Abstract
Purpose
Parathyroid carcinoma (PC) is an endocrine malignancy with a poor prognosis. The tumour immune microenvironment is a critical factor influencing the outcomes of multiple cancer types. However, knowledge of the immune microenvironment in PC remains limited.
Methods
The intratumoural density of immunocytes and the Ki-67 index were evaluated immunohistochemically in 51 PC patient samples and were compared with clinicopathological features and parafibromin staining results. The Kaplan–Meier method and Cox proportional hazards analysis were used to estimate the effects of these variables on clinical outcomes.
Results
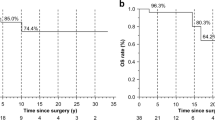
Intratumoural immunocyte density was not correlated with age, gender, urolithiasis, or palpation of a neck mass. The Ki-67 index was correlated with the intratumoural density of CD3+ cells (P = 0.022) and CD8+ cells (P = 0.021) and serum calcium levels (P = 0.022). In the intratumoural area of primary foci, Kaplan–Meier method showed that the risk factors associated with recurrence/metastasis were a low density of CD3+ (P = 0.017), CD8+ (P = 0.019) and CD45+ cells (P = 0.047), a high density of CD163+ cells (P = 0.003) and a high Ki-67 index (P = 0.004). Cox regression multivariate analysis revealed that CD163+ cell density (hazard ratio (HR) 16.19, 95% confidence interval (CI) 1.99—131.66; P = 0.009) and CD8+ cell density (HR 0.13, 95% CI 0.02—0.76, P = 0.024) were independent factors associated with PC relapse.
Conclusion
The immune microenvironment is an important factor influencing the relapse of PC. The intratumoural density of CD3+, CD8+, CD45+, and CD163+ immunocytes was correlated with disease-free survival (DFS) in patients with PC. Immunotherapy based on T lymphocytes or tumour-associated macrophages may be a promising treatment strategy.




Similar content being viewed by others
Data availability
All related data in this manuscript are stored and can be accessed in our database of hyperparathyroidism.
References
Lee PK, Jarosek SL, Virnig BA et al (2007) Trends in the incidence and treatment of parathyroid cancer in the United States. Cancer 109:1736–1741. https://doi.org/10.1002/cncr.22599
Ryhanen EM, Leijon H, Metso S et al (2017) A nationwide study on parathyroid carcinoma. Acta Oncol 56:991–1003. https://doi.org/10.1080/0284186x.2017.1306103
Xue S, Chen H, Lv C et al (2016) Preoperative diagnosis and prognosis in 40 parathyroid carcinoma patients. Clin Endocrinol 85:29–36. https://doi.org/10.1111/cen.13055
Saponaro F, Cetani F, Mazoni L et al (2019) Hypercalciuria: its value as a predictive risk factor for nephrolithiasis in asymptomatic primary hyperparathyroidism? J Endocrinol Investig. https://doi.org/10.1007/s40618-019-01162-y
Cetani F, Pardi E, Marcocci C (2016) Update on parathyroid carcinoma. J Endocrinol Investig 39:595–606. https://doi.org/10.1007/s40618-016-0447-3
Salcuni AS, Cetani F, Guarnieri V et al (2018) Parathyroid carcinoma. Best Pract Res Clin Endocrinol Metab 32:877–889. https://doi.org/10.1016/j.beem.2018.11.002
Gajewski TF, Schreiber H, Fu YX (2013) Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol 14:1014–1022. https://doi.org/10.1038/ni.2703
Vitale I, Sistigu A, Manic G et al (2019) Mutational and antigenic landscape in tumor progression and cancer immunotherapy. Trends Cell Biol 29:396–416. https://doi.org/10.1016/j.tcb.2019.01.003
Fink PJ (2018) The cancer immunotherapy revolution: mechanistic insights. J Immunol 200:371–372. https://doi.org/10.4049/jimmunol.1790024
Kelly PN (2018) The cancer immunotherapy revolution. Science 359:1344–1345. https://doi.org/10.1126/science.359.6382.1344
Bommareddy PK, Shettigar M, Kaufman HL (2018) Integrating oncolytic viruses in combination cancer immunotherapy. Nat Rev Immunol 18:498–513. https://doi.org/10.1038/s41577-018-0014-6
June CH, O'Connor RS, Kawalekar OU et al (2018) CAR T cell immunotherapy for human cancer. Science 359:1361–1365. https://doi.org/10.1126/science.aar6711
Talat N, Diaz-Cano S, Schulte KM (2011) Inflammatory diseases of the parathyroid gland. Histopathology 59:897–908. https://doi.org/10.1111/j.1365-2559.2011.04001.x
Shi Y, Hogue J, Dixit D et al (2014) Functional and genetic studies of isolated cells from parathyroid tumors reveal the complex pathogenesis of parathyroid neoplasia. Proc Natl Acad Sci USA 111:3092–3097. https://doi.org/10.1073/pnas.1319742111
Haglund F, Hallstrom BM, Nilsson IL et al (2017) Inflammatory infiltrates in parathyroid tumors. Eur J Endocrinol 177:445–453. https://doi.org/10.1530/eje-17-0277
Silva-Figueroa A, Villalobos P, Williams MD et al (2018) Characterizing parathyroid carcinomas and atypical neoplasms based on the expression of programmed death-ligand 1 expression and the presence of tumor-infiltrating lymphocytes and macrophages. Surgery 164:960–964. https://doi.org/10.1016/j.surg.2018.06.013
Hu Y, Bi Y, Cui M et al (2019) The influence of surgical extent and parafibromin staining on the outcome of parathyroid carcinoma: 20-year experience from a single institute. Endocr Pract 25:634–641. https://doi.org/10.4158/ep-2018-0538
Camp RL, Dolled-Filhart M, Rimm DL (2004) X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res 10:7252–7259. https://doi.org/10.1158/1078-0432.ccr-04-0713
Prenen H, Mazzone M (2019) Tumor-associated macrophages: a short compendium. Cell Mol Life Sci 76:1447–1458. https://doi.org/10.1007/s00018-018-2997-3
Heusinkveld M, van der Burg SH (2011) Identification and manipulation of tumor associated macrophages in human cancers. J Transl Med 9:216. https://doi.org/10.1186/1479-5876-9-216
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141:39–51. https://doi.org/10.1016/j.cell.2010.03.014
Hori S, Nomura T, Sakaguchi S (2003) Control of regulatory T cell development by the transcription factor Foxp3. Science 299:1057–1061. https://doi.org/10.1126/science.1079490
Wing JB, Tanaka A, Sakaguchi S (2019) Human FOXP3(+) regulatory T cell heterogeneity and function in autoimmunity and cancer. Immunity 50:302–316. https://doi.org/10.1016/j.immuni.2019.01.020
Wang O, Wang CY, Shi J et al (2012) Expression of Ki-67, galectin-3, fragile histidine triad, and parafibromin in malignant and benign parathyroid tumors. Chin Med J 125:2895–2901
Erickson LA, Mete O (2018) Immunohistochemistry in diagnostic parathyroid pathology. Endocr Pathol 29:113–129. https://doi.org/10.1007/s12022-018-9527-6
Funding
This work was supported by the Chinese Academy of Medical Sciences (CAMS) Initiative for Innovative Medicine (CAMS-I2M) (2017-I2M-1-001), Peking Union Medical College Innovative Team Development Program (2016) and Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2018PT32014).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Institutional Ethics Review Board of Peking Union Medical College Hospital (S-K624). All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hu, Y., Cui, M., Bi, Y. et al. Immunocyte density in parathyroid carcinoma is correlated with disease relapse. J Endocrinol Invest 43, 1453–1461 (2020). https://doi.org/10.1007/s40618-020-01224-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-020-01224-6