Abstract
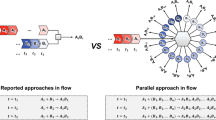
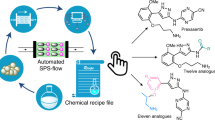
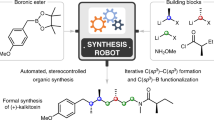
Automated synthesis platforms accelerate and simplify the preparation of molecules by removing the physical barriers to organic synthesis. This provides unrestricted access to biopolymers and small molecules via reproducible and directly comparable chemical processes. Current automated multistep syntheses rely on either iterative1,2,3,4 or linear processes5,6,7,8,9, and require compromises in terms of versatility and the use of equipment. Here we report an approach towards the automated synthesis of small molecules, based on a series of continuous flow modules that are radially arranged around a central switching station. Using this approach, concise volumes can be exposed to any reaction conditions required for a desired transformation. Sequential, non-simultaneous reactions can be combined to perform multistep processes, enabling the use of variable flow rates, reuse of reactors under different conditions, and the storage of intermediates. This fully automated instrument is capable of both linear and convergent syntheses and does not require manual reconfiguration between different processes. The capabilities of this approach are demonstrated by performing optimizations and multistep syntheses of targets, varying concentrations via inline dilutions, exploring several strategies for the multistep synthesis of the anticonvulsant drug rufinamide10, synthesizing eighteen compounds of two derivative libraries that are prepared using different reaction pathways and chemistries, and using the same reagents to perform metallaphotoredox carbon–nitrogen cross-couplings11 in a photochemical module—all without instrument reconfiguration.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this Article and its Supplementary Information file.
Code availability
The complete software package and instructions necessary for assembling and operating the radial synthesizer are freely available to academic users by request at http://synthesizer.mpikg.mpg.de/.
References
Merrifield, R. B. Automated synthesis of peptides. Science 150, 178–185 (1965).
Caruthers, M. H. Gene synthesis machines: DNA chemistry and its uses. Science 230, 281–285 (1985).
Plante, O. J., Palmacci, E. R. & Seeberger, P. H. Automated solid-phase synthesis of oligosaccharides. Science 291, 1523–1527 (2001).
Li, J. et al. Synthesis of many different types of organic small molecules using one automated process. Science 347, 1221–1226 (2015).
Zhang, P. et al. Advanced continuous flow platform for on-demand pharmaceutical manufacturing. Chem. Eur. J. 24, 2776–2784 (2018).
Adamo, A. et al. On-demand continuous-flow production of pharmaceuticals in a compact, reconfigurable system. Science 352, 61–67 (2016).
Bédard, A. C. et al. Reconfigurable system for automated optimization of diverse chemical reactions. Science 361, 1220–1225 (2018).
Ghislieri, D., Gilmore, K. & Seeberger, P. H. Chemical assembly systems: layered control for divergent, continuous, multistep syntheses of active pharmaceutical ingredients. Angew. Chem. Int. Ed. 54, 678–682 (2015).
Britton, J. & Jamison, T. F. A unified continuous flow assembly-line synthesis of highly subsituted pyrazoles and pyrazolines. Angew. Chem. Int. Ed. 56, 8823–8827 (2017).
Padmaja, R. D. & Chanda, K. A short review on synthetic advances towards the synthesis of rufinamide, an entiepileptic drug. Org. Process Res. Dev. 22, 457–466 (2018).
Corcoran, E. B. et al. Aryl amination using ligand-free Ni(ii) salts and photoredox catalysis. Science 353, 279–283 (2016).
Plutschack, M. B., Pieber, B., Gilmore, K. & Seeberger, P. H. The hitchhiker’s guide to flow chemistry. Chem. Rev. 117, 11796–11893 (2017).
Britton, J. & Raston, C. L. Multi-step continuous-flow synthesis. Chem. Soc. Rev. 46, 1250–1271 (2017).
Gérardy, R. et al. Continuous flow organic chemistry: successes and pitfalls at the interface with current societal challenges. Eur. J. Org. Chem. 2018, 2301–2351 (2018).
Pastre, J. C., Browne, D. L. & Ley, S. V. Flow chemistry syntheses of natural products. Chem. Soc. Rev. 42, 8849–8869 (2013).
Malet-Sanz, L. & Susanne, F. Continuous flow synthesis. A pharma perspective. J. Med. Chem. 55, 4062–4098 (2012).
Gutmann, B., Cantillo, D. & Kappe, C. O. Continuous-flow technology—a tool for the safe manufacturing of active pharmaceutical ingredients. Angew. Chem. Int. Ed. 54, 6688–6728 (2015).
Steiner, S. et al. Organic synthesis in a modular robotic system driven by a chemical programming language. Science 363, eaav2211 (2019).
Schotten, C., Leist, L. G. T., Semrau, A. L. & Browne, D. L. A machine-assisted approach for the preparation of follow-on pharmaceutical compound libraries. React. Chem. Eng. 3, 210–215 (2018).
Hwang, Y. J. et al. A segmented flow platform for on-demand medicinal chemistry and compound synthesis in oscillating droplets. Chem. Commun. 53, 6649–6652 (2017).
Hepler, L. G. Correct calculation of ΔH°, ΔC°P, and ΔV° from temperature and pressure dependences of equilibrium constants: the importance of thermal expansion and compressibility of the solvent. Thermochim. Acta 50, 69–72 (1981).
Fitzpatrick, D. E., Maujean, T., Evans, A. C. & Ley, S. V. Across-the-world automated optimization and continuous-flow synthesis of pharmaceutical agents operating through a cloud-based server. Angew. Chem. Int. Ed. 57, 15128–15132 (2018).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Milligan, J. A., Phelan, J. P., Badir, S. O. & Molander, G. A. Alkyl carbon–carbon bond formation by nickel/photoredox cross-coupling. Angew. Chem. Int. Ed. 58, 6152–6163 (2019).
Acknowledgements
We acknowledge financial support from the Max-Planck Society, the Army Research Office under contract W911NF-16-1-0557, and the DFG InCHeM (FOR 2177). We thank B. Pieber, J. Malik, S. Moon and F. Hentschel for suggestions and support; J. von Szada-Borryszkowski and J. Petersen for construction of the instrument housing; and Vapourtec for software support with the incorporation of their equipment.
Author information
Authors and Affiliations
Contributions
K.G. designed the approach and system, and refined the instrument and chemistries. M.G. and K.G. wrote the draft of the manuscript, and all authors participated in revising the manuscript. S.C. designed, wrote and developed the software, controls, user interface and hardware layout. M.G. developed chemistry, designed and executed optimizations, developed pathways for multistep processes and refined the system. P.H.S. and K.G. provided oversight to the project and secured funding.
Corresponding author
Ethics declarations
Competing interests
The Max-Planck Society has been granted patent EP3386628B1, which covers the system described here and lists S.C., P.H.S. and K.G. as inventors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file contains Supplementary Figures S1–S146, Supplementary Tables S1–S23 and Supplementary Schemes S1–S34.
Rights and permissions
About this article
Cite this article
Chatterjee, S., Guidi, M., Seeberger, P.H. et al. Automated radial synthesis of organic molecules. Nature 579, 379–384 (2020). https://doi.org/10.1038/s41586-020-2083-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2083-5
This article is cited by
-
A dynamic knowledge graph approach to distributed self-driving laboratories
Nature Communications (2024)
-
Universal chemical programming language for robotic synthesis repeatability
Nature Synthesis (2024)
-
Identifying general reaction conditions by bandit optimization
Nature (2024)
-
An integrated self-optimizing programmable chemical synthesis and reaction engine
Nature Communications (2024)
-
Inverse design of chiral functional films by a robotic AI-guided system
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.