Abstract
Purpose
To update the clinical practice guidelines for the use of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the prevention and/or treatment of oral mucositis (OM).
Methods
A systematic review was conducted by the Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO). The body of evidence for each intervention, in each cancer treatment setting, was assigned an evidence level. The findings were added to the database used to develop the 2014 MASCC/ISOO clinical practice guidelines. Based on the evidence level, the following guidelines were determined: Recommendation, Suggestion, and No Guideline Possible.
Results
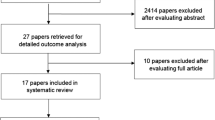
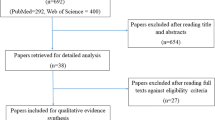
A total of 9 new papers were identified within the scope of this section, adding to the 62 papers reviewed in this section previously. A new Suggestion was made for topical 0.2% morphine for the treatment of OM-associated pain in head and neck (H&N) cancer patients treated with RT-CT (modification of previous guideline). A previous Recommendation against the use of sucralfate-combined systemic and topical formulation in the prevention of OM in solid cancer treatment with CT was changed from Recommendation Against to No Guideline Possible. Suggestion for doxepin and fentanyl for the treatment of mucositis-associated pain in H&N cancer patients was changed to No Guideline Possible.
Conclusions
Of the agents studied for the management of OM in this paper, the evidence supports a Suggestion in favor of topical morphine 0.2% in H&N cancer patients treated with RT-CT for the treatment of OM-associated pain.

Similar content being viewed by others
References
Sonis ST (2004) The pathobiology of mucositis. Nat Rev Cancer 4(4):277–284. https://doi.org/10.1038/nrc1318
Lalla RV, Sonis ST, Peterson DE (2008) Management of oral mucositis in patients who have cancer. Dent Clin N Am 52(1):61–77, viii. https://doi.org/10.1016/j.cden.2007.10.002
Lalla RV, Bowen J, Barasch A, Elting L, Epstein J, Keefe DM, McGuire D, Migliorati C, Nicolatou-Galitis O, Peterson DE, Raber-Durlacher JE, Sonis ST, Elad S, Mucositis Guidelines Leadership Group of the Multinational Association of Supportive Care in Cancer and International Society of Oral Oncology (MASCC/ISOO) (2014) MASCC/ISOO clinical practice guidelines for the management of mucositis secondary to cancer therapy. Cancer 120(10):1453–1461. https://doi.org/10.1002/cncr.28592
Rubenstein EB, Peterson DE, Schubert M, Keefe D, McGuire D, Epstein J, Elting LS, Fox PC, Cooksley C, Sonis ST, Mucositis Study Section of the Multinational Association for Supportive Care in Cancer, International Society for Oral Oncology (2004) Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer 100(9 Suppl):2026–2046. https://doi.org/10.1002/cncr.20163
Saunders DP, Epstein JB, Elad S, Allemano J, Bossi P, van de Wetering M, Rao NG, Potting C, Cheng KK, Freidank A, Brennan MT, Bowen J, Dennis K, Lalla RV, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) (2013) Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients. Support Care Cancer 21(11):3191–3207. https://doi.org/10.1007/s00520-013-1871-y
Ranna V, Cheng KKF, Castillo DA, Porcello L, Vaddi A, Lalla RV, Bossi P, Elad S, On behalf of the Mucositis Study group of the Multinational Association of Supportive Care in Cancer/International Society for Oral Oncology (MASCC/ISOO) (2019) Development of the MASCC/ISOO clinical practice guidelines for mucositis: an overview of the methods. Support Care Cancer 27:1–16. https://doi.org/10.1007/s00520-019-04891-1
Somerfield MR, Padberg J, Pfister D et al (2000) ASCO clinical practice guidelines: process, progress, pitfalls, and prospects. Class Pap Curr Comments 4(4):881–886
Hadorn DC, Baker D, Hodges JS, Hicks N (1996) Rating the quality of evidence for clinical practice guidelines. J Clin Epidemiol 49(7):749–754. https://doi.org/10.1016/0895-4356(96)00019-4
Corvo R, Amichetti M, Ascarelli A et al (2008) Effects of fluconazole in the prophylaxis of oropharyngeal candidiasis in patients undergoing radiotherapy for head and neck tumour: results from a double-blind placebo-controlled trial. Eur J Cancer Care (Engl) 17(3):270–277. https://doi.org/10.1111/j.1365-2354.2007.00855.x
Nicolatou-Galitis O, Velegraki A, Sotiropoulou-Lontou A, Dardoufas K, Kouloulias V, Kyprianou K, Kolitsi G, Skarleas C, Pissakas G, Papanicolaou VS, Kouvaris J (2006) Effect of fluconazole antifungal prophylaxis on oral mucositis in head and neck cancer patients receiving radiotherapy. Support Care Cancer 14(1):44–51. https://doi.org/10.1007/s00520-005-0835-2
Rao NG, Han G, Greene JN, Tanvetyanon T, Kish JA, de Conti RC, Chuong MD, Shridhar R, Biagioli MC, Caudell JJ, Trotti AM 3rd (2013) Effect of prophylactic fluconazole on oral mucositis and candidiasis during radiation therapy for head-and-neck cancer. Pract Radiat Oncol 3(3):229–233. https://doi.org/10.1016/j.prro.2012.05.008
Orvain C, Moles-Moreau MP, Francois S et al (2015) Miconazole mucoadhesive buccal tablet in high-dose therapy with autologous stem cell transplantation (HDT/ASCT)-induced mucositis. Support Care Cancer 23(2):359–364. https://doi.org/10.1007/s00520-014-2365-2
Allison RR, Ambrad AA, Arshoun Y, Carmel RJ, Ciuba DF, Feldman E, Finkelstein SE, Gandhavadi R, Heron DE, Lane SC, Longo JM, Meakin C, Papadopoulos D, Pruitt DE, Steinbrenner LM, Taylor MA, Wisbeck WM, Yuh GE, Nowotnik DP, Sonis ST (2014) Multi-institutional, randomized, double-blind, placebo-controlled trial to assess the efficacy of a mucoadhesive hydrogel (MuGard) in mitigating oral mucositis symptoms in patients being treated with chemoradiation therapy for cancers of the head and neck. Cancer 120(9):1433–1440. https://doi.org/10.1002/cncr.28553
Barber C, Powell R, Ellis A, Hewett J (2007) Comparing pain control and ability to eat and drink with standard therapy vs Gelclair: a preliminary, double centre, randomised controlled trial on patients with radiotherapy-induced oral mucositis. Support Care Cancer 15(4):427–440. https://doi.org/10.1007/s00520-006-0171-1
Lindsay G, Rushton R, Harris T (2009) The clinical effectiveness of Gelclair in the management of oral mucositis. Austal Nurs J 16:30–33
Vokurka S, Skardova J, Hruskova R et al (2011) The effect of polyvinylpyrrolidone-sodium hyaluronate gel (Gelclair) on oral microbial colonization and pain control compared with other rinsing solutions in patients with oral mucositis after allogeneic stem cells transplantation. Med Sci Monit 17(10):CR572-6. https://doi.org/10.12659/msm.881983
Szabo S, Hollander D (1989) Pathways of gastrointestinal protection and repair: mechanisms of action of sucralfate. Am J Med 86(6A):23–31. https://doi.org/10.1016/0002-9343(89)90153-8
McCarthy DM (1991) Sucralfate. N Engl J Med 325(14):1017–1025. https://doi.org/10.1056/NEJM199110033251407
Szabo S, Vattay P, Scarbrough E, Folkman J (1991) Role of vascular factors, including angiogenesis, in the mechanisms of action of sucralfate. Am J Med 91(2A):158S–160S. https://doi.org/10.1016/0002-9343(91)90469-e
Ala S, Saeedi M, Janbabai G, Ganji R, Azhdari E, Shiva A (2016) Efficacy of sucralfate mouth wash in prevention of 5-fluorouracil induced oral mucositis: a prospective, randomized, double-blind, controlled trial. Nutr Cancer 68(3):456–463. https://doi.org/10.1080/01635581.2016.1153666
Nottage M, McLachlan SA, Brittain MA, Oza A, Hedley D, Feld R, Siu LL, Pond G, Moore MJ (2003) Sucralfate mouthwash for prevention and treatment of 5-fluorouracil-induced mucositis: a randomized, placebo-controlled trial. Support Care Cancer 11(1):41–47. https://doi.org/10.1007/s00520-002-0378-8
Shenep JL, Kalwinsky DK, Hutson PR et al (1988) Efficacy of oral sucralfate suspension in prevention and treatment of chemotherapy-induced mucositis. J Pediatr 113(4):758–763. https://doi.org/10.1016/s0022-3476(88)80397-4
Etiz D, Erkal HS, Serin M, Küçük B, Hepari A, Elhan AH, Tulunay O, Cakmak A (2000) Clinical and histopathological evaluation of sucralfate in prevention of oral mucositis induced by radiation therapy in patients with head and neck malignancies. Oral Oncol 36(1):116–120. https://doi.org/10.1016/S1368-8375(99)00075-5
Cengiz M, Ozyar E, Ozturk D et al (1999) Sucralfate in the prevention of radiation-induced oral mucositis. J Clin Gastroenterol 28(1):40–43. https://doi.org/10.1097/00004836-199901000-00009
Matthews RH, Ercal N (1996) Prevention of mucositis in irradiated head and neck cancer patients. J Exp Ther Oncol 1(2):135–138
Epstein JB, Wong FL (1994) The efficacy of sucralfate suspension in the prevention of oral mucositis due to radiation therapy. Int J Radiat Oncol Biol Phys 28(3):693–698. https://doi.org/10.1016/0360-3016(94)90195-3
Makkonen TA, Bostrom P, Vilja P et al (1994) Sucralfate mouth washing in the prevention of radiation-induced mucositis: a placebo-controlled double-blind randomized study. Int J Radiat Oncol Biol Phys 30(1):177–182. https://doi.org/10.1016/0360-3016(94)90533-9
Evensen JF, Bjordal K, Jacobsen AB, Løkkevik E, Tausjø JE (2001) Effects of Na-sucrose octasulfate on skin and mucosa reactions during radiotherapy of head and neck cancers--a randomized prospective study. Acta Oncol 40(6):751–755. https://doi.org/10.1080/02841860152619188
Carter DL, Hebert ME, Smink K, Leopold KA, Clough RL, Brizel DM (1999) Double blind randomized trial of sucralfate vs placebo during radical radiotherapy for head and neck cancers. Head Neck 21(8):760–766. https://doi.org/10.1002/(SICI)1097-0347(199912)21:8<760::AID-HED12>3.0.CO;2-%23
Pfeiffer P, Hansen O, Madsen EL, May O (1990) A prospective pilot study on the effect of sucralfate mouth-swishing in reducing stomatitis during radiotherapy of the oral cavity. Acta Oncol 29(4):471–473. https://doi.org/10.3109/02841869009090032
Giorgi F, Bascioni R, De Signoribus G et al (1996) Sucralfate prophylaxis of fluorouracil-induced stomatitis. Tumori 82(6):585–587. https://doi.org/10.1177/030089169608200614
Castagna L, Benhamou E, Pedraza E, Luboinski M, Forni M, Brandes I, Pico JL, Dietrich PY (2001) Prevention of mucositis in bone marrow transplantation: a double blind randomised controlled trial of sucralfate. Ann Oncol 12(7):953–955. https://doi.org/10.1023/a:1011119721267
Lievens Y, Haustermans K, Van den Weyngaert D et al (1998) Does sucralfate reduce the acute side-effects in head and neck cancer treated with radiotherapy? A double-blind randomized trial. Radiother Oncol 47(2):149–153. https://doi.org/10.1016/S0167-8140(97)00231-4
Barker G, Loftus L, Cuddy P et al (1991) The effects of sucralfate suspension and diphenhydramine syrup plus kaolin-pectin on radiotherapy-induced mucositis. Oral Surg Oral Med Oral Pathol 71(3):288–293. https://doi.org/10.1016/0030-4220(91)90301-r
Meredith R, Salter M, Kim R et al (1997) Sucralfate for radiation mucositis: results of a double-blind randomized trial. Int J Radiat Oncol Biol Phys 37(2):275–279. https://doi.org/10.1016/s0360-3016(96)00531-7
Dodd MJ, Miaskowski C, Greenspan D, MacPhail L, Shih AS, Shiba G, Facione N, Paul SM (2003) Radiation-induced mucositis: a randomized clinical trial of micronized sucralfate versus salt & soda mouthwashes. Cancer Investig 21(1):21–33. https://doi.org/10.1081/CNV-120016400
Franzén L, Henriksson R, Littbrand BO et al (1995) Effects of sucralfate on mucositis during and following radiotherapy of malignancies in the head and neck region: a double-blind placebo-controlled study. Acta Oncol 34(2):219–223. https://doi.org/10.3109/02841869509093959
Chiara S, Nobile MT, Vincenti M, Gozza A, Pastrone I, Rosso M, Rosso R (2001) Sucralfate in the treatment of chemotherapy-induced stomatitis: a double-blind, placebo-controlled pilot study. Anticancer Res 21(5):3707–3710
Loprinzi CL, Ghosh C, Camoriano J, Sloan J, Steen PD, Michalak JC, Schaefer PL, Novotny PJ, Gerstner JB, White DF, Hatfield AK, Quella SK (1997) Phase III controlled evaluation of sucralfate to alleviate stomatitis in patients receiving fluorouracil-based chemotherapy. J Clin Oncol 15(3):1235–1238. https://doi.org/10.1200/JCO.1997.15.3.1235
Saarilahti K, Kajanti M, Joensuu T et al (2002) Comparison of granulocyte-macrophage colony-stimulating factor and sucralfate mouthwashes in the prevention of radiation-induced mucositis: a double-blind prospective randomized phase III study. Int J Radiat Oncol Biol Phys 54(2):479–485. https://doi.org/10.1016/s0360-3016(02)02935-8
Stein C (1995) The control of pain in peripheral tissue by opioids. N Engl J Med 332(25):1685–1690. https://doi.org/10.1056/NEJM199506223322506
Cerchietti LC, Navigante AH, Bonomi MR, Zaderajko MA, Menéndez PR, Pogany CE, Roth BM (2002) Effect of topical morphine for mucositis-associated pain following concomitant chemoradiotherapy for head and neck carcinoma. Cancer 95(10):2230–2236. https://doi.org/10.1002/cncr.10938
Cerchietti LC, Navigante AH, Korte MW et al (2003) Potential utility of the peripheral analgesic properties of morphine in stomatitis-related pain: a pilot study. Pain 105(1-2):265–273. https://doi.org/10.1016/S0304-3959(03)00227-6
Vayne-Bossert P, Escher M, de Vautibault CG, Dulguerov P, Allal A, Desmeules J, Herrmann FR, Pautex S (2010) Effect of topical morphine (mouthwash) on oral pain due to chemotherapy- and/or radiotherapy-induced mucositis: a randomized double-blinded study. J Palliat Med 13(2):125–128. https://doi.org/10.1089/jpm.2009.0195
Sarvizadeh M, Hemati S, Meidani M, Ashouri M, Roayaei M, Shahsanai A (2015) Morphine mouthwash for the management of oral mucositis in patients with head and neck cancer. Adv Biomed Res 4:44. https://doi.org/10.4103/2277-9175.151254
Krajnik M, Zylicz Z, Finlay I, Luczak J, van Sorge A (1999) Potential uses of topical opioids in palliative care--report of 6 cases. Pain 80(1-2):121–125. https://doi.org/10.1016/S0304-3959(98)00211-5
Shaiova L, Mori M, Anderson K, Loewen G, Ghalie R, Homel P, Portenoy R (2007) Administration of morphine sulfate extended-release capsules via gastrostomy: dissolution study and case reports. J Palliat Med 10(5):1063–1067. https://doi.org/10.1089/jpm.2006.0262
Atkin T, Comai S, Gobbi G (2018) Drugs for insomnia beyond benzodiazepines: pharmacology, clinical applications, and discovery. Pharmacol Rev 70(2):197–245. https://doi.org/10.1124/pr.117.014381
Mika J, Zychowska M, Popiolek-Barczyk K, Rojewska E, Przewlocka B (2013) Importance of glial activation in neuropathic pain. Eur J Pharmacol 716(1-3):106–119. https://doi.org/10.1016/j.ejphar.2013.01.072
Leenstra JL, Miller RC, Qin R, Martenson JA, Dornfeld KJ, Bearden JD, Puri DR, Stella PJ, Mazurczak MA, Klish MD, Novotny PJ, Foote RL, Loprinzi CL (2014) Doxepin rinse versus placebo in the treatment of acute oral mucositis pain in patients receiving head and neck radiotherapy with or without chemotherapy: a phase III, randomized, double-blind trial (NCCTG-N09C6 [Alliance]). J Clin Oncol 32(15):1571–1577. https://doi.org/10.1200/JCO.2013.53.2630
Epstein JB, Silverman S Jr, Paggiarino DA et al (2001) Benzydamine HCl for prophylaxis of radiation-induced oral mucositis: results from a multicenter, randomized, double-blind, placebo-controlled clinical trial. Cancer 92(4):875–885. https://doi.org/10.1002/1097-0142(20010815)92:4<875::aid-cncr1396>3.0.co;2-1
Epstein JB, Epstein JD, Epstein MS, Oien H, Truelove EL (2008) Doxepin rinse for management of mucositis pain in patients with cancer: one week follow-up of topical therapy. Spec Care Dentist 28(2):73–77. https://doi.org/10.1111/j.1754-4505.2008.00015.x
Xing SZ, Zhang Y (2015) Efficacy and safety of transdermal fentanyl for the treatment of oral mucositis pain caused by chemoradiotherapy in patients with esophageal squamous cell carcinoma. Support Care Cancer 23(3):753–759. https://doi.org/10.1007/s00520-014-2419-5
Guo SP, Wu SG, Zhou J, Feng HX, Li FY, Wu YJ, Sun JY, He ZY (2014) Transdermal fentanyl for pain due to chemoradiotherapy-induced oral mucositis in nasopharyngeal cancer patients: evaluating efficacy, safety, and improvement in quality of life. Drug Des Devel Ther 8:497–503. https://doi.org/10.2147/DDDT.S60187
Cai Q, Huang H, Sun X, Xia Z, Li Y, Lin X, Guo Y (2008) Efficacy and safety of transdermal fentanyl for treatment of oral mucositis pain caused by chemotherapy. Expert Opin Pharmacother 9(18):3137–3144. https://doi.org/10.1517/14656560802504508
Demarosi F, Lodi G, Soligo D, Sardella A, Volpe AD, Carrassi A, Deliliers GL (2004) Transdermal fentanyl in HSCT patients: an open trial using transdermal fentanyl for the treatment of oral mucositis pain. Bone Marrow Transplant 33(12):1247–1251. https://doi.org/10.1038/sj.bmt.1704515
Kim JG, Sohn SK, Kim DH, Baek JH, Chae YS, Bae NY, Kim SY, Lee KB (2005) Effectiveness of transdermal fentanyl patch for treatment of acute pain due to oral mucositis in patients receiving stem cell transplantation. Transplant Proc 37(10):4488–4491. https://doi.org/10.1016/j.transproceed.2005.11.038
Strupp C, Sudhoff T, Germing U, Hunerliturkoglu A, Schneider P, Niederste-Hollenberg A, Heyll A, Aul C (2000) Transdermal fentanyl during high-dose chemotherapy and autologous stem cell support. Oncol Rep 7(3):659–661. https://doi.org/10.3892/or.7.3.659
Bossi P, Locati L, Bergamini C, Mirabile A, Granata R, Imbimbo M, Resteghini C, Licitra L (2014) Fentanyl pectin nasal spray as treatment for incident predictable breakthrough pain (BTP) in oral mucositis induced by chemoradiotherapy in head and neck cancer. Oral Oncol 50(9):884–887. https://doi.org/10.1016/j.oraloncology.2014.06.013
McCullough RW (2019) Practice insights on patient care-management overview for chemoradiation toxic mucositis-guidelines, guideline-supported therapies and high potency polymerized cross-linked sucralfate (ProThelial). J Oncol Pharm Pract 25(2):409–422. https://doi.org/10.1177/1078155218758864
Mogensen S, Treldal C, Kristensen CA, Bentzen J, Lawson-Smith L, Petersen J, Andersen O (2017) Effect of bupivacaine lozenges on oral mucositis pain: a randomized controlled multicenter phase II study. Pain Rep 2(5):e619. https://doi.org/10.1097/PR9.0000000000000619
Cheng Y, Qin SK, Chen YP, Dong LH, Sun XD, Yu SY, Wu SK (2018) Local analgesic effect of a bioadhesive barrier-forming oral liquid in cancer patients with oral mucositis caused by chemotherapy and/or radiotherapy: a randomized multicenter, single-use, positive-controlled, open-label study. Onco Targets Ther 11:8555–8564. https://doi.org/10.2147/OTT.S185915
Acknowledgments
The authors are thankful for the medical librarians for their valuable contribution to this project: Lorraine Porcello, MSLIS, MSIM—Bibby Dental Library, Eastman Institute for Oral Health, University of Rochester Medical Center, Rochester, NY, USA; Daniel A. Castillo, MLIS—Edward G. Miner Library, University of Rochester Medical Center, Rochester, NY, USA.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
According to the MASCC Guideline Policy, employees of commercial entities were not eligible to serve on this MASCC Guideline Panel. All authors completed a Conflict of Interest disclosure form and conflicts are disclosed in the guideline’s publications. The authors disclose no conflict of interest (TR, AK, KKFC, NY, JJ, KB, MW, NBD, TK, KC, VR, AV, and SE). DPS has received research support to her institution from Galera Therapeutics. PB has served an advisory role for AstraZeneca, Helsinn, and Kyowa Kirin and received grants from Merck, Kyowa Kirin, and Roche. RVL has served as a consultant for Colgate Oral Pharmaceuticals, Galera Therapeutics, Ingalfarma SpA, Monopar Therapeutics, Mundipharma, and Sucampo Pharma; has received research support to his institution from Galera Therapeutics, Novartis, Oragenics, and Sucampo Pharma; and has received stock in Logic Biosciences. JE has received grants from Prysma Inc. for a funded trial.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saunders, D.P., Rouleau, T., Cheng, K. et al. Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer 28, 2473–2484 (2020). https://doi.org/10.1007/s00520-019-05181-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-05181-6