJA-induced active ingredient synthesis in P. angulata. Systematic correlation analysis identified a number of novel candidate genes associated with bioactive compound biosynthesis. These results may help to elucidate the regulatory mechanism underlying MeJA-induced active compound accumulation and provide several valuable candidate genes for further functional study.







Similar content being viewed by others
References
Adamski J (2015) Perspectives in steroid research. J Steroid Biochem Mol Biol 153:1–2
Auer CA (2002) Discoveries and dilemmas concerning cytokinin metabolism. J Plant Growth Regul 21:24–31
Azam SS, Abro A, Raza S, Saroosh A (2014) Structure and dynamics studies of sterol 24-C-methyltransferase with mechanism based inactivators for the disruption of ergosterol biosynthesis. Mol Biol Rep 41:4279–4293
Buchfink B, Xie C, Huson DH (2014) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12:59
Chae HS, Kieber JJ (2005) Eto Brute? Role of ACS turnover in regulating ethylene biosynthesis. Trends Plant Sci 10:291–296
Chen LX, Xia GY, Liu QY, Xie YY, Qiu F (2014) Chemical constituents from the calyces of Physalis alkekengi var. franchetii. Biochem Syst Ecol 54:31–35
Chen M, Zhu WJ, You X, Liu YD, Kaleri GM, Yang Q (2015a) Isolation and characterization of a chalcone isomerase gene promoter from potato cultivars. Genet Mol Res 14:18872–18885
Chen RB, Li Q, Tan HX, Chen JF, Xiao Y, Ma RF, Gao SH, Zerbe P, Chen W, Zhang L (2015b) Gene-to-metabolite network for biosynthesis of lignans in MeJA-elicited Isatis indigotica hairy root cultures. Front Plant Sci 6:952
Chen RB, Chen XH, Zhu TT, Liu JH, Xiang X, Yu J, Tan HX, Gao SH, Li Q, Fang YC, Chen WS, Zhang L, Huang BK (2018) Integrated transcript and metabolite profiles reveal that EbCHI plays an important role in scutellarin accumulation in Erigeron breviscapus hairy roots. Front Plant Sci 9:789
Ciura J, Szeliga M, Grzesik M, Tyrka M (2018) Changes in fenugreek transcriptome induced by methyl jasmonate and steroid precursors revealed by RNA-Seq. Genomics 110:267–276
Corey EJ, Matsuda SP, Bartel B (1993) Isolation of an Arabidopsis thaliana gene encoding cycloartenol synthase by functional expression in a yeast mutant lacking lanosterol synthase by the use of a chromatographic screen. Proc Natl Acad Sci USA 90:11628
Dao TTH, Linthorst HJM, Verpoorte R (2011) Chalcone synthase and its functions in plant resistance. Phytochem Rev: Proc Phytochem Soc Europe 10:397–412
Dunn WB, Broadhurst D, Begley P, Zelena E, Francis-McIntyre S, Anderson N, Brown M, Knowles JD, Halsall A, Haselden JN, Nicholls AW, Wilson ID, Kell DB, Goodacre R, The Human Serum Metabolome Consortium (2011) Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat Protoc 6:1060
Enciso-Rodriguez FE, Gonzalez C, Rodriguez EA, Lopez CE, Landsman D, Barrero LS, Marino-Ramirez L (2013) Identification of immunity related genes to study the Physalis peruviana–Fusarium oxysporum pathosystem. PLoS ONE 8:e68500
Fukushima A, Nakamura M, Suzuki H, Yamazaki M, Knoch E, Mori T, Umemoto N, Morita M, Hirai G, Sodeoka M, Saito K (2016) Comparative characterization of the leaf tissue of Physalis alkekengi and Physalis peruviana using RNA-seq and metabolite profiling. Front Plant Sci 7:1883
Garzon-Martinez GA, Zhu ZI, Landsman D, Barrero LS, Marino-Ramirez L (2012) The Physalis peruviana leaf transcriptome: assembly, annotation and gene model prediction. BMC Genom 13:151
Gas-Pascual E, Simonovik B, Schaller H, Bach TJ (2015) Inhibition of cycloartenol synthase (CAS) function in tobacco BY-2 cells. Lipids 50:761–772
Godoy JA, Lunar R, Torres-Schumann S, Moreno J, Rodrigo RM, Pintor-Toro JA (1994) Expression, tissue distribution and subcellular localization of dehydrin TAS14 in salt-stressed tomato plants. Plant Mol Biol 26:1921–1934
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, Adiconis X, Fan L, Raychowdhury R, Zeng Q, Chen Z, Mauceli E, Hacohen N, Gnirke A, Rhind N, di Palma F, Birren BW, Nusbaum C, Lindblad-Toh K, Friedman N, Regev A (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644
Guimaraes ET, Lima MS, Santos LA, Ribeiro IM, Tomassini TB, Ribeiro dos Santos R, dos Santos WL, Soares MB (2009) Activity of physalins purified from Physalis angulata in in vitro and in vivo models of cutaneous leishmaniasis. J Antimicrob Chemother 64:84–87
Han JY, In JG, Kwon YS, Choi YE (2010) Regulation of ginsenoside and phytosterol biosynthesis by RNA interferences of squalene epoxidase gene in Panax ginseng. Phytochemistry 71:36–46
Ho TT, Lee JD, Jeong CS, Paek KY, Park SY (2018) Improvement of biosynthesis and accumulation of bioactive compounds by elicitation in adventitious root cultures of Polygonum multiflorum. Appl Microbiol Biotechnol 102:199–209
Ismail N, Alam M (2001) A novel cytotoxic flavonoid glycoside from Physalis angulata. Fitoterapia 72:676–679
Jin ML, Lee WM, Kim OT (2017) Two cycloartenol synthases for phytosterol biosynthesis in Polygala tenuifolia Willd. Int J Mol Sci 18:2426
Kang N, Kang N, Jian JF, Cao SJ, Zhang Q, Mao YW, Huang YY, Peng YF, Qiu F, Gao XM (2016) Physalin A induces G2/M phase cell cycle arrest in human non-small cell lung cancer cells: involvement of the p38 MAPK/ROS pathway. Mol Cell Biochem 415:145–155
Kranjc E, Albreht A, Vovk I, Glavnik V (2017) High performance thin-layer chromatography-mass spectrometry enables reliable analysis of physalins in different plant parts of Physalis alkekengi L. J Chromatogr A 1526:137–150
Li AL, Chen BJ, Li GH, Zhou MX, Li YR, Ren DM, Lou HX, Wang XN, Shen T (2018) Physalis alkekengi L. var. franchetii (Mast.) Makino: an ethnomedical, phytochemical and pharmacological review. J Ethnopharmacol 210:260–274
Liu J, Liu Y, Wang Y, Zhang ZH, Zu YG, Efferth T, Tang ZH (2016) The combined effects of ethylene and MeJA on metabolic profiling of phenolic compounds in Catharanthus roseus revealed by metabolomics analysis. Front Physiol 7:217
Liu XG, Bian J, Li DQ, Liu CF, Xu SS, Zhang GL, Zhang LX, Gao PY (2019) Structural features, antioxidant and acetylcholinesterase inhibitory activities of polysaccharides from stem of Physalis alkekengi L. Ind Crops Prod 129:654–661
Llano SM, Munoz-Jimenez AM, Jimenez-Cartagena C, Londono-Londono J, Medina S (2018) Untargeted metabolomics reveals specific withanolides and fatty acyl glycoside as tentative metabolites to differentiate organic and conventional Physalis peruviana fruits. Food Chem 244:120–127
Lusakibanza M, Mesia G, Tona G, Karemere S, Lukuka A, Tits M, Angenot L, Frederich M (2010) In vitro and in vivo antimalarial and cytotoxic activity of five plants used in congolese traditional medicine. J Ethnopharmacol 129:398–402
Ma DM, Wang Z, Wang L, Alejos-Gonzales F, Sun MA, Xie DY (2015) A genome-wide scenario of terpene pathways in self-pollinated Artemisia annua. Mol Plant 8:1580–1598
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J 2011(17):3
Maruenda H, Cabrera R, Canari-Chumpitaz C, Lopez JM, Toubiana D (2018) NMR-based metabolic study of fruits of Physalis peruviana L. grown in eight different Peruvian ecosystems. Food Chem 262:94–101
Matsuura T, Kawai M, Nakashima R, Butsugan Y (1969) Bitter principles of Physalis alkekengi var. Francheti: structure of physalin a. Tetrahedron Lett 10:1083–1086
Mirjalili MH, Moyano E, Bonfill M, Cusido RM, Palazon J (2009) Steroidal lactones from Withania somnifera, an ancient plant for novel medicine. Molecules 14:2373–2393
Munkert J, Ernst M, Muller-Uri F, Kreis W (2014) Identification and stress-induced expression of three 3beta-hydroxysteroid dehydrogenases from Erysimum crepidifolium Rchb. and their putative role in cardenolide biosynthesis. Phytochemistry 100:26–33
Nabavi SM, Samec D, Tomczyk M, Milella L, Russo D, Habtemariam S, Suntar I, Rastrelli L, Daglia M, Xiao J, Giampieri F, Battino M, Sobarzo-Sanchez E, Nabavi SF, Yousefi B, Jeandet P, Xu S, Shirooie S (2020) Flavonoid biosynthetic pathways in plants: versatile targets for metabolic engineering. Biotechnol Adv 38:107316
Nakashima T, Inoue T, Oka A, Nishino T, Osumi T, Hata S (1995) Cloning, expression, and characterization of cDNAs encoding Arabidopsis thaliana squalene synthase. Proc Natl Acad Sci USA 92:2328–2332
Phimchan P, Chanthai S, Bosland PW, Techawongstien S (2014) Enzymatic changes in phenylalanine ammonia-lyase, cinnamic-4-hydroxylase, capsaicin synthase, and peroxidase activities in capsicum under drought stress. J Agric Food Chem 62:7057–7062
Pinto LA, Meira CS, Villarreal CF, Vannier-Santos MA, de Souza CV, Ribeiro IM, Tomassini TC, Galvao-Castro B, Soares MB, Grassi MF (2016) Physalin F, a seco-steroid from Physalis angulata L., has immunosuppressive activity in peripheral blood mononuclear cells from patients with HTLV1-associated myelopathy. Biomed Pharmacother 79:129–134
Ryder NS (1991) Squalene epoxidase as a target for the allylamines. Biochem Soc Trans 19:774–777
Sawai S, Akashi T, Sakurai N, Suzuki H, Shibata D, Ayabe S, Aoki T (2006) Plant lanosterol synthase: divergence of the sterol and triterpene biosynthetic pathways in eukaryotes. Plant Cell Physiol 47:673–677
Schaller H (2004) New aspects of sterol biosynthesis in growth and development of higher plants. Plant Physiol Biochem 42:465–476
Flora compilation committee of Chinese academy of science (2008) Flora of China. Science Press, Beijing, p 50
Shackleton CH (2012) Role of a disordered steroid metabolome in the elucidation of sterol and steroid biosynthesis. Lipids 47:1–12
Shih CH, Chu H, Tang LK, Sakamoto W, Maekawa M, Chu IK, Wang M, Lo C (2008) Functional characterization of key structural genes in rice flavonoid biosynthesis. Planta 228:1043–1054
Simbaqueba J, Catanzariti AM, Gonzalez C, Jones DA (2018) Evidence for horizontal gene transfer and separation of effector recognition from effector function revealed by analysis of effector genes shared between cape gooseberry- and tomato-infecting formae speciales of Fusarium oxysporum. Mol Plant Pathol 19:2302–2318
Sonnante G, D'Amore R, Blanco E, Pierri CL, De Palma M, Luo J, Tucci M, Martin C (2010) Novel hydroxycinnamoyl-coenzyme A quinate transferase genes from artichoke are involved in the synthesis of chlorogenic acid. Plant Physiol 153:1224–1238
Suzuki M, Xiang T, Ohyama K, Seki H, Saito K, Muranaka T, Hayashi H, Katsube Y, Kushiro T, Shibuya M, Ebizuka Y (2006) Lanosterol synthase in dicotyledonous plants. Plant Cell Physiol 47:565–571
Tu YH, Liu F, Guo DD, Fan LJ, Zhu ZX, Xue YR, Gao Y, Guo ML (2016) Molecular characterization of flavanone 3-hydroxylase gene and flavonoid accumulation in two chemotyped safflower lines in response to methyl jasmonate stimulation. BMC Plant Biol 16:132
Uno T, Nakano R, Kitagawa R, Okada M, Kanamaru K, Takenaka S, Uno Y, Imaishi H (2018) Metabolism of steroids by cytochrome P450 2C9 variants. Biopharm Drug Dispos 39:371–377
Vieira AT, Pinho V, Lepsch LB, Scavone C, Ribeiro IM, Tomassini T, Ribeiro-dos-Santos R, Soares MB, Teixeira MM, Souza DG (2005) Mechanisms of the anti-inflammatory effects of the natural secosteroids physalins in a model of intestinal ischaemia and reperfusion injury. Br J Pharmacol 146:244–251
Vranova E, Hirsch-Hoffmann M, Gruissem W (2011) AtIPD: a curated database of Arabidopsis isoprenoid pathway models and genes for isoprenoid network analysis. Plant Physiol 156:1655–1660
Wang AQ, Wang SP, Zhou FY, Li P, Wang YT, Gan LS, Lin LG (2018a) Physalin B induces cell cycle arrest and triggers apoptosis in breast cancer cells through modulating p53-dependent apoptotic pathway. Biomed Pharmacother 101:334–341
Wang ZB, Yu QB, Shen WX, El Mohtar CA, Zhao XC, Gmitter FG Jr (2018b) Functional study of CHS gene family members in citrus revealed a novel CHS gene affecting the production of flavonoids. BMC Plant Biol 18:189
Wen B, Mei ZL, Zeng CW, Liu SQ (2017) metaX: a flexible and comprehensive software for processing metabolomics data. BMC Bioinform 18:183
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Yang YK, Xie SD, Xu WX, Nian Y, Liu XL, Peng XR, Ding ZT, Qiu MH (2016) Six new physalins from Physalis alkekengi var. franchetii and their cytotoxicity and antibacterial activity. Fitoterapia 112:144–152
Yang YJ, Yi L, Wang Q, Xie BB, Dong Y, Sha CW (2017) Anti-inflammatory effects of physalin E from Physalis angulata on lipopolysaccharide-stimulated RAW 264.7 cells through inhibition of NF-kappaB pathway. Immunopharm Immunot 39:74–79
Yu YJ, Sun LJ, Ma L, Li JY, Hu LH, Liu JW (2010) Investigation of the immunosuppressive activity of Physalin H on T lymphocytes. Int Immunopharmacol 10:290–297
Zhan XR, Liao XY, Luo XJ, Zhu YJ, Feng SG, Yu CN, Lu JJ, Shen CJ, Wang HZ (2018) Comparative metabolomic and proteomic analyses reveal the regulation mechanism underlying MeJA-induced bioactive compound accumulation in cutleaf groundcherry (Physalis angulata L.) hairy roots. J Agric Food Chem 66:6336–6347
Zhang WN, Tong W-Y (2016) Chemical constituents and biological activities of plants from the genus Physalis. Chem Biodivers 13:48–65
Zhang M, Dong JF, Jin HH, Sun LN, Xu MJ (2011) Ultraviolet-B-induced flavonoid accumulation in Betula pendula leaves is dependent upon nitrate reductase-mediated nitric oxide signaling. Tree Physiol 31:798–807
Zhang SC, Yan Y, Wang BQ, Liang ZS, Liu Y, Liu FH, Qi ZH (2014) Selective responses of enzymes in the two parallel pathways of rosmarinic acid biosynthetic pathway to elicitors in Salvia miltiorrhiza hairy root cultures. J Biosci Bioeng 117:645–651
Zhang MY, Wang SY, Yin J, Li CX, Zhan YG, Xiao JL, Liang T, Li X (2016) Molecular cloning and promoter analysis of squalene synthase and squalene epoxidase genes from Betula platyphylla. Protoplasma 253:1347–1363
Zheng YL, Cao C, Lin MH, Zhai Y, Ge ZW, ShenTu JZ, Wu LH, Hu XJ (2017) Identification and quantitative analysis of physalin D and its metabolites in rat urine and feces by liquid chromatography with triple quadrupole time-of-flight mass spectrometry. J Sep Sci 40:2355–2365
Zhou B, Wang Y, Zhan YG, Li YH, Kawabata S (2013) Chalcone synthase family genes have redundant roles in anthocyanin biosynthesis and in response to blue/UV-A light in turnip (Brassica rapa; Brassicaceae). Am J Bot 100:2458–2467
Zhu FF, Dai CY, Fu YF, Loo JFC, Xia DJ, Gao SZ, Ma ZJ, Chen Z (2016) Physalin A exerts anti-tumor activity in non-small cell lung cancer cell lines by suppressing JAK/STAT3 signaling. Oncotarget 7:9462
Acknowledgements
This work was funded by the National Natural Science Foundation of China (Grant Nos. 31470407 and 31970346), and the Major Increase or Decrease Program In The Central Finance Level (Grant No. 2060302). The authors also acknowledge Prof. Zhongjun Ma from Zhejiang University for supporting the authentic standards of Physalins.
Author information
Authors and Affiliations
Contributions
JJL and HZW conceived and designed the study. XRZ, XJL, JYH, CCZ, XYL, YJM and XYX collected and taken care of the plant samples. SGF, CNY and ZFJ performed the experiments. XRZ and JJL analyzed the data. CJS and JJL wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhan, X., Luo, X., He, J. et al. Bioactive compounds induced in Physalis angulata L. by methyl-jasmonate: an investigation of compound accumulation patterns and biosynthesis-related candidate genes. Plant Mol Biol 103, 341–354 (2020). https://doi.org/10.1007/s11103-020-00996-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-00996-y