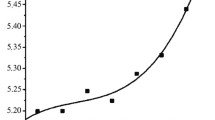
Self-propagating high temperature synthesis (SHS) was used to obtain porous cermet membranes based on a mixture of aluminum oxide, silicon carbide, and magnesium oxide powders. The obtained membranes are catalytically active. The surfaces of the open pores in them were modified by rhenium-tungsten active components using the alkoxy method. The highest selectivity in terms of propane (about 60%) is achieved at temperature 600°C. In addition, the optimal conditions for implementing the propane dehydrogenation process is temperature 650°C at which the quite high selectivity for the most important monomers remains with a relatively small contribution of accessory processes.






Similar content being viewed by others
References
I. Amghizar, L. A. Vandewalle, K. M. Van Geem, and G. B. Marin, “New trends in olefin production,” Engineering, 3(2), 171 – 178 (2017); URL: https://doi.org/10.1016/J.ENG.2017.02.006.
N. L. Morrow, “The industrial production and use of 1,3-butadiene,” Environmental Health Perspectives, 86(7 – 8) (1990); URL: https://doi.org/10.1289/ehp.90867.
W. C. White, “Butadiene production process overview,” Chem.-Biolog. Interactions, 166(1 – 3), 10 – 14 (2007); URL: https://doi.org/10.1016/j.cbi.2007.01.009.
G. O. Ezinkwo, V. F. Tretjakov, R. M. Talyshinky, et al., “Overview of the catalytic production of isoprene from different raw materials; prospects of isoprene production from bio-ethanol,” Catalysis for Sustainable Energy, No. 1, 100 – 111 (2013); URL: https://doi.org/10.2478/cse-2013-0006.
W.W. Kaeding, “Styrene monomer,” Catal. Rev., 8(1), 307 – 316 (1974); URL: https://doi.org/10.1080/01614947408071865.
I. A. Makaryan and V. I. Savchenko, “On the way to develop an alternative energy-saving ‘on-purpose’ propylene production,” Alternative Energy and Ecology, No. 10, 99 – 121 (2009); URL: https://elibrary.ru/download/elibrary_12963698_30637490.pdf.
A. V. Lavrenov, L. F. Saifulina, E. A. Buluchevskii, and E. N. Bogdanets, “Propylene production technology: Today and tomorrow,” Catalysis in Industry, 7(3), 175 – 187 (2015); URL: https://doi.org/10.1134/S2070050415030083.
Chuck Carr, Global Propylene Market, Motivated to Change; URL: http://cdn.ihs.com/www/pdf/asia-chem-conf/Carr.pdf.
Global Production of Propylene Running at 90% Capacity, Information and Analytical Center RUPEC; URL: http://rupec.ru/news/39475/.
IXS Markit: Propylene, Chemical Economics Handbook; URL: https://ihsmarkit.com/products/propylene-chemical-economicshandbook.html.
I. M. Kurchatov, N. I. Laguntsov, M. V. Tsodikov, et al., “The nature of permeability anisotropy and catalytic activity,” Kinetics and Catalysis, 49(1), 121 – 126 (2008); URL: https://doi.org/10.1134/S0023158408010151.
M. V. Tsodikov, A. S. Fedotov, V. V. Zhmakin, et al., “Carbon dioxide reforming of alcohols on porous membrane catalyst systems,” Petroleum Chem., 51(7), 568 – 576 (2011); URL: https://doi.org/10.1134/S0965544111070127.
A. S. Fedotov, D. O. Antonov, V. I. Uvarov, et al., “Synergistic effect in the dry methane reforming on porous ceramic Ni–Co membranes,” Dokl. Chem., 459(1), 202 – 204 (2014); URL: https://doi.org/10.1134/S0012500814110020.
A. S. Fedotov, D. O. Antonov, O. V. Bukhtenko, et al., “The role of aluminum in the formation of Ni–Al–Co-containing porous ceramic converters with high activity in dry and steam reforming of methane and ethanol,” Int. J. Hydrogen Energy, 42(38), 24131 – 24141 (2017); URL: https://doi.org/10.1016/j.ijhydene.2017.07.095.
A. S. Fedotov, D. O. Antonov, V. I. Uvarov, and M. V. Tsodikov, “Original hybrid membrane-catalytic reactor for the CoProduction of syngas and ultrapure hydrogen in the processes of dry and steam reforming of methane, ethanol and DME,” Int. J. Hydrogen Energy, 43(14), 7046 – 7054 (2018); URL: https://doi.org/10.1016/j.ijhydene.2018.02.060.
Author information
Authors and Affiliations
Additional information
Translated from Steklo i Keramika, No. 11, pp. 35 – 39, November, 2019.
Rights and permissions
About this article
Cite this article
Uvarov, V.I., Loryan, V.É., Kapustin, R.D. et al. Using Porous Ceramic Catalytic Converters for Dehydrogenation of Propane in Propylene. Glass Ceram 76, 428–431 (2020). https://doi.org/10.1007/s10717-020-00216-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10717-020-00216-5