Abstract
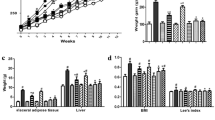
Alginate oligosaccharides are associated with some beneficial health effects. Gut microbiota is one of the most recently identified factors in the development of several metabolic diseases induced by high-fat diet. Our objective was to evaluate how alginate oligosaccharides impact on high-fat diet‑induced features of metabolic disorders and whether this impact is related to modulations in the modulation of the gut microbiota. C57BL/6J mice were fed with chow diet, high-fat diet, or high-fat diet supplemented with alginate oligosaccharides for 10 weeks. Alginate oligosaccharide treatment improved lipid metabolism, such as reducing levels of TG and LDL-C and inhibiting expression of lipogenesis genes. Alginate oligosaccharide administration reduced the levels of fasting blood glucose and increased the levels of serum insulin. Alginate oligosaccharide treatment was found to lower the expression of markers of inflammation, including IL1β and CD11c. Alginate oligosaccharide treatment modulated gut microbial communities and markedly prompted the growth of Akkermansia muciniphila, Lactobacillus reuteri, and Lactobacillus gasseri. Additionally, alginate oligosaccharide intervention significantly increased concentrations of short-chain fatty acids, such as acetic acid, propionic acid, and butyric acid, as well as decreased levels of endotoxin. Alginate oligosaccharides exert beneficial effects via alleviating metabolic metrics induced by high-fat diet, which is associated with increase in A. muciniphila, L. reuteri, and L. gasseri, as well as the release of microbiota-dependent short-chain fatty acids and inhibition of endotoxin levels.









Similar content being viewed by others
References
Al-Lahham SH, Peppelenbosch MP, Roelofsen H, Vonk RJ, Venema K (2010) Biological effects of propionic acid in humans; metabolism, potential applications and underlying mechanisms. Bba-Mol Cell Biol L 1801(11):1175–1183. https://doi.org/10.1016/j.bbalip.2010.07.007
Anhê FF, Roy D, Pilon G (2015) A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 64(4):872–883. https://doi.org/10.1136/gutjnl-2014-307142
Chakraborti CK (2015) New-found link between microbiota and obesity. World J Gastrointest Pathophysiol 6(4):110–119. https://doi.org/10.4291/wjgp.v6.i4.110
Chen YT, Yang NS, Lin YC, Ho ST, Li KY, Lin JS, Liu JR, Chen MJ (2018) A combination of Lactobacillus mali APS1 and dieting improved the efficacy of obesity treatment via manipulating gut microbiome in mice. Sci Rep 8(1):6153. https://doi.org/10.1038/s41598-018-23844-y
Cox AJ, West NP, Cripps AW (2015) Obesity, inflammation, and the gut microbiota. Lancet Diabetes Endocrinol 3(3):207–215. https://doi.org/10.1016/S2213-8587(14)70134-2
de la Cuesta-Zuluaga J, Mueller NT, Corrales-Agudelo V, Velasquez-Mejia EP, Carmona JA, Abad JM, Escobar JS (2017) Metformin is associated with higher relative abundance of mucin-degrading Akkermansia muciniphila and several short-chain fatty acid-producing microbiota in the gut. Diabetes Care 40(1):54–62. https://doi.org/10.2337/dc16-1324
den Besten G, Bleeker A, Gerding A, van Eunen K, Havinga R, van Dijk TH, Oosterveer MH, Jonker JW, Groen AK, Reijngoud DJ, Bakker BM (2015) Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes 64(7):2398–2408. https://doi.org/10.2337/db14-1213
Depommier C, Everard A, Druart C, Plovier H, Van Hul M, Vieira-Silva S, Falony G, Raes J, Maiter D, Delzenne NM, de Barsy M, Loumaye A, Hermans MP, Thissen JP, de Vos WM, Cani PD (2019) Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med 25(7):1096–1103. https://doi.org/10.1038/s41591-019-0495-2
Everard A, Belzer C, Geurts L, Ouwerkerk JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne NM, de Vos WM, Cani PD (2013) Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. P Natl Acad Sci USA 110(22):9066–9071. https://doi.org/10.1073/pnas.1219451110
Everard A, Lazarevic V, Derrien M, Girard M, Muccioli GG, Neyrinck AM, Possemiers S, Van Holle A, Francois P, de Vos WM, Delzenne NM, Schrenzel J, Cani PD (2011) Responses of gut microbiota and glucose and lipid metabolism to prebiotics in genetic obese and diet-induced leptin-resistant mice. Diabetes 60(12):3307–3307. https://doi.org/10.2337/db11-0227
Fak F, Backhed F (2012) Lactobacillus reuteri prevents diet-induced obesity, but not atherosclerosis, in a strain dependent fashion in Apoe−/− mice. PLoS One 7(10):e46837. https://doi.org/10.1371/journal.pone.0046837
Gupta S, Lokesh J, Abdelhafiz Y, Siriyappagouder P, Pierre R, Sorensen M, Fernandes JMO, Kiron V (2019) Macroalga-derived alginate oligosaccharide alters intestinal bacteria of Atlantic salmon. Front Microbiol 10:2037. https://doi.org/10.3389/fmicb.2019.02037
Han F, Wang Y, Han Y, Zhao J, Han F, Song G, Jiang P, Miao H (2018) Effects of whole-grain rice and wheat on composition of gut microbiota and short-chain fatty acids in rats. J Agric Food Chem 66(25):6326–6335. https://doi.org/10.1021/acs.jafc.8b01891
Han ZL, Yang M, Fu XD, Chen M, Su Q, Zhao YH, Mou HJ (2019) Evaluation of prebiotic potential of three marine algae oligosaccharides from enzymatic hydrolysis. Mar Drugs 17(3). https://doi.org/10.3390/md17030173
Hong J, Jia YM, Pan S, Jia LF, Li HF, Han ZQ, Cai DM, Zhao RQ (2016) Butyrate alleviates high fat diet-induced obesity through activation of adiponectin-mediated pathway and stimulation of mitochondrial function in the skeletal muscle of mice. Oncotarget 7(35):56071–56075. https://doi.org/10.18632/oncotarget.112676082
Juarez GE, Villena J, Salva S, de Valdez GF, Rodriguez AV (2013) Lactobacillus reuteri CRL1101 beneficially modulate lipopolysaccharide-mediated inflammatory response in a mouse model of endotoxic shock. J Funct Foods 5(4):1761–1773. https://doi.org/10.1016/j.jff.2013.08.002
Kadooka Y, Sato M, Imaizumi K, Ogawa A, Ikuyama K, Akai Y, Okano M, Kagoshima M, Tsuchida T (2010) Regulation of abdominal adiposity by probiotics (Lactobacillus gasseri SBT2055) in adults with obese tendencies in a randomized controlled trial. Eur J Clin Nutr 64(6):636–643. https://doi.org/10.1038/ejcn.2010.19
Kawano M, Miyoshi M, Ogawa A, Sakai F, Kadooka Y (2016) Lactobacillus gasseri SBT2055 inhibits adipose tissue inflammation and intestinal permeability in mice fed a high-fat diet. J Nutr Sci 5:e23. https://doi.org/10.1017/jns.2016.12
Kim J, Yun JM, Kim MK, Kwon O, Cho B (2018) Lactobacillus gasseri BNR17 supplementation reduces the visceral fat accumulation and waist circumference in obese adults: a randomized, double-blind, placebo-controlled trial. J Med Food 21(5):454–461. https://doi.org/10.1089/jmf.2017.3937
Kumar MV, Shimokawa T, Nagy TR, Lane MD (2002) Differential effects of a centrally acting fatty acid synthase inhibitor in lean and obese mice. Proc Natl Acad Sci U S A 99(4):1921–1925. https://doi.org/10.1073/pnas.042683699
Li M, van Esch B, Henricks PAJ, Folkerts G, Garssen J (2018) The anti-inflammatory effects of short chain fatty acids on lipopolysaccharide- or tumor necrosis factor alpha-stimulated endothelial cells via activation of GPR41/43 and inhibition of HDACs. Front Pharmacol 9:533. https://doi.org/10.3389/fphar.2018.00533
Long T, Yu ZJ, Wang J, Liu J, He BS (2018) Orally administered chitooligosaccharides modulate colon microbiota in normal and colitis mice. Int J Pharmacol 14(2):291–300. https://doi.org/10.3923/ijp.2018.291.300
Ma DL, Forsythe P, Bienenstock J (2004) Live Lactobacillus reuteri is essential for the inhibitory effect on tumor necrosis factor alpha-induced interleukin-8 expression. Infect Immun 72(9):5308–5314. https://doi.org/10.1128/Iai.72.9.5308-5314.2004
Makki K, Froguel P, Wolowczuk I (2013) Adipose tissue in obesity-related inflammation and insulin resistance: cells, cytokines, and chemokines. ISRN Inflamm 2013:139239. https://doi.org/10.1155/2013/139239
McNabney SM, Henagan TM (2017) Short chain fatty acids in the colon and peripheral tissues: a focus on butyrate, colon cancer, obesity and insulin resistance. Nutrients 9(12). https://doi.org/10.3390/nu9121348
Mehrpouya-Bahrami P, Chitrala KN, Ganewatta MS, Tang C, Murphy EA, Enos RT, Velazquez KT, McCellan J, Nagarkatti M, Nagarkatti P (2017) Blockade of CB1 cannabinoid receptor alters gut microbiota and attenuates inflammation and diet-induced obesity. Sci Rep 7(1):15645–15660. https://doi.org/10.1038/s41598-017-15154-6
Melo AD, Silveira H, Bortoluzzi C, Lara LJ, Garbossa CA, Preis G, Costa LB, Rostagno MH (2016) Intestinal alkaline phosphatase and sodium butyrate may be beneficial in attenuating LPS-induced intestinal inflammation. Genet Mol Res 15(4):1–10. https://doi.org/10.4238/gmr15048875
Piazzon MC, Calduch-Giner JA, Fouz B, Estensoro I, Simo-Mirabet P, Puyalto M, Karalazos V, Palenzuela O, Sitja-Bobadilla A, Perez-Sanchez J (2017) Under control: how a dietary additive can restore the gut microbiome and proteomic profile, and improve disease resilience in a marine teleostean fish fed vegetable diets. Microbiome 5(164):1–23. https://doi.org/10.1186/s40168-017-0390-3
Rogero MM, Calder PC (2018) Obesity, inflammation, toll-like receptor 4 and fatty acids. Nutrients 10(4):432. https://doi.org/10.3390/nu10040432
Schachter J, Martel J, Lin CS, Chang CJ, Wu TR, Lu CC, Ko YF, Lai HC, Ojcius DM, Young JD (2018) Effects of obesity on depression: a role for inflammation and the gut microbiota. Brain Behav Immun 69:1–8. https://doi.org/10.1016/j.bbi.2017.08.026
Shi L, Li M, Miyazawa K, Li Y, Hiramatsu M, Xu J, Gong C, Jing X, He F, Huang C (2013) Effects of heat-inactivated Lactobacillus gasseri TMC0356 on metabolic characteristics and immunity of rats with the metabolic syndrome. Br J Nutr 109(2):263–272. https://doi.org/10.1017/S000711451200116X
Shimizu T, Mori K, Ouchi K, Kushida M, Tsuduki T (2018) Effects of dietary intake of japanese mushrooms on visceral fat accumulation and gut microbiota in mice. Nutrients 10(5). https://doi.org/10.3390/nu10050610
Simon MC, Strassburger K, Nowotny B, Kolb H, Nowotny P, Burkart V, Zivehe F, Hwang JH, Stehle P, Pacini G, Hartmann B, Holst JJ, MacKenzie C, Bindels LB, Martinez I, Walter J, Henrich B, Schloot NC, Roden M (2015) Intake of Lactobacillus reuteri improves incretin and insulin secretion in glucose-tolerant humans: a proof of concept. Diabetes Care 38(10):1827–1834. https://doi.org/10.2337/dc14-2690
Terakado S, Ueno M, Tamura Y, Toda N, Yoshinaga M, Otsuka K, Numabe A, Kawabata Y, Murota I, Sato N, Uehara Y (2012) Sodium alginate oligosaccharides attenuate hypertension and associated kidney damage in dahl salt-sensitive rats fed a high-salt diet. Clin Exp Hypertens 34(2):99–106. https://doi.org/10.3109/10641963.2011.618196
Thupari JN, Landree LE, Ronnett GV, Kuhajda FP (2002) C75 increases peripheral energy utilization and fatty acid oxidation in diet-induced obesity. Proc Natl Acad Sci U S A 99(14):9498–9502. https://doi.org/10.1073/pnas.132128899
Ueno M, Tamura Y, Toda N, Yoshinaga M, Terakado S, Otsuka K, Numabe A, Kawabata Y, Murota I, Sato N, Uehara Y (2012) Sodium alginate oligosaccharides attenuate hypertension in spontaneously hypertensive rats fed a low-salt diet. Clin Exp Hypertens 34(5):305–310. https://doi.org/10.3109/10641963.2011.577484
Wang J, Qin C, He T, Qiu K, Sun W, Zhang X, Jiao N, Zhu W, Yin J (2018) Alfalfa-containing diets alter luminal microbiota structure and short chain fatty acid sensing in the caecal mucosa of pigs. J Anim Sci Biotechnol 9:11. https://doi.org/10.1186/s40104-017-0216-y
Wang Z, Bao ZJ (2017) Effect of anti-gut inflammatory agent on insulin resistance and lipid profile of mice fed different diets. Trop J Pharm Res 16(11):2651–2658. https://doi.org/10.4314/tjpr.v16i11.12
Yan SK, Shi RJ, Li L, Ma SB, Zhang HB, Ye J, Wang JM, Pan JR, Wang QX, Jin X, Liu XB, Liu ZG (2019) Mannan oligosaccharide suppresses lipid accumulation and appetite in western-diet-induced obese mice via reshaping gut microbiome and enhancing short-chain fatty acids production. Mol Nutr Food Res 63(23):e1900521. https://doi.org/10.1002/mnfr.201900521
Yin J, Li Y, Han H, Chen S, Gao J, Liu G, Wu X, Deng J, Yu Q, Huang X, Fang R, Li T, Reiter RJ, Zhang D, Zhu C, Zhu G, Ren W, Yin Y (2018) Melatonin reprogramming of gut microbiota improves lipid dysmetabolism in high fat diet-fed mice. J Pineal Res 65:e12524. https://doi.org/10.1111/jpi.12524
Zhai SX, Song Q, Li LL, Zhu LM, Zou ZQ, Wang L (2019) Dietary butyrate suppresses inflammation through modulating gut microbiota in high-fat diet-fed mice. FEMS Microbiol Lett 366(13):1–7. https://doi.org/10.1093/femsle/fnz153
Zhang L, Qin QQ, Liu MN, Zhang XL, He F, Wang GQ (2018) Akkermansia muciniphila can reduce the damage of gluco/lipotoxicity, oxidative stress and inflammation, and normalize intestine microbiota in streptozotocin-induced diabetic rats. Pathog Dis 76(4). https://doi.org/10.1093/femspd/fty028
Zhou R, Shi X, Gao Y, Cai N, Jiang Z, Xu X (2015) Anti-inflammatory activity of guluronate oligosaccharides obtained by oxidative degradation from alginate in lipopolysaccharide-activated murine macrophage RAW 264.7 cells. J Agric Food Chem 63(1):160–168. https://doi.org/10.1021/jf503548a
Funding
This work was supported by the National Key R&D Program of China (2017YFC0506200) and Youth Innovation Promotion Association of Chinese Academy of Sciences (2018246).
Author information
Authors and Affiliations
Contributions
YW, LL, and SQ designed the research. YW and JY were responsible for the execution of the study, data collection, and analysis. YW, LL, and SQ interpreted the data. YW, LL, and CY played major roles in drafting, writing, and revising this manuscript. All authors have known and agreed to this final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Permissions for this animal procedure had been requested and approved from the Institutional Animal Care and Use Committee of Binzhou Medical University.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 634 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Li, L., Ye, C. et al. Alginate oligosaccharide improves lipid metabolism and inflammation by modulating gut microbiota in high-fat diet fed mice. Appl Microbiol Biotechnol 104, 3541–3554 (2020). https://doi.org/10.1007/s00253-020-10449-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10449-7