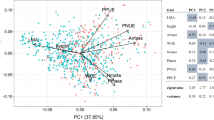
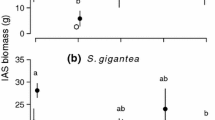
Abstract
Two contradictory hypotheses have been put forth to forecast alien invasiveness: being either functionally similar, or dissimilar, to resident natives along environmental gradients. The ‘try-harder’ hypothesis predicts that alien plants will be functionally dissimilar to natives and should thus exhibit exaggerated trait values when compared to natives in respect to resource extraction or stress tolerance. In contrast, the ‘join-the-locals’ hypothesis, which is compatible with “environmental filtering”, predicts functional similarities among alien and native species in richer, but not in resource-limited environments. Here, we propose a framework that links the successful alien plant strategy, i.e. being functionally similar or dissimilar to natives, to the harshness of the environment and the availability of resources. We tested these two hypotheses using a trait-environment dataset of 33 alien and 130 native plants in 96 sites covering a gradient of soil resources (organic matter, nitrogen, soil moisture) in Saint-Katherine, an arid protected area in Egypt. We estimated 18 interaction coefficients between three candidate traits (specific leaf area, above-ground biomass, height) of alien and native plants as well as soil resources using linear mixed-effects models. Additionally, we calculated the mean and the hierarchical functional distance among aliens and natives along soil gradients. Our results revealed that in extreme resource-limited environments, aliens and natives were functionally similar and had relatively equal trait values consistent with environmental filtering, thus supporting the ‘join-the-locals’ hypothesis. However, in environments richer in resources, aliens and natives were functionally dissimilar with aliens exhibiting more exploitative trait values (taller, higher SLA and biomass) than natives, providing support for the ‘try-harder’ hypothesis. While demonstrated in only one arid system, results suggest that linking soil resource availability with functional divergence and convergence among native and alien plants could be used as an informative strategy to predict alien invasiveness. Hence, future studies should investigate the functional response of alien and native plants in different environments against different resource gradients to test for the generality of the patterns we found.




Similar content being viewed by others
References
Abrams P (1983) The theory of limiting similarity. Annu Rev Ecol Syst 14:359–376
Aerts R (1999) Interspecific competition in natural plant communities: mechanisms, trade-offs and plant–soil feedbacks. J Exp Bot 50:29–37. https://doi.org/10.1093/jxb/50.330.29
Aerts R, Chapin III FS (1999) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. In: Advances in ecological research, vol 30. Academic Press, pp 1–67
Allen SE, Grimshaw HM, Parkinson JA, Quarmby C (1974) Chemical analysis of ecological materials. Blackwell, Hoboken
Allen CR, Nemec KT, Wardwell DA et al (2013) Predictors of regional establishment success and spread of introduced non-indigenous vertebrates. Glob Ecol Biogeogr 22:889–899
Ayyad MA, Fakhry AM, Moustafa ARA (2000) Plant biodiversity in the Saint Catherine area of the Sinai peninsula, Egypt. Biodivers Conserv 9(2):265–281
Bartoń K (2016) MuMIn: multi-model inference. R package version 1.15.6. https://CRAN.R-project.org/package=MuMIn
Basuki TM, Van Laake PE, Skidmore AK, Hussin YA (2009) Allometric equations for estimating the above-ground biomass in tropical lowland Dipterocarp forests. For Ecol Manag 257:1684–1694
Bates D, Maechler M, Bolker B, Walker S (2015) lme4: linear mixed-effects models using Eigen and S4. R package version 1.1–7. 2014
Blomberg SP, Garland T Jr, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–745
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889. https://doi.org/10.2307/2261425
Blumenthal D (2005) Interrelated causes of plant invasion. Science 310:243–244. https://doi.org/10.1126/science.1114851
Blumenthal DM (2006) Interactions between resource availability and enemy release in plant invasion. Ecol Lett 9:887–895
Bullock J, Joe F, Mark JS et al (2001) A plant trait analysis of responses to grazing in a long-term experiment. J Appl Ecol 38:253–267. https://doi.org/10.1046/j.1365-2664.2001.00599.x
Burns JH (2006) Relatedness and environment affect traits associated with invasive and noninvasive introduced Commelinaceae. Ecol Appl 16:1367–1376. https://doi.org/10.1890/1051-0761(2006)016%5b1367:RAEATA%5d2.0.CO;2
Cadotte MW, Campbell SE, Li S et al (2018) Preadaptation and naturalization of non-native species: Darwin’s two fundamental insights into species invasion. Annu Rev Plant Biol 69:661–684. https://doi.org/10.1146/annurev-arplant-042817-040339
Carboni M, Calderon-Sanou I, Pollock L et al (2018) Functional traits modulate the response of alien plants along abiotic and biotic gradients. Glob Ecol Biogeogr 27:1173–1185. https://doi.org/10.1111/geb.12775
Catford JA, Jansson R, Nilsson C (2009) Reducing redundancy in invasion ecology by integrating hypotheses into a single theoretical framework. Divers Distrib 15:22–40. https://doi.org/10.1111/j.1472-4642.2008.00521.x
Cavender-Bares J, Ackerly DD, Baum DA, Bazzaz FA (2004) Phylogenetic overdispersion in Floridian oak communities. Am Nat 163:823–843. https://doi.org/10.1086/386375
Chapin FS III (1980) The mineral nutrition of wild plants. Annu Rev Ecol Syst 11:233–260
Conti L, Block S, Parepa M et al (2018) Functional trait differences and trait plasticity mediate biotic resistance to potential plant invaders. J Ecol 106:1607–1620
Craine JM (2009) Resource strategies of wild plants. Princeton University Press, Princeton
Crawley MJ, Harvey PH, Purvis A (1996) Comparative ecology of the native and alien floras of the British Isles. Philos Trans R Soc B 351:1251–1259. https://doi.org/10.1098/rstb.1996.0108
Daehler CC (2001) Darwin’s naturalization hypothesis revisited. Am Nat 158:324–330
Daehler CC (2003) Performance comparisons of co-occurring native and alien invasive plants: implications for conservation and restoration. Annu Rev Ecol Evol Syst 34:183–211
Davies TJ (2006) Evolutionary ecology: when relatives cannot live together. Evol Ecol 16:R645–R647
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534
Dawson W, Burslem DF, Hulme PE (2011) The comparative importance of species traits and introduction characteristics in tropical plant invasions. Divers Distrib 17:1111–1121
Dawson W, Rohr RP, van Kleunen M, Fischer M (2012) Alien plant species with a wider global distribution are better able to capitalize on increased resource availability. New Phytol 194:859–867
Duncan RP, Williams PA (2002) Ecology: Darwin’s naturalization hypothesis challenged. Nature 417:608
Dyderski MK, Jagodziński AM (2019) Functional traits of acquisitive invasive woody species differ from conservative invasive and native species. NeoBiota 41:91–113. https://doi.org/10.3897/neobiota.41.31908
Feng Y-L, Lei Y-B, Wang R-F et al (2009) Evolutionary tradeoffs for nitrogen allocation to photosynthesis versus cell walls in an invasive plant. Proc Natl Acad Sci USA 106:1853–1856
Funk JL, Vitousek PM (2007) Resource-use efficiency and plant invasion in low-resource systems. Nature 446:1079–1081. https://doi.org/10.1038/nature05719
Funk JL, Cleland EE, Suding KN, Zavaleta ES (2008) Restoration through reassembly: plant traits and invasion resistance. Trends Ecol Evol 23:695–703
Funk JL, Glenwinkel LA, Sack L (2013) Differential allocation to photosynthetic and non-photosynthetic nitrogen fractions among native and invasive species. PLoS ONE 8:e64502
Funk JL, Standish RJ, Stock WD, Valladares F (2016) Plant functional traits of dominant native and invasive species in mediterranean-climate ecosystems. Ecology 97:75–83
Gallien L, Carboni M (2017) The community ecology of invasive species: where are we and what’s next? Ecography 40:335–352. https://doi.org/10.1111/ecog.02446
Gallien L, Mazel F, Lavergne S et al (2015) Contrasting the effects of environment, dispersal and biotic interactions to explain the distribution of invasive plants in alpine communities. Biol Invasions 17:1407–1423. https://doi.org/10.1007/s10530-014-0803-1
Gallien L, Zimmermann NE, Levine JM, Adler PB (2017) The effects of intransitive competition on coexistence. Ecol Lett 20:791–800
Gaudet CL, Keddy PA (1988) A comparative approach to predicting competitive ability from plant traits. Nature 334:242. https://doi.org/10.1038/334242a0
González AL, Kominoski JS, Danger M et al (2010) Can ecological stoichiometry help explain patterns of biological invasions? Oikos 119:779–790
González-Suárez M, Bacher S, Jeschke JM (2015) Intraspecific trait variation is correlated with establishment success of alien mammals. Am Nat 185:737–746. https://doi.org/10.1086/681105
Grime JP (2006) Plant strategies, vegetation processes, and ecosystem properties. Wiley, Hoboken
Grotkopp E, Rejmánek M (2007) High seedling relative growth rate and specific leaf area are traits of invasive species: phylogenetically independent contrasts of woody angiosperms. Am J Bot 94:526–532
Heberling JM, Fridley JD (2013) Resource-use strategies of native and invasive plants in Eastern North American forests. New Phytol 200:523–533
Holeksa J, Woźniak G (2005) Biased vegetation patterns and detection of vegetation changes using phytosociological databases: a case study in the forests of the Babia Góra National Park (the West Carpathians, Poland). Phytocoenologia 35(1):1–18
Jauni M, Hyvönen T (2012) Interactions between alien plant species traits and habitat characteristics in agricultural landscapes in Finland. Biol Invasions 14:47–63. https://doi.org/10.1007/s10530-011-0058-z
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends Ecol Evol 19:101–108. https://doi.org/10.1016/j.tree.2003.10.013
Kembel SW, Hubbell SP (2006) The phylogenetic structure of a neotropical forest tree community. Ecology 87:S86–S99
Klute A (1986) Methods of soil analysis. No 9 Part 1, physical and mineralogical methods. American Society of Agronomy Madison, Wisconsin
Kunstler G, Lavergne S, Courbaud B et al (2012) Competitive interactions between forest trees are driven by species’ trait hierarchy, not phylogenetic or functional similarity: implications for forest community assembly. Ecol Lett 15:831–840. https://doi.org/10.1111/j.1461-0248.2012.01803.x
Lake JC, Leishman MR (2004) Invasion success of exotic plants in natural ecosystems: the role of disturbance, plant attributes and freedom from herbivores. Biol Conserv 117:215–226. https://doi.org/10.1016/S0006-3207(03)00294-5
Laliberté E, Shipley B, Norton DA, Scott D (2012) Which plant traits determine abundance under long-term shifts in soil resource availability and grazing intensity? J Ecol 100:662–677
Leffler AJ, James JJ, Monaco TA, Sheley RL (2014) A new perspective on trait differences between native and invasive exotic plants. Ecology 95:298–305. https://doi.org/10.1890/13-0102.1
Leishman MR, Thomson VP (2005) Experimental evidence for the effects of additional water, nutrients and physical disturbance on invasive plants in low fertility Hawkesbury Sandstone soils, Sydney, Australia. J Ecol 93:38–49. https://doi.org/10.1111/j.1365-2745.2004.00938.x
Leishman MR, Haslehurst T, Ares A, Baruch Z (2008) Leaf trait relationships of native and invasive plants: community- and global-scale comparisons. New Phytol. https://doi.org/10.1111/j.1469-8137.2007.02189.x
Leishman MR, Thomson VP, Cooke J (2010) Native and exotic invasive plants have fundamentally similar carbon capture strategies: leaf trait relationships of natives and exotic invasives. J Ecol 98:28–42. https://doi.org/10.1111/j.1365-2745.2009.01608.x
Lepš J, Hadincová V (1992) How reliable are our vegetation analyses? J Veg Sci 3(1):119–124
Li S, Cadotte MW, Meiners SJ et al (2015) The effects of phylogenetic relatedness on invasion success and impact: deconstructing Darwin’s naturalisation conundrum. Ecol Lett 18:1285–1292. https://doi.org/10.1111/ele.12522
Macarthur R, Levins R (1967) The limiting similarity, convergence, and divergence of coexisting species. Am Nat 101:377–385. https://doi.org/10.1086/282505
MacDougall AS, Gilbert B, Levine JM (2009) Plant invasions and the niche. J Ecol 97:609–615. https://doi.org/10.1111/j.1365-2745.2009.01514.x
Maitner BS, Rudgers JA, Dunham AE, Whitney KD (2012) Patterns of bird invasion are consistent with environmental filtering. Ecography 35:614–623. https://doi.org/10.1111/j.1600-0587.2011.07176.x
Mayfield MM, Levine JM (2010) Opposing effects of competitive exclusion on the phylogenetic structure of communities: phylogeny and coexistence. Ecol Lett 13:1085–1093. https://doi.org/10.1111/j.1461-0248.2010.01509.x
Nakagawa S, Schielzeth H (2013) A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol Evol 4:133–142
Onoda Y, Hikosaka K, Hirose T (2004) Allocation of nitrogen to cell walls decreases photosynthetic nitrogen-use efficiency. Funct Ecol 18:419–425. https://doi.org/10.1111/j.0269-8463.2004.00847.x
Ordonez A, Olff H (2013) Do alien plant species profit more from high resource supply than natives? A trait-based analysis: Alien-native leaf trait differences. Glob Ecol Biogeogr 22:648–658. https://doi.org/10.1111/geb.12019
Park DS, Potter D (2015) A reciprocal test of Darwin’s naturalization hypothesis in two Mediterranean-climate regions. Glob Ecol Biogeogr 24:1049–1058. https://doi.org/10.1111/geb.12352
Penuelas J, Sardans J, Llusià J et al (2010) Faster returns on ‘leaf economics’ and different biogeochemical niche in invasive compared with native plant species. Glob Change Biol 16:2171–2185. https://doi.org/10.1111/j.1365-2486.2009.02054.x
Pérez-Harguindeguy N, Diaz S, Gamier E et al (2013) New handbook for stand-ardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234
Pollock LJ, Morris WK, Vesk PA (2012) The role of functional traits in species distributions revealed through a hierarchical model. Ecography 35:716–725. https://doi.org/10.1111/j.1600-0587.2011.07085.x
Poorter H, Evans JR (1998) Photosynthetic nitrogen-use efficiency of species that differ inherently in specific leaf area. Oecologia 116:26–37. https://doi.org/10.1007/s004420050560
Poorter H, Nagel O (2000) The role of biomass allocation in the growth response of plants to different levels of light, CO2, nutrients and water: a quantitative review. Funct Plant Biol 27:1191. https://doi.org/10.1071/pp99173_co
Prinzing A, Durka W, Klotz S, Brandl R (2002) Which species become aliens? Evol Ecol Res 4:385–405
Pyšek P, Richardson DM (2007) Traits associated with invasiveness in alien plants: where do we stand? In: Nentwig W (ed) Biological invasions. Springer, Berlin, pp 97–125
Pyšek P, Prach K, Smilauer P (1995) Relating invasion success to plant traits: an analysis of the Czech alien flora. In: Pyšek P, Prach K, Rejmánek M, Wade M (eds) Plant invasions: general aspects and special problems. SPB Academic Publishers, Amsterdam, pp 39–60
Pyšek P, Jarošík V, Chytrý M et al (2005) Alien plants in temperate weed communities: prehistoric and recent invaders occupy different habitats. Ecology 86:772–785. https://doi.org/10.1890/04-0012
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rejmánek M, Richardson DM, Pyšek P (2005) Plant invasions and invasibility of plant communities. Veg Ecol 20:332–355
Revell LJ (2010) Phylogenetic signal and linear regression on species data. Methods Ecol Evol 1:319–329. https://doi.org/10.1111/j.2041-210X.2010.00044.x
Revell LJ (2012) Phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol Evol 3:217–223. https://doi.org/10.1111/j.2041-210X.2011.00169.x
Richardson DM, Pyšek P (2006) Plant invasions: merging the concepts of species invasiveness and community invasibility. Prog Phys Geogr 30:409–431
Rose KE, Atkinson RL, Turnbull LA, Rees M (2009) The costs and benefits of fast living. Ecol Lett 12:1379–1384
Schielzeth H (2010) Simple means to improve the interpretability of regression coefficients. Methods Ecol Evol 1:103–113. https://doi.org/10.1111/j.2041-210X.2010.00012.x
Schieving F, Poorter H (1999) Carbon gain in a multispecies canopy: the role of specific leaf area and photosynthetic nitrogen-use efficiency in the tragedy of the commons. New Phytol 143(1):201–211. https://doi.org/10.1046/j.1469-8137.1999.00431.x
Takashima T, Hikosaka K, Hirose T (2004) Photosynthesis or persistence: nitrogen allocation in leaves of evergreen and deciduous Quercus species. Plant, Cell Environ 27:1047–1054. https://doi.org/10.1111/j.1365-3040.2004.01209.x
Tecco PA, DÃ-az S, Cabido M, Urcelay C (2010) Functional traits of alien plants across contrasting climatic and land-use regimes: do aliens join the locals or try harder than them? Alien traits across climates and land uses. J Ecol 98:17–27. https://doi.org/10.1111/j.1365-2745.2009.01592.x
Tecco PA, Urcelay C, Díaz S et al (2013) Contrasting functional trait syndromes underlay woody alien success in the same ecosystem: contrasting syndromes among coexisting aliens. Aust Ecol 38:443–451. https://doi.org/10.1111/j.1442-9993.2012.02428.x
Thuiller W, Gallien L, Boulangeat I et al (2010) Resolving Darwin’s naturalization conundrum: a quest for evidence. Divers Distrib 16:461–475. https://doi.org/10.1111/j.1472-4642.2010.00645.x
van Kleunen M, Weber E, Fischer M (2010) A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol Lett 13:235–245. https://doi.org/10.1111/j.1461-0248.2009.01418.x
Vitousek P (1982) Nutrient cycling and nutrient use efficiency. Am Nat 119:553–572. https://doi.org/10.1086/283931
Warren CR, Adams MA (2001) Distribution of N, Rubisco and photosynthesis in Pinus pinaster and acclimation to light. Plant, Cell Environ 24:597–609
Weiher E, Keddy PA (1995) Assembly rules, null models, and trait dispersion: new questions from old patterns. Oikos 74(1):159–164
Westoby M (1998) A leaf-height-seed (LHS) plant ecology strategy scheme. Plant Soil 199:213–227
Westoby M, Falster DS, Moles AT et al (2002) Plant ecological strategies: some leading dimensions of variation between species. Annu Rev Ecol Syst 33:125–159. https://doi.org/10.1146/annurev.ecolsys.33.010802.150452
Wright IJ, Reich PB, Westoby M et al (2004) The worldwide leaf economics spectrum. Nature 428:821. https://doi.org/10.1038/nature02403
Acknowledgements
The study was financed by Swiss Government Excellence Scholarship (2018.0730). We are grateful to Caroline Curtis and Giovanni Vimercati (Biology Dept., University of Fribourg) for their comments on earlier drafts of the manuscript.
Funding
Funding was provided by Office Fédéral de l’Education et de la Science (ESKAS 2018.07.30).
Author information
Authors and Affiliations
Contributions
RFE conceived the approach with substantial contributions from SB. IA and AK collected and compiled the datasets. RFE carried out the analyses with advice from RR. RFE wrote the first draft with substantial contributions of SB. AFP helped with the writing of the manuscript. All authors gave final approval for publication.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix: Estimation of native SLA and biomass data using allometric equations
Appendix: Estimation of native SLA and biomass data using allometric equations
Because there was no allowance of destructively measuring native biomass and SLA in SKP, we estimated the above-ground biomass and SLA of native plants using allometric equations (Basuki et al. 2009). First, we used four non-destructive traits directly measured in the field per native plant individual. These traits were plant height (cm), leaf area (cm2) (drawn in the field then scanned using IMAGEJ), number of leaves and number of flowers and fruits. Second, we constructed series of multivariate regression models to calibrate a predictive model of aliens above-ground biomass and SLA as a function of their height (cm), number of leaves, flowers, fruits and leaf area (cm2). Then, we validated the predictive power of this model through model selection criteria using Akaike Information Criterion (AIC) and Akaike Weights (AW) in addition to diagnostic regression plots (e.g. QQ plots) to infer the best regression models. Additionally, we found high correlation between the actual observed values of biomass and SLA and the predicted values from the best regression models. Based on this validation, the best fitted model was used to predict natives’ biomass and SLA in each stand based on their empirical, field-measured trait values (see Table 3).
Initially, we considered using biomass and SLA data from global databases such as TRY or LEDA, because trait values were found for only 24 native species (out of 133). Likewise, database trait values of these native species that could also be measured non-destructively were much larger compared to the ones that were measured in the field. This difference might indicate the effects of the hyper arid conditions prevailing in Saint-Katherine. We also considered imputing missing values from less than 20% of the native species as extremely unreliable. Thus, we refrained from using trait values derived from databases and believe that the field - measured trait values (and their allometric derivatives) are more representative and relevant for this study.
Rights and permissions
About this article
Cite this article
El-Barougy, R.F., Elgamal, I., Rohr, R.P. et al. Functional similarity and dissimilarity facilitate alien plant invasiveness along biotic and abiotic gradients in an arid protected area. Biol Invasions 22, 1997–2016 (2020). https://doi.org/10.1007/s10530-020-02235-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02235-3