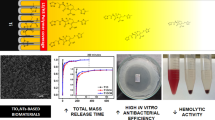
Abstract
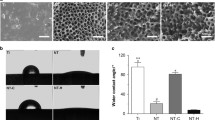
The anodized titanium nanotubes (TiO2-NTs) are considered to be a potential material in clinical therapy. To enhance the antibacterial property of TiO2-NTs, cefuroxime is introduced into TiO2-NTs, and then, different chitosan layers are coated to control the release of cefuroxime. SEM and FTIR are adapted for the characterization of prepared TiO2-NTs. The effects of chitosan coating thickness on release of cefuroxime are also investigated, followed with the antibacterial property evaluation. The results show TiO2-NTs are fabricated by anodization method and cefuroxime is also successfully loaded into the nanotubes. The thickness of chitosan coating is an important factor to the release rate of cefuroxime. Antimicrobial detection and morphology observation of S. aureus show a sustained 7-day drug release and strong negative effect on bacteria. The approach in this study provides a broadly applicable method to fabricate titanium-based orthopedic implants with enhanced antibacterial properties.







Similar content being viewed by others
References
Cabral H, Kataoka K (2014) Progress of drug-loaded polymeric micelles into clinical studies. J Control Release 190:465–476. https://doi.org/10.1016/j.jconrel.2014.06.042
Chennell P, Feschet-Chassot E, Devers T, Awitor KO, Descamps S, Sautou V (2013) In vitro evaluation of TiO2 nanotubes as cefuroxime carriers on orthopaedic implants for the prevention of periprosthetic joint infections. Int J Pharm 455(1–2):298–305. https://doi.org/10.1016/j.ijpharm.2013.07.014
Dang M, Saunders L, Niu XF, Fan YB, Ma PX (2018) Biomimetic delivery of signals for bone tissue engineering. Bone Res 6:1–12. https://doi.org/10.1038/s41413-018-0025-8
Delmar K, Bianco-Peled H (2016) Composite chitosan hydrogels for extended release of hydrophobic drugs. Carbohydr Polym 136:570–580. https://doi.org/10.1016/j.carbpol.2015.09.072
El Saeed AM, Abd El-Fattah M, Dardir MM (2015) Synthesis and characterization of titanium oxide nanotubes and its performance in epoxy nanocomposite coating. Prog Org Coat 78:83–89. https://doi.org/10.1016/j.porgcoat.2014.09.019
Ercan B, Taylor E, Alpaslan E, Webster TJ (2011) Diameter of titanium nanotubes influences anti-bacterial efficacy. Nanotechnology 22(29). https://doi.org/10.1088/0957-4484/22/29/295102
Feng WC, Geng Z, Li ZY, Cui ZD, Zhu SL, Liang YQ, Liu YD, Wang RF, Yang XJ (2016) Controlled release behaviour and antibacterial effects of antibiotic-loaded titania nanotubes. Mater Sci Eng C 62:105–112. https://doi.org/10.1016/j.msec.2016.01.046
Fux CA, Costerton JW, Stewart PS, Stoodley P (2005) Survival strategies of infectious biofilms. Trends Microbiol 13(1):34–40. https://doi.org/10.1016/j.tim.2004.11.010
Gao A, Hang RQ, Chu PK (2017) Recent advances in anti-infection surfaces fabricated on biomedical implants by plasma-based technology. Surf Coat Technol 312:2–6. https://doi.org/10.1016/j.surfcoat.2016.04.020
Goodman SB, Yao ZY, Keeney M, Yang F (2013) The future of biologic coatings for orthopaedic implants. Biomaterials 34(13):3174–3183. https://doi.org/10.1016/j.biomaterials.2013.01.074
Goodman SB, Gibon E, Pajarinen J, Lin TH, Keeney M, Ren PG, Nich C, Yao Z, Egashira K, Yang F, Konttinen YT (2014) Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J R Soc Interface 11(93):20130962. https://doi.org/10.1098/Rsif.2013.0962
Ivashchenko O, Wozniak A, Coy E, Peplinska B, Gapinski J, Jurga S (2017) Release and cytotoxicity studies of magnetite/Ag/antibiotic nanoparticles: an interdependent relationship. Colloids Surf B Biointerfaces 152:85–94. https://doi.org/10.1016/j.colsurfb.2017.01.009
Jelinska A, Dudzinska I, Zajac M, Oszczpowicz I (2006) The stability of the amorphous form of cefuroxime axetil in solid state. J Pharmaceut Biomed 41(3):1075–1081. https://doi.org/10.1016/j.jpba.2006.02.008
Li HR, Cui Q, Feng B, Wang JX, Lu X, Weng J (2013) Antibacterial activity of TiO2 nanotubes: influence of crystal phase, morphology and Ag deposition. Appl Surf Sci 284:179–183. https://doi.org/10.1016/j.apsusc.2013.07.076
Mandal A, Bisht R, Rupenthal ID, Mitra AK (2017) Polymeric micelles for ocular drug delivery: from structural frameworks to recent preclinical studies. J Control Release 248:96–116. https://doi.org/10.1016/j.jconrel.2017.01.012
Niu XF, Chen P, Jia XL, Wang LZ, Li P, Yang L, Wang YL, Fan YB (2014) Microencapsulation of mechano growth factor E peptide for sustained delivery and bioactivity maintenance. Int J Pharm 469(1):214–221. https://doi.org/10.1016/j.ijpharm.2014.04.054
Niu XF, Liu ZN, Hu J, Rambhia KJ, Fan YB, Ma PX (2016) Microspheres assembled from chitosan-graft-poly(lactic acid) micelle-like core-shell nanospheres for distinctly controlled release of hydrophobic and hydrophilic biomolecules. Macromol Biosci 16(7):1039–1047. https://doi.org/10.1002/mabi.201600020
Peng LL, Mendelsohn AD, LaTempa TJ, Yoriya S, Grimes CA, Desai TA (2009) Long-term small molecule and protein elution from TiO2 nanotubes. Nano Lett 9(5):1932–1936. https://doi.org/10.1021/nl9001052
Peng J, Zhang XM, Li ZY, Liu YD, Yang XJ (2015) Titania nanotube delivery fetal bovine serum for enhancing MC3T3-E1 activity and osteogenic gene expression. Mater Sci Eng C 56:438–443. https://doi.org/10.1016/j.msec.2015.07.005
Popat KC, Eltgroth M, La Tempa TJ, Grimes CA, Desai TA (2007a) Titania nanotubes: a novel platform for drug-eluting coatings for medical implants? Small 3(11):1878–1881. https://doi.org/10.1002/smll.200700412
Popat KC, Eltgroth M, LaTempa TJ, Grimes CA, Desai TA (2007b) Decreased Staphylococcus epidermis adhesion and increased osteoblast functionality on antibiotic-loaded titania nanotubes. Biomaterials 28(32):4880–4888. https://doi.org/10.1016/j.biomaterials.2007.07.037
Popat KC, Leoni L, Grimes CA, Desai TA (2007c) Influence of engineered titania nanotubular surfaces on bone cells. Biomaterials 28(21):3188–3197. https://doi.org/10.1016/j.biomaterials.2007.03.020
Qin H, Cao HL, Zhao YC, Zhu C, Cheng T, Wang QJ, Peng XC, Cheng MQ, Wang JX, Jin GD, Jiang Y, Zhang XL, Liu XY, Chu PK (2014) In vitro and in vivo anti-biofilm effects of silver nanoparticles immobilized on titanium. Biomaterials 35(33):9114–9125. https://doi.org/10.1016/j.biomaterials.2014.07.040
Shard AG, Tomlins PE (2006) Biocompatibility and the efficacy of medical implants. Regen Med 1(6):789–800. https://doi.org/10.2217/17460751.1.6.789
Tsuchiya H, Macak JM, Ghicov A, Taveira L, Schmuki P (2005) Self-organized porous TiO2 and ZrO2 produced by anodization. Corros Sci 47(12):3324–3335. https://doi.org/10.1016/j.corsci.2005.05.041
Yang FH, Niu XF, Gu XN, Xu CP, Wang W, Fan YB (2019) Biodegradable magnesium-incorporated poly(L-lactic acid) microspheres for manipulation of drug release and alleviation of inflammatory response. ACS Appl Mater Interfaces 11(26):23546–23557. https://doi.org/10.1021/acsami.9b03766
Yao C, Webster TJ (2009) Prolonged antibiotic delivery from anodized nanotubular titanium using a co-precipitation drug loading method. J Biomed Mater Res B 91B(2):587–595. https://doi.org/10.1002/jbm.b.31433
Zhang L, Niu XF, Sun L, She ZD, Tan RW, Wang W (2018) Immune response of bovine sourced cross-linked collagen sponge for hemostasis. J Biomater Appl 32(7):920–931. https://doi.org/10.1177/0885328217744080
Zhao LZ, Chu PK, Zhang YM, Wu ZF (2009) Antibacterial coatings on titanium implants. J Biomed Mater Res B 91B(1):470–480. https://doi.org/10.1002/jbm.b.31463
Funding
This study was funded by the National Natural Science Foundation of China (Nos. 11872097, 11421202, and 11827803), Beijing Natural Science Foundation (No. L182017), Shenzhen Science and Technology Project (No. JCYJ20170817140537062), the Fundamental Research Funds for the Central Universities (No. YWF-19-BJ-J-234), the 111 Project (No. B13003), and the International Joint Research Center of Aerospace Biotechnology and Medical Engineering, Ministry of Science and Technology of China.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Niu, X., Sun, L., Zhang, X. et al. Fabrication and antibacterial properties of cefuroxime-loaded TiO2 nanotubes. Appl Microbiol Biotechnol 104, 2947–2955 (2020). https://doi.org/10.1007/s00253-020-10446-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10446-w