Key message
A novel QTL (qSCN-PL10) for SCN resistance and related candidate genes were identified in the soybean variety Pingliang xiaoheidou, and plant basal immunity seems to contribute to the SCN resistance.
Abstract
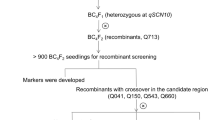
Soybean cyst nematode (SCN, Heterodera glycines Ichinohe) is one of the most devastating soybean pests worldwide. The development of host plant resistance represents an effective strategy to control SCN. However, owing to the lack of diversity of resistance genes in soybean varieties, further investigation is necessary to identify new SCN resistance genes. By analyzing the resistance phenotypes of soybean variety Pingliang xiaoheidou (Pingliang, ZDD 11047), we found that it exhibited the different resistance phenotypes from PI 88788 and Peking varieties. Because Pingliang variety contains the Rhg1-a (low copy) haplotype and lacks the resistant Rhg4 haplotype, novel quantitative trait locus might account for their SCN resistance. After sequencing parental lines (Magellan and Pingliang) and 200 F2:3 progenies, a high-density genetic map was constructed using the specific length amplified fragment sequencing method and qSCN-PL10 was identified as a novel locus for SCN resistance. Candidate genes were predicted by RNA sequencing (RNA-seq) in the qSCN-PL10 locus region. The RNA-seq analysis performed also indicated that plant basal immunity plays an important role in the resistance of Pingliang to SCN. These results lay a foundation for the use of marker-assisted breeding to enhance the resistance to SCN.







Similar content being viewed by others
Data availability
RNA-Seq‘s raw data has been published at WEBLINK https://www.ncbi.nlm.nih.gov/bioproject/579241 with BioProject ID PRJNA579241.
References
Afzal AJ, Srour A, Saini N, Hemmati N, El Shemy HA, Lightfoot DA (2012) Recombination suppression at the dominant Rhg1/Rfs2 locus underlying soybean resistance to the cyst nematode. Theor Appl Genet 124:1027–1039
Arelli AP, Wilcox JA, Myers O Jr, Gibson PT (1997) Soybean germplasm resistant to races 1 and 2 of Heterodera glycines. Crop Sci 37:1367–1369
Arelli PR, Sleper DA, Pin Y, Wilcox JA (2000) Soybean reaction to Races 1 and 2 of Heterodera glycines. Crop Sci 40:824–826
Brown S, Yeckel G, Heinz R, Clark K, Sleper D, Mitchum MG (2010) A high-throughput automated technique for counting females of Heterodera glycines using a fluorescence-based imaging system. J Nematol 42:201
Brucker E, Carlson S, Wright E, Niblack T, Diers B (2005) Rhg1 alleles from soybean PI 437654 and PI 88788 respond differentially to isolates of Heterodera glycines in the greenhouse. Theor Appl Genet 111:44–49
Chen JC, Lu HC, Chen CE, Hsu HF, Chen HH, Yeh HH (2013) The NPR1 ortholog PhaNPR1 is required for the induction of PhaPR1 in Phalaenopsis aphrodite. Bot Stud 54:1–11
Chen JS, Li XB, Li ZY, Zhou CJ, Luo X, Wang D, Duan YX, Chen Lj (2015) Identification of the virulence type of soybean cyst nematode under continuous cropping in Daqing and Anda. Soybean Sci 34:676–678
Concibido VC, Diers BW, Arelli PR (2004) A decade of QTL mapping for cyst nematode resistance in soybean. Crop Sci 44:1121–1131
Cook DE, Bayless AM, Wang K, Guo X, Song Q, Jiang J, Bent AF (2014) Distinct copy number, coding sequence, and locus methylation patterns underlie Rhg1-mediated soybean resistance to soybean cyst nematode. Plant Physiol 165:630–647
Cook DE, Lee TG, Guo X, Melito S, Wang K, Bayless AM, Wang J, Hughes TJ, Willis DK, Clemente TE et al (2012) Copy number variation of multiple genes at Rhg1 mediates nematode resistance in soybean. Science 338:1206–1209
Fricke W (2001) Xylem: differentiation, water transport and ecology. eLS 1–7
Goodstein DM, Shu S, Howson R, Neupane R, Hayes RD, Fazo J, Mitros T, Dirks W, Hellsten U, Putnam N et al (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40:D1178–1186
Guo B, Sleper DA, Arelli PR, Shannon JG, Nguyen HT (2005) Identification of QTLs associated with resistance to soybean cyst nematode races 2, 3 and 5 in soybean PI 90763. Theor Appl Genet 111:965–971
Guo W, Zhang F, Bao A, You Q, Li Z, Chen J, Cheng Y, Zhao W, Shen X, Zhou X et al (2019) The soybean Rhg1 amino acid transporter gene alters glutamate homeostasis and jasmonic acid-induced resistance to soybean cyst nematode. Mol Plant Pathol 20:270–286
He Y, Xu J, Wang X, He X, Wang Y, Zhou J, Zhang S, Meng X (2019) The Arabidopsis pleiotropic drug resistance transporters PEN3 and PDR12 mediate camalexin secretion for resistance to Botrytis cinerea. Plant Cell 31:2206–2222
Healey A, Furtado A, Cooper T, Henry RJ (2014) Protocol: a simple method for extracting next-generation sequencing quality genomic DNA from recalcitrant plant species. Plant Methods 10:21
Hernandez-Sanchez IE, Maruri-Lopez I, Graether SP, Jimenez-Bremont JF (2017) In vivo evidence for homo- and heterodimeric interactions of Arabidopsis thaliana dehydrins AtCOR47, AtERD10, and AtRAB18. Sci Rep 7:17036
Holbein J, Grundler FM, Siddique S (2016) Plant basal resistance to nematodes: an update. J Exp Bot 67:2049–2061
Irieda H, Inoue Y, Mori M, Yamada K, Oshikawa Y, Saitoh H, Uemura A, Terauchi R, Kitakura S, Kosaka A et al (2019) Conserved fungal effector suppresses PAMP-triggered immunity by targeting plant immune kinases. Proc Natl Acad Sci USA 116:496–505
Jiao Y, Vuong TD, Liu Y, Meinhardt C, Liu Y, Joshi T, Cregan PB, Xu D, Shannon JG, Nguyen HT (2015) Identification and evaluation of quantitative trait loci underlying resistance to multiple HG types of soybean cyst nematode in soybean PI 437655. Theor Appl Genet 128:15–23
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T et al (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–484
Kim M, Hyten DL, Niblack TL, Diers BW (2011) Stacking resistance alleles from wild and domestic soybean sources improves soybean cyst nematode resistance. Crop Sci 51:934–943
Kosambi DD (1943) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Kud J, WangW GR, Fan Y, Huang L, Yuan Y, Gray A, Duarte A, Kuhl JC, Caplan A et al (2019) The potato cyst nematode effector RHA1B is a ubiquitin ligase and uses two distinct mechanisms to suppress plant immune signaling. PLoS Pathog 15:e1007720
Lee MW, Huffaker A, Crippen D, Robbins RT, Goggin FL (2018) Plant elicitor peptides promote plant defences against nematodes in soybean. Mol Plant Pathol 19:858–869
Lee TG, Kumar I, Diers BW, Hudson ME (2015) Evolution and selection of Rhg1, a copy-number variant nematode-resistance locus. Mol Ecol 24:1774–1791
Li R, Yu C, Li Y, Lam TW, Yiu SM, Kristiansen K, Wang J (2009) SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics 25:1966–1967
Li Y, Cui H, Cui X, Wang A (2016) The altered photosynthetic machinery during compatible virus infection. Curr Opin Virol 17:19–24
Lin B, Zhuo K, Chen S, Hu L, Sun L, Wang X, Zhang LH, Liao J (2015) A novel nematode effector suppresses plant immunity by activating host reactive oxygen species-scavenging system. New Phytol 209:1159–1173
Liu D, Ma C, Hong W, Huang L, Liu M, Liu H, Zeng H, Deng D, Xin H, Song J et al (2014) Construction and analysis of high-density linkage map using high-throughput sequencing data. PLoS ONE 9:e98855
Liu S, Kandoth PK, Warren SD, Yeckel G, Heinz R, Alden J, Yang C, Jamai A, El-Mellouki T, Juvale PS et al (2012) A soybean cyst nematode resistance gene points to a new mechanism of plant resistance to pathogens. Nature 492:256–260
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Lu Y, Yao J (2018) Chloroplasts at the crossroad of photosynthesis, pathogen infection and plant defense. Int J Mol Sci. https://doi.org/10.3390/ijms19123900
Manohar M, Tenjo-Castano F, Chen S, Zhang YK, Kumari A, Williamson VM, Wang X, Klessig DF, Schroeder FC (2020) Plant metabolism of nematode pheromones mediates plant-nematode interactions. Nat Commun 11:208
Manosalva P, Manohar M, von Reuss SH, Chen S, Koch A, Kaplan F, Choe A, Micikas RJ, Wang X, Kogel KH et al (2015) Conserved nematode signalling molecules elicit plant defenses and pathogen resistance. Nat Commun 6:7795
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–3793
Mendy B, Wang'ombe MW, Radakovic ZS, Holbein J, Ilyas M, Chopra D, Holton N, Zipfel C, Grundler FM, Siddique S (2017) Arabidopsis leucine-rich repeat receptor-like kinase NILR1 is required for induction of innate immunity to parasitic nematodes. PLoS Pathog 13:e1006284
Mitchum MG (2016) Soybean resistance to the soybean cyst nematode Heterodera glycines: an update. Phytopathology 106:1444–1450
Mitchum MG, Wrather JA, Heinz RD, Shannon JG, Danekas G (2007) Variability in distribution and virulence phenotypes of Heterodera glycines in missouri during 2005. Plant Dis 91:1473–1476
Monecke T, Buschmann J, Neumann P, Wahle E, Ficner R (2014) Crystal structures of the novel cytosolic 5'-nucleotidase IIIB explain its preference for m7GMP. PLoS ONE 9:e90915
Moran-Diez E, Rubio B, Dominguez S, Hermosa R, Monte E, Nicolas C (2012) Transcriptomic response of Arabidopsis thaliana after 24 h incubation with the biocontrol fungus Trichoderma harzianum. J Plant Physiol 169:614–620
Mulo P (2011) Chloroplast-targeted ferredoxin-NADP(+) oxidoreductase (FNR): structure, function and location. Biochim Biophys Acta 1807:927–934
Niblack TL, Lambert KN, Tylka GL (2006) A model plant pathogen from the kingdom Animalia: Heterodera glycines, the soybean cyst nematode. Annu Rev Phytopathol 44:283–303
Niblack TL, Tylka GL, Arelli P, Bond J, Diers B, Donald P, Faghihi J, Gallo K, Heinz RD, Lopez-Nicora H et al (2009) A standard greenhouse method for assessing soybean cyst nematode resistance in soybean: SCE08 (standardized cyst evaluation 2008). Plant Health Prog 10(1):33
Nivedita Verma PK, Upadhyaya KC (2017) Lectin protein kinase is induced in plant roots in response to the endophytic fungus, Piriformospora indica. Plant Mol Biol Reporter 35:323–332
Noctor G, Mhamdi A (2017) Climate change, CO2, and defense: the metabolic, redox, and signaling perspectives. Trends Plant Sci 22:857–870
Noman A, Aqeel M, Lou Y (2019) PRRs and NB-LRRs: from signal perception to activation of plant innate immunity. Int J Mol Sci. https://doi.org/10.3390/ijms20081882
Patil GB, Lakhssassi N, Wan J, Song L, Zhou Z, Klepadlo M, Vuong TD, Stec AO, Kahil SS, Colantonio V, Valliyodan B, Rice JH, Piya S, Hewezi T, Stupar RM, Meksem K, Nguyen HT (2019) Whole-genome re-sequencing reveals the impact of the interaction of copy number variants of the rhg1 and Rhg4 genes on broad-based resistance to soybean cyst nematode. Plant Biotechnol J 17(8):1595–1611
Plett JM, Tisserant E, Brun A, Morin E, Grigoriev IV, Kuo A, Martin F, Kohler A (2015) The mutualist laccaria bicolor expresses a core gene regulon during the colonization of diverse host plants and a variable regulon to counteract host-specific defenses. Mol Plant Microb Interact 28:261–273
Sato K, Kadota Y, Shirasu K (2019) Plant immune responses to parasitic nematodes. Front Plant Sci 10:1165
Schaff JE, Nielsen DM, Smith CP, Scholl EH, Bird DM (2007) Comprehensive transcriptome profiling in tomato reveals a role for glycosyltransferase in Mi-mediated nematode resistance. Plant Physiol 144:1079–1092
Serrano I, Audran C, Rivas S (2016) Chloroplasts at work during plant innate immunity. J Exp Bot 67:3845–3854
Shah SJ, Anjam MS, Mendy B, Anwer MA, Habash SS, Lozano-Torres JL, Grundler FMW, Siddique S (2017) Damage-associated responses of the host contribute to defence against cyst nematodes but not root-knot nematodes. J Exp Bot 68:5949–5960
Sun X, Liu D, Zhang X, Li W, Liu H, Hong W, Jiang C, Guan N, Ma C, Zeng H et al (2013) SLAF-seq: an efficient method of large-scale de novo SNP discovery and genotyping using high-throughput sequencing. PLoS ONE 8:e58700
Teixeira MA, Wei L, Kaloshian I (2016) Root-knot nematodes induce pattern-triggered immunity in Arabidopsis thaliana roots. New Phytol 211:276–287
Tiwari M, Sharma D, Singh M, Tripathi RD, Trivedi PK (2014) Expression of OsMATE1 and OsMATE2 alters development, stress responses and pathogen susceptibility in Arabidopsis. Sci Rep 4:3964
Upadhyay N, Kar D, Deepak Mahajan B, Nanda S, Rahiman R, Panchakshari N, Bhagavatula L, Datta S (2019) The multitasking abilities of MATE transporters in plants. J Exp Bot 70:4643–4656
van Ooijen JW (2011) Multipoint maximum likelihood mapping in a full-sib family of an outbreeding species. Genet Res (Camb) 93:343–349
van Os H, Stam P, Visser RG, van Eck HJ (2005) SMOOTH: a statistical method for successful removal of genotyping errors from high-density genetic linkage data. Theor Appl Genet 112:187–194
Vuong TD, Sleper DA, Shannon JG, Nguyen HT (2010) Novel quantitative trait loci for broad-based resistance to soybean cyst nematode (Heterodera glycines Ichinohe) in soybean PI 567516C. Theor Appl Genet 121:1253–1266
Wang Y, Bouwmeester K (2017) L-type lectin receptor kinases: new forces in plant immunity. PLoS Pathog 13:e1006433
Wang Z, Gerstein M, Snyder M (2009) RNA-seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10:57–63
Wanke D, Kolukisaoglu HU (2010) An update on the ABCC transporter family in plants: many genes, many proteins, but how many functions? Plant Biol (Stuttg) 12(Suppl 1):15–25
Wen Z, Tan R, Zhang S, Collins PJ, Yuan J, Du W, Gu C, Ou S, Song Q, An YC et al (2018) Integrating GWAS and gene expression data for functional characterization of resistance to white mould in soya bean. Plant Biotechnol J 16:1825–1835
Winter SM, Shelp BJ, Anderson TR, Welacky TW, Rajcan I (2007) QTL associated with horizontal resistance to soybean cyst nematode in Glycine soja PI464925B. Theor Appl Genet 114:461–472
Wu CH, Abd-El-Haliem A, Bozkurt TO, Belhaj K, Terauchi R, Vossen JH, Kamoun S (2017) NLR network mediates immunity to diverse plant pathogens. Proc Natl Acad Sci USA 114:8113–8118
Wu G, Liu S, Zhao Y, Wang W, Kong Z, Tang D (2015) ENHANCED DISEASE RESISTANCE4 associates with CLATHRIN HEAVY CHAIN2 and modulates plant immunity by regulating relocation of EDR1 in Arabidopsis. Plant Cell 27:857–873
Wu X, Blake S, Sleper DA, Shannon JG, Cregan P, Nguyen HT (2009) QTL, additive and epistatic effects for SCN resistance in PI 437654. Theor Appl Genet 118:1093–1105
Yang Y, Zhou Y, Chi Y, Fan B, Chen Z (2017) characterization of soybean WRKY gene family and identification of soybean WRKY genes that promote resistance to soybean cyst nematode. Sci Rep 7:17804
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11(2):R14
Zhang H, Kjemtrup-Lovelace S, Li C, Luo Y, Chen LP, Song BH (2017) Comparative rna-seq analysis uncovers a complex regulatory network for soybean cyst nematode resistance in wild soybean (Glycine soja). Sci Rep 7:9699
Acknowledgements
This work was supported by the National Key R&D Program of China (2016YFD0100603), the Major project of Research and Development of Applied Technology in Heilongjiang Province (GA18B101), the Agricultural Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences, the National Natural Science Foundation of China (31501334 and 31522042).
Author information
Authors and Affiliations
Contributions
WG, XJS, JSC, and YQJ designed the study; WG, JSC, FZ, CJZ, and HFC performed the QTL and genomic qPCR analyses; LMC, SLY, RL, ZYL, WJD, and JSC performed the nematode assays; SLC, ZHS, ZLY, XJZ, DZQ, QBY, and XJS performed the qRT-PCR analysis; DC, QNH, WG, and JSC analyzed the data; WG, XJS, XAZ, and JSC wrote the paper; WG, XJS, and YQJ coordinated the study. All authors have read and approved the manuscript for submission.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Guo, W., Chen, J.S., Zhang, F. et al. Characterization of Pingliang xiaoheidou (ZDD 11047), a soybean variety with resistance to soybean cyst nematode Heterodera glycines. Plant Mol Biol 103, 253–267 (2020). https://doi.org/10.1007/s11103-020-00990-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-00990-4