Abstract
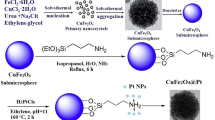
Zr-based metal-organic framework nanocable catalysts comprising MTe(M = Pt, PtCu)@UIO-67 resembling “sugarcoated haws on a stick” were assembled. Ultrathin Te nanowires with diameters of about 8.48 nm served as a sacrificial template for the epitaxial growth of an outer M (Pt or PtCu) to obtain cable cores comprising MTe nanowires with good dispersity, which were then successfully encapsulated in the center of a monodisperse MTe@UIO-67 cable nano-structured catalyst. Transmission electron microscopy (TEM), scanning electron microscopy (SEM), powder X-ray diffraction (XRD) and energy dispersive X-ray (EDX) element mapping were used to investigate the morphology, structure and composition of the nanocable catalysts. Six types of catalysts were synthesized, and all of the catalysts demonstrated superior product selectivity to CO for reverse water-gas shift (RWGS) reaction. Especially, the morphology and dispersion of PtCuTe@UIO-67 nanocable catalyst can be maintained to some extent after catalysis at high temperature, and PtTe@UIO-67 catalyzed CO2 conversion was achieved with 99.86% CO selectivity.
摘要
本文以锆系金属有机骨架为载体合成了具有“糖葫芦”结构形貌的纳米电缆状催化剂MTe (M = Pt, PtCu)@UIO-67. 首先以直径大约为8.48 nm的超细Te纳米线作为牺牲模板, 用于外延生长外部的金属粒子(Pt或PtCu), 得到具有良好分散性的电缆芯形状的MTe纳米线, 然后成功地封装在单分散的MTe@UIO-67电缆状纳米结构催化剂的中心. 采用透射电子显微镜、 扫描电子显微镜、 粉末X射线衍射和能谱等方法对纳米电缆催化剂的形貌结构和组成进行了表征. 本文合成的六种催化剂均表现出良好的CO产物选择性, 在高温催化后, PtCuTe@UIO-67催化剂的形貌和分散性能得到一定程度的保持, 而PtTe@UIO-67催化CO2转化成CO的选择性可以达到99.86%.
Article PDF
Similar content being viewed by others
References
Aminu MD, Nabavi SA, Rochelle CA, et al. A review of developments in carbon dioxide storage. Appl Energy, 2017, 208: 1389–1419
Rahman FA, Aziz MMA, Saidur R, et al. Pollution to solution: Capture and sequestration of carbon dioxide (CO2) and its utilization as a renewable energy source for a sustainable future. Renew Sustain Energy Rev, 2017, 71: 112–126
Li B, Duan Y, Luebke D, et al. Advances in CO2 capture technology: A patent review. Appl Energy, 2013, 102: 1439–1447
Jiang X, Guan D. The global CO2 emissions growth after international crisis and the role of international trade. Energy Policy, 2017, 109: 734–746
Saeidi S, Amin NAS, Rahimpour MR. Hydrogenation of CO2 to value-added products—A review and potential future developments. J CO2 Utilization, 2014, 5: 66–81
Goeppert A, Czaun M, Jones JP, et al. Recycling of carbon dioxide to methanol and derived products—closing the loop. Chem Soc Rev, 2014, 43: 7995–8048
Aziz MAA, Jalil AA, Triwahyono S, et al. Highly active Ni-pro-moted mesostructured silica nanoparticles for CO2 methanation. Appl Catal B-Environ, 2014, 147: 359–368
Guo L, Sun J, Ge Q, et al. Recent advances in direct catalytic hydrogenation of carbon dioxide to valuable C2+ hydrocarbons. J Mater Chem A, 2018, 6: 23244–23262
Hu B, Guild C, Suib SL. Thermal, electrochemical, and photochemical conversion of CO2 to fuels and value-added products. CO2 Utilization, 2013, 1: 18–27
Melaet G, Ralston WT, Li CS, et al. Evidence of highly active cobalt oxide catalyst for the Fischer-Tropsch synthesis and CO2 hydrogenation. J Am Chem Soc, 2014, 136: 2260–2263
Khodakov AY, Chu W, Fongarland P. Advances in the development of novel cobalt Fischer-Tropsch catalysts for synthesis of long-chain hydrocarbons and clean fuels. Chem Rev, 2007, 107: 1692–1744
Goguet A, Meunier FC, Tibiletti D, et al. Spectrokinetic investigation of reverse water-gas-shift reaction intermediates over a Pt/CeO2 catalyst. J Phys Chem B, 2004, 108: 20240–20246
Kim DH, Han SW, Yoon HS, et al. Reverse water gas shift reaction catalyzed by Fe nanoparticles with high catalytic activity and stability. J Industrial Eng Chem, 2015, 23: 67–71
Dagle RA, Platon A, Palo DR, et al. PdZnAl catalysts for the reactions of water-gas-shift, methanol steam reforming, and reverse-water-gas-shift. Appl Catal A-General, 2008, 342: 63–68
Tsuchiya K, Huang JD, Tominaga K. Reverse water-gas shift reaction catalyzed by mononuclear Ru complexes. ACS Catal, 2013, 3: 2865–2868
Xia BY, Wu HB, Li N, et al. One-pot synthesis of Pt-Co alloy nanowire assemblies with tunable composition and enhanced electrocatalytic properties. Angew Chem Int Ed, 2015, 54: 3797–3801
Hong W, Wang J, Wang E. Dendritic Au/Pt and Au/PtCu nano-wires with enhanced electrocatalytic activity for methanol electrooxidation. Small, 2014, 10: 3262–3265
Hong W, Shang C, Wang J, et al. Bimetallic PdPt nanowire networks with enhanced electrocatalytic activity for ethylene glycol and glycerol oxidation. Energy Environ Sci, 2015, 8: 2910–2915
Sai Siddhardha RS, Kumar MA, Lakshminarayanan V, et al. Anti-fouling response of gold-carbon nanotubes composite for enhanced ethanol electrooxidation. J Power Sources, 2014, 271: 305–311
Zhao GY, Xu CL, Guo DJ, et al. Template preparation of Pt-Ru and Pt nanowire array electrodes on a Ti/Si substrate for methanol electro-oxidation. J Power Sources, 2006, 162: 492–496
Zhu C, Guo S, Dong S. PdM (M = Pt, Au) bimetallic alloy nano-wires with enhanced electrocatalytic activity for electro-oxidation of small molecules. Adv Mater, 2012, 24: 2326–2331
Qi Y, Bian T, Choi SI, et al. Kinetically controlled synthesis of Pt-Cu alloy concave nanocubes with high-index facets for methanol electro-oxidation. Chem Commun, 2014, 50: 560–562
Fu X, Zhao Z, Wan C, et al. Ultrathin wavy Rh nanowires as highly effective electrocatalysts for methanol oxidation reaction with ultrahigh ECSA. Nano Res, 2019, 12: 211–215
Chen B, Cheng D, Zhu J. Synthesis of PtCu nanowires in non-aqueous solvent with enhanced activity and stability for oxygen reduction reaction. J Power Sources, 2014, 267: 380–387
Yu M, Wang Z, Hou C, et al. Nitrogen-doped Co3O4 mesoporous nanowire arrays as an additive-free air-cathode for flexible solidstate zinc-air batteries. Adv Mater, 2017, 29: 1602868
Bai Y, Dou Y, Xie LH, et al. Zr-based metal-organic frameworks: design, synthesis, structure, and applications. Chem Soc Rev, 2016, 45: 2327–2367
Lustig WP, Mukherjee S, Rudd ND, et al. Metal-organic frameworks: functional luminescent and photonic materials for sensing applications. Chem Soc Rev, 2017, 46: 3242–3285
Li JR, Sculley J, Zhou HC. Metal-organic frameworks for separations. Chem Rev, 2012, 112: 869–932
Huang YB, Liang J, Wang XS, et al. Multifunctional metal-organic framework catalysts: synergistic catalysis and tandem reactions. Chem Soc Rev, 2017, 46: 126–157
Zhu Y, Ciston J, Zheng B, et al. Unravelling surface and interfacial structures of a metal-organic framework by transmission electron microscopy. Nat Mater, 2017, 16: 532–536
Furukawa H, Cordova KE, O’Keeffe M, et al. The chemistry and applications of metal-organic frameworks. Science, 2013, 341: 1230444
Rowsell JLC, Yaghi OM. Metal-organic frameworks: a new class of porous materials. Microporous Mesoporous Mater, 2004, 73: 3–14
Liu J, Strachan DM, Thallapally PK. Enhanced noble gas adsorption in Ag@MOF-74Ni. Chem Commun, 2014, 50: 466–468
Wang X, Xie L, Huang KW, et al. A rationally designed amino-borane complex in a metal organic framework: a novel reusable hydrogen storage and size-selective reduction material. Chem Commun, 2015, 51: 7610–7613
Adhikari AK, Lin KS. Improving CO2 adsorption capacities and CO2/N2 separation efficiencies of MOF-74(Ni, Co) by doping palladium-containing activated carbon. Chem Eng J, 2016, 284: 1348–1360
Zhu H, Yang X, Cranston ED, et al. Flexible and porous nano-cellulose aerogels with high loadings of metal-organic-framework particles for separations applications. Adv Mater, 2016, 28: 7652–7657
Kreno LE, Leong K, Farha OK, et al. Metal-organic framework materials as chemical sensors. Chem Rev, 2012, 112: 1105–1125
Lee JY, Farha OK, Roberts J, et al. Metal-organic framework materials as catalysts. Chem Soc Rev, 2009, 38: 1450–1459
Mondloch JE, Katz MJ, Isley III WC, et al. Destruction ofchemical warfare agents using metal-organic frameworks. Nat Mater, 2015, 14: 512–516
Zhou HC, Long JR, Yaghi OM. Introduction to metal-organic frameworks. Chem Rev, 2012, 112: 673–674
Wang X, Chen W, Zhang L, et al. Uncoordinated amine groups of metal-organic frameworks to anchor single Ru sites as chemose-lective catalysts toward the hydrogenation of quinoline. J Am Chem Soc, 2017, 139: 9419–9422
Zhao C, Dai X, Yao T, et al. Ionic exchange of metal-organic frameworks to access single nickel sites for efficient electroreduction of CO2. J Am Chem Soc, 2017, 139: 8078–8081
Zhang H, Zhao W, Zou M, et al. 3D, mutually embedded MOF@carbon nanotube hybrid networks for high-performance lithium-sulfur batteries. Adv Energy Mater, 2019, 9: 1805764
Zhang W, Wu ZY, Jiang HL, et al. Nanowire-directed templating synthesis of metal-organic framework nanofibers and their derived porous doped carbon nanofibers for enhanced electrocatalysis. J Am Chem Soc, 2014, 136: 14385–14388
Drobek M, Kim JH, Bechelany M, et al. MOF-based membrane encapsulated ZnO nanowires for enhanced gas sensor selectivity. ACS Appl Mater Interfaces, 2016, 8: 8323–8328
Zang X, Zhang X, Chang Q, et al. Metal-organic framework UiO-67-coated fiber for the solid-phase microextraction of nitrobenzene compounds from water. J Sep Sci, 2016, 39: 2770–2776
Hester P, Xu S, Liang W, et al. On thermal stability and catalytic reactivity of Zr-based metal-organic framework (UiO-67) encapsulated Pt catalysts. J Catal, 2016, 340: 85–94
Cavka JH, Jakobsen S, Olsbye U, et al. A new zirconium inorganic building brick forming metal organic frameworks with exceptional stability. J Am Chem Soc, 2008, 130: 13850–13851
Na K, Choi KM, Yaghi OM, et al. Metal nanocrystals embedded in single nanocrystals of MOFs give unusual selectivity as heterogeneous catalysts. Nano Lett, 2014, 14: 5979–5983
Qian HS, Yu SH, Gong JY, et al. High-quality luminescent tellurium nanowires of several nanometers in diameter and high aspect ratio synthesized by a poly(vinyl pyrrolidone)-assisted hydrothermal process. Langmuir, 2006, 22: 3830–3835
Huang Z, Zhou H, Chen Z, et al. Facile synthesis of porous Pt botryoidal nanowires and their electrochemical properties. Electrochim Acta, 2014, 147: 643–649
Hong W, Wang J, Wang E. Facile synthesis of PtCu nanowires with enhanced electrocatalytic activity. Nano Res, 2015, 8: 2308–2316
Cao X, Wang N, Jia S, et al. Detection of glucose based on bimetallic PtCu nanochains modified electrodes. Anal Chem, 2013, 85: 5040–5046
Mintsouli I, Georgieva J, Armyanov S, et al. Pt-Cu electrocatalysts for methanol oxidation prepared by partial galvanic replacement of Cu/carbon powder precursors. Appl Catal B-Environ, 2013, 136–137: 160–167
Fu S, Zhu C, Shi Q, et al. PtCu bimetallic alloy nanotubes with porous surface for oxygen reduction reaction. RSC Adv, 2016, 6: 69233–69238
Su L, Shrestha S, Zhang Z, et al. Platinum-copper nanotube electrocatalyst with enhanced activity and durability for oxygen reduction reactions. J Mater Chem A, 2013, 1: 12293–12301
Xu H, Li Y, Luo X, et al. Monodispersed gold nanoparticles supported on a zirconium-based porous metal-organic framework and their high catalytic ability for the reverse water-gas shift reaction. Chem Commun, 2017, 53: 7953–7956
Zhao Y, Zhang Q, Li Y, et al. Large-scale synthesis ofmonodisperse UiO-66 crystals with tunable sizes and missing linker defects via acid/base Co-modulation. ACS Appl Mater Interfaces, 2017, 9: 15079–15085
Masoomi MY, Beheshti S, Morsali A. Shape control of Zn(II) metal-organic frameworks by modulation synthesis and their morphology-dependent catalytic performance. Cryst Growth Des, 2015, 15: 2533–2538
Cai X, Lin J, Pang M. Facile synthesis of highly uniform Fe-MIL-88B particles. Cryst Growth Des, 2016, 16: 3565–3568
Lu G, Li S, Guo Z, et al. Imparting functionality to a metal-organic framework material by controlled nanoparticle encapsulation. Nat Chem, 2012, 4: 310–316
Keturakis CJ, Zhu M, Gibson EK, et al. Dynamics of CrO3-Fe2O3 catalysts during the high-temperature water-gas shift reaction: molecular structures and reactivity. ACS Catal, 2016, 6: 4786–4798
Cheng Z, Sherman BJ, Lo CS. Carbon dioxide activation and dissociation on ceria (110): A density functional theory study. J Chem Phys, 2013, 138: 014702
Daza YA, Kuhn JN. CO2 conversion by reverse water gas shift catalysis: comparison of catalysts, mechanisms and their consequences for CO2 conversion to liquid fuels. RSC Adv, 2016, 6: 49675–49691
Du C, Chen M, Wang W, et al. Platinum-based intermetallic nanotubes with a core-shell structure as highly active and durable catalysts for fuel cell applications. J Power Sources, 2013, 240: 630–635
Jiang Y, Jia Y, Zhang J, et al. Underpotential deposition-induced synthesis of composition-tunable Pt-Cu nanocrystals and their catalytic properties. Chem Eur J, 2013, 19: 3119–3124
Xu D, Liu Z, Yang H, et al. Solution-based evolution and enhanced methanol oxidation activity of monodisperse platinum-copper nanocubes. Angew Chem Int Ed, 2009, 48: 4217–4221
Yu X, Wang D, Peng Q, et al. Pt-M (M=Cu, Co, Ni, Fe) nano-crystals: from small nanoparticles to wormlike nanowires by oriented attachment. Chem Eur J, 2013, 19: 233–239
Gutterød ES, Øien-Ødegaard S, Bossers K, et al. CO2 hydrogenation over Pt-containing UiO-67 Zr-MOFs—the base case. Ind Eng Chem Res, 2017, 56: 13206–13218
Zhao X, Xu H, Wang XX, et al. Monodisperse metal-organic framework nanospheres with encapsulated core-shell nano-particles Pt/Au@Pd@{Co2(oba)4(3-bpdh)2}4H2O for the highly selective conversion of CO2 to CO. ACS Appl Mater Interfaces, 2018, 10: 15096–15103
Chen CS, Wu JH, Lai TW. Carbon dioxide hydrogenation on Cu nanoparticles. J Phys Chem C, 2010, 114: 15021–15028
Han Y, Xu H, Su Y, et al. Noble metal (Pt, Au@Pd) nanoparticles supported on metal organic framework (MOF-74) nanoshuttles as high-selectivity CO2 conversion catalysts. J Catal, 2019, 370: 70–78
Andersson MP, Abild-Pedersen F, Remediakis IN, et al. Structure sensitivity of the methanation reaction: H2-induced CO dissociation on nickel surfaces. J Catal, 2008, 255: 6–19
Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Foundation of China (21371058).
Author information
Authors and Affiliations
Contributions
Author contributions Zhang H performed the experiments, analyzed the data and wrote the draft of manuscript under the guidance of Xu H; Li Y, Pan X and Li L provided some suggestions.
Corresponding author
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Additional information
Huaqian Zhang is a master candidate at East China University of Science and Technology (ECUST). Her current research interest focuses on the synthesis of functional nanomaterials and CO2 conversion.
Haitao Xu obtained his Master and PhD degrees from the University of Science and Technology of China (USTC) in 1999 and 2002, respectively. After a postdoctoral fellowship at Tsinghua University, he became an associate professor at Tongji University in 2005. He studied in Kyushu University as a JSPS fellow in 2007. He moved to ECUST in 2009. His research focuses on multifunction materials, metal-organic frameworks, nanocatalyst, adsorption/separation, and CO2 conversion.
Supplementary Material
Rights and permissions
About this article
Cite this article
Zhang, H., Xu, H., Li, Y. et al. Nanocable catalysts MTe (M = Pt, PtCu)@UIO-67 for CO2 conversion. Sci. China Mater. 63, 769–778 (2020). https://doi.org/10.1007/s40843-019-1258-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-019-1258-8