Abstract
Mitochondrial fission factor (Mff) has been demonstrated to play a role in the activation of mitochondrial cleavage and mitochondrial death, denoting its role in the regulation of mitochondrial quality control. Recent evidence suggested that the mRNA translation of Mff is under the negative regulation by the RNA-binding protein Pumilio2 (Pum2). This study was designed to examine the role of Pum2 and Mff in the governance of mitochondrial quality control in a murine model of acute ischemic kidney injury. Our results indicated that genetic deletion of Mff overtly attenuated ischemic acute kidney injury (AKI)–induced renal failure through inhibition of pro-inflammatory response, tubular oxidative stress, and ultimately cell death in the kidney. Furthermore, Mff inhibition effectively preserved mitochondrial homeostasis through amelioration of mitochondrial mitosis, restoration of Sirt1/3 expression, and boost of mitochondrial respiration. Western blot analysis revealed that levels of Pum2 were significantly downregulated by ischemic AKI, inversely coinciding with levels of Mff. Overexpression of Pum2 reduced ischemic AKI-mediated Mff upregulation and offered protection on renal tubules through modulation of mitochondrial quality control. Taken together, our data have unveiled the molecular mechanism of the Pum2-Mff axis in mitochondrial quality control in a mouse model of ischemic AKI. These data indicated the therapeutic potential of Pum2 activation and Mff inhibition in the management of ischemic AKI.
Similar content being viewed by others
Introduction
The onset of acute kidney injury (AKI) is mainly triggered by pathological insults including renal ischemia-reperfusion hit, cardiogenic shock, nephrotoxicity, and inflammation (Zhu et al. 2019). In general, ischemic AKI is characterized by critical damage to renal tubule, resulting in dynamically reversible oliguria and abrupt loss of kidney function (Biernacki et al. 2018; Qian et al. 2019). Ample epidemiological evidence has described a tight association between ischemic AKI and in-hospital morbidity/mortality (Soranno et al. 2019), although underlying molecular mechanisms behind both pathogenesis and regulatory components remain at large (Kong et al. 2019).
Mitochondrion is the cornerstone of cellular energy metabolism. Not surprisingly, mitochondrial quality control is cardinal to safeguard inheritance, dynamics, and bioenergetics in mitochondria (Sedlackova and Korolchuk 2019). In particular, mitochondrial dysfunction plays an essential role in the development of ischemic AKI, a process where apoptosis, redox imbalance, and metabolic impairment are sequentially turned on and function as key messengers to provoke tubular death and subsequently renal failure (Bhargava and Schnellmann 2017; Lin et al. 2019b; Sun et al. 2019). In an effort to maintain mitochondrial quality control, mitochondrial fission is perhaps the very initial step to induce mitochondrial structural deformation prior to mitochondrial functional repression (Tahrir et al. 2019). Fragmented mitochondria, triggered by dynamic-related protein 1 (Drp1)–mediated mitochondrial fission, have been documented in reperfused renal tubules, while dysregulated mitochondrial division is connected to prominent tubular death through either endogenous or exogenous mechanisms, revealing a rather critical role for mitochondrial mitosis in the regulation of renal tubular fate (Tanaka and Nangaku 2019). Findings from our previous studies have noted the involvement of the mitochondrial fission factor (Mff)–mediated mitochondrial mitosis in the development of myocardial ischemia-reperfusion injury both in vivo and in vitro (Zhou et al. 2017; Zhou et al. 2018a). Both genetic ablation and pharmacologic inhibition of Mff were demonstrated to drastically improve myocardial performance (Zhou et al. 2017). Mechanistically, mitochondrial fission is initiated by Drp1 shuttling from cytoplasm onto the outer membrane of mitochondria where Drp1 forms a potential contractive ring around mitochondria, leading to cleavage of one mitochondria into small daughter mitochondria (Osellame et al. 2016; Otera et al. 2010; Zhang et al. 2016). During this process, Drp1 localization onto mitochondria requires its receptors residing on mitochondria. Mff, one of the receptors of Drp1, is considered as an essential hub that tightly recruits Drp1 localization onto mitochondria (Ji et al. 2015; Liu and Chan 2015). The coordination between Mff and Drp1 seems to be the prerequisite for mitochondrial fission. Nonetheless, the role of Mff in ischemic AKI remains largely unknown.
In-depth attempt has been engaged towards the understanding of the upstream regulator for Mff. In our earlier studies, two mechanisms were identified in the regulation of Mff: one is handled through DUSP1-mediated Mff protein upregulation (Jin et al. 2018), and the other involves NR4A1-induced Mff phosphorylation (Zhou et al. 2018a). Nonetheless, both regulatory mechanisms function at the post-transcriptional levels, and little information is available for transcriptional regulation of Mff in pathological settings such as ischemia-reperfusion injury. A recent study from aged mice noted that the stability of Mff mRNA seems to be affected by the RNA-binding protein Pumilio2 (Pum2), which inhibits translation of mRNA encoding for Mff. Interestingly, mitophagy and mitochondrial performance are also affected by Pum2 (D'Amico et al. 2019). These findings are also in concordance with the notion that Pum2 downregulation is instrumental in the pathogenesis of brain injury (Kanemaru et al. 2013). To this end, our present study was designed to understand the role of the Pum2-Mff axis in the orchestration of mitochondrial quality control in ischemic AKI with a particular focus on mitochondrial mitosis.
Methods
Animal model
Adult male C57BL/6 wild-type (WT) and Mff-knockout (Mff-KO) mice were employed to induce the ischemic AKI mode. Generation of Mff-KO mice was described in detail in our previous report (Zhou et al. 2017). All animal procedures were approved by the animal care and use committee from the PLA General Hospital and were in compliance with the NIH guideline. In brief, a renal ischemia-reperfusion injury (IRI) model was achieved through bilateral renal pedicle clamping for 30-min ischemia followed by 24-h reperfusion (Perry et al. 2018). Levels of blood urea nitrogen (BUN) and serum creatinine (Scr) were measured using a metabolic assay kit (Sigma, St. Louis, MO) per manufacturer’s instruction.
Histology
Kidney samples were fixed in a 4% phosphate-buffered formaldehyde solution (Klinipath, Olen, Belgium). Samples were then dehydrated, embedded in paraffin, and stained with hematoxylin and eosin (H&E) (Vial et al. 2019).
ELISA for intracellular glutathione and glutathione peroxidase
Cells were rinsed three times using ice-cold PBS (pH 7.4). Then, cells were homogenized in 200 μL ice-cold lysis buffer (PBS containing 0.5% NP-40) for 30 min. Subsequently, samples were centrifuged for 10 min at 1000–3000 rpm and supernatant was immediately examined to detect levels of intracellular glutathione (GSH) and glutathione peroxidase (GPx) per the manufacturer’s guide (Curley et al. 2018) (Mouse GSH ELISA Kit, Catalog: MBS267424, MyBioSource; Mouse GPX ELISA Kit, Catalog: MBS776262, MyBioSource).
Cell culture and treatment
Primary proximal tubule cells were prepared as described previously (Xie et al. 2018b). Isolated proximal tubular cells were cultured in the DMEM medium containing 10% fetal bovine serum (FBS). For in vitro study, an IRI model mimicking ischemic AKI was established using ATP depletion-induced metabolic stress by adding 10 mM rotenone (a complex I inhibitor) in a glucose-free DMEM for approximately 30 min(Biernacki et al. 2018). Primary proximal tubular cells were cultured under normal condition for 24 h.
CCK8 analysis
Cell viability was analyzed using a Cell Counting Kit-8 (CCK-8, Dojindo Laboratories, Kumamoto, Japan) assay. Cells were seeded onto 96-well plates at a density of 1500 cells per well. Cultures were stained using CCK-8 at the indicated time points. In brief, 10 μL of CCK-8 solution was added to each well and incubated for 2 h at 37 °C (Huang et al. 2019). The absorption was measured in a microplate reader using a spectrophotometer at the wavelength of 450 nm (Bio-Rad Laboratories Inc., Tokyo, Japan) (Jung et al. 2018).
Immunofluorescent staining
Cells (1 × 105/well) were seeded on a slide. Cells under different conditions were fixed in 4% formaldehyde for 20 min. Slides were incubated for 30 min in 0.1% Triton X-100 in PBS. Primary antibodies (Tim23, 1:1000, Santa Cruz Biotechnology, #sc-13,298) were added on the slide and were kept overnight at 4 °C. The primary antibody was labeled using the specific secondary antibody with Alexa Fluor 405, 488, or 568 (Invitrogen; A31553, A11008, or A11004, respectively) (Han et al. 2019). Fluorescent change of cells was measured using confocal microscopy (Olympus, FV-1000, Tokyo, Japan)(Kim et al. 2019).
Detection of inflammation factors expression by qPCR
Total RNA was extracted from tubular cells using a single-step method with the TRIzol reagent (Invitrogen). For reverse transcription-polymerase chain reaction (RT-PCR), the cDNA template was synthesized using 1 μg of total RNA with an oligo-dT primer and the Moloney murine leukemia virus (MMLV) reverse transcriptase (Promega, Madison, USA)(DeLeon-Pennell et al. 2018). The sequences of PCR primers were as follows: TNFα (Forward, 5′-AGATGGAGCAACCTAAGGTC-3′; Reverse, 5′GCAGACCTCGCTGTTCTAGC-3′), IL6 (Forward, 5′-CAGACTCGCGCCTCTAAGGAGT-3′; Reverse, 5′-GATAGCCGATCCGTCGAA-3′), MCP1 (Forward, 5′-GGATGGATTGCACAGCCATT-3′; Reverse, 5′-GCGCCGACTCAGAGGTGT-3′), KIM1 (Forward, 5′-ACATATCGTGGAATCACAACGAC-3′; Reverse, 5′-ACTGCTCTTCTGATAGGTGACA-3′), NGAL (Forward, 5′-CAAGCAATACTTCAAAATTACCCTGTA-3′; Reverse, 5′-GCAAAGCGGGTGAAACGTT-3′).
Adenovirus-mediated Pum2 overexpression
Pum2 adenovirus was constructed by GenePharma Co., Ltd. (Shanghai, China). Primary proximal tubule cells were infected with Pum2 adenovirus for 24 h at 37 °C under 5% CO2. Proteins were isolated from cells and Western blotting was used to verify the adenovirus-mediated Pum2 overexpression efficiency (Zhu et al. 2018b).
Western blot analysis
Total protein extract was obtained by homogenizing tissue in a RIPA buffer supplemented with 5.5% β-glycerophosphate, 1M dithiothreitol, and complete protease and phosphatase inhibitors (Roche Diagnostics, Vilvoorde, Belgium), and then total protein concentrations were determined (Bio-Rad) (Li et al. 2018a; Moore et al. 2018). Thirty micrograms of protein (one mouse or a mixture of two mice per group) was separated on a 4–20% Criterion Stain-Free gradient gel (Chemidoc MP Imaging system, BioRad), and the proteins were transferred to a nitrocellulose membrane (Bio-Rad). The membranes were blocked with 5% skim milk in Tris buffered saline with 0.1% Tween-20 (TBST) (Sigma-Aldrich) for 1 h and incubated overnight at 4 °C with Pum2 (1:1000, Abcam, #ab92390), HtrA2/Omi (1:1000, Abcam, #ab75982), PCNA (1:1000, Abcam, #ab29), Drp1 (1:1000, Abcam, #ab56788), Bax (1:1000, Cell Signaling Technology, #2772), and Mff (1:1000; Abcam; #ab129075) in blocking buffer. Next, blots were incubated for 1 h at room temperature with a horseradish peroxidase–conjugated secondary antibody (1/2000 dilution, Cell Signaling Technology, Leiden, The Netherlands). Bands were visualized using chemiluminescence and imaged on a Chemidoc MP Imager (Bio-Rad). Band intensity and total protein quantification and normalization were performed using ImageLab 5.2 software (Bio-Rad) (Na et al. 2019). All uncropped original gels of western blots have been provided in Supplemental Figures.
ROS evaluation
Cytoplasmic ROS and mitochondrial ROS were stained using DCF-DA (10 μM) and MitoSOX Red probe, respectively (Zhu et al. 2018a). Fluorescence intensity of ROS was converted into a grayscale picture to quantify ROS levels. To observe levels of mito-ROS, flow cytometry was employed. In brief, cells were firstly treated with mIRI and were isolated in an ice-cold PBS. Then, MitoSOX Red probe was incubated with cells for 30 min in the dark. Then, samples were immediately analyzed using the flow cytometry (BD FACSCalibur cytometry).
Mitochondrial fission staining
Mitochondrial morphology was visualized using MitoTracker red staining according to the manufacturer’s instruction (Zhou et al. 2018b). The nuclei of cells were stained using 4′, 6-diamidino-2-phenylindole (DAPI). Fluorescent images were visualized and captured through × 100 oil immersion using a Zeiss fluorescence microscope. Mitochondrial fission was quantified based on our previous study (Jin et al. 2018). In brief, mitochondrial length was evaluated in at least 100 cells. Meanwhile, fragmented mitochondria were also recorded in at least 200 cells, and then the percentage of cells with fragmented mitochondria was calculated.
Mitochondrial potential and respiration detection
Mitochondrial potential was visualized using a JC-1 probe based on our earlier reports (Li et al. 2018b). In brief, primary proximal tubule cells were stained with JC-1 and then the fluorescent images were captured through a Zeiss fluorescence microscope. Mitochondrial respiration was analyzed as previously described using a Biological Oxygen Monitor System (Hansatech Instruments, King’s Lynn, UK). Then mitochondrial state 3/4 respiration rate and ADP phosphorylation were calculated (Sajib et al. 2018).
Statistical analysis
Data were shown as mean ± SEM. Differences between groups were analyzed by ANOVA analysis using the GraphPad Prism software. A p value less than 0.05 was considered statistically significant.
Results
Genetic ablation of Mff attenuated ischemic AKI
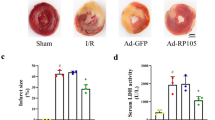
AKI was established following 30-min ischemia and 24-h reperfusion. Renal failure was featured by overtly elevated serum creatinine (Scr) and BUN levels (Fig. 1a, b). In addition, other kidney damage biomarkers such as KIM-1 and NGAL (Fig. 1c, d), as assessed using qPCR, were also upregulated after ischemic AKI. Interestingly, Mff ablation significantly decreased the levels of Scr and BUN in ischemic AKI. Consistently, Mff ablation negated ischemic AKI-induced elevation in transcriptional levels of KIM-1 and NGAL. Histologic observation also noted prominent loss of tubule-cell viability as an abrupt ischemic AKI, with a lesser tubular damage in Mff-KO mice (Fig. 1e, f). In vivo, a protein-activity assay for caspase-3 indicated that ischemic AKI abruptly triggered apoptosis initiation as shown by increased caspase-3 activity, the effect of which was drastically attenuated by Mff knockout (Fig. 1g). In vitro study further examined isolated primary renal tubule cells to mimic ischemia-reperfusion injury (mIRI). mIRI repressed tubule-cell viability, as determined by CCK8 assay (Fig. 1h). However, tubular cells from Mff knockout mice exhibited normal cell viability. Altogether, levels of Mff were elevated in a murine kidney ischemia-reperfusion injury model and were correlated with renal damage, confirming its pathological role in ischemic AKI.
Mff deletion attenuates ischemic AKI. a, b AKI was established through 30 min of ischemia and 24 h of reperfusion. Blood was collected and levels of serum creatinine and BUN were determined using ELISA. c, d Reperfused kidney tissues were isolated and qPCR was used to evaluate transcription of KIM-1 and NGAL. e, f HE staining for acute tubular injury. g Proteins were isolated after ischemic AKI and then activity of caspase-3 was determined using ELISA. h CCK8 assay was used to assess cell viability of primary proximal tubule cells. mIRI model was established using ATP deletion–mediated metabolic stress through addition of rotenone (10 nM). Mean ± SEM, n = 6/group, #p < 0.05 vs. sham/control group; *p < 0.05 vs. IRI/mIRI group
Mff deficiency interrupted the inflammation reaction and oxidative stress
In addition to the appearance of renal failure, our data further showed overt inflammation reaction provoked by ischemic AKI, an effect that was mitigated by Mff knockout (Fig. 2a–c). In addition, the renal proliferative index, as assessed using PCNA expression (Fig. 2d, e), was inhibited in response to ischemic AKI. However, Mff deletion effectively restored the proliferative capacity in renal tissues.
Mff deletion reduces inflammation damage and oxidative stress. a–c Reperfused kidney tissues were isolated and qPCR was employed to assess transcription of TGFα, IL-6, and MCP1. d, e Proteins were isolated after ischemic AKI and then the expression of PCNA was determined using western blotting. e, f In vitro, intracellular GSH and GPx were measured through ELISA kit. h, i Tubular cells were treated with mIRI and then the production of mito-ROS was measured via flow cytometry. j–l ROS probe was used to observe levels of mitochondrial ROS (mito-ROS) and cytoplasmic ROS (cyto-ROS) using MitoSOX Red and DCF-DA, respectively. Mean ± SEM, n = 6/group, #p < 0.05 vs. sham/control group; *p < 0.05 vs. IRI/mIRI group
The pro-inflammatory response was typically followed by the uncontrolled oxidative stress due to neutrophile-mediated ROS eruption. In vitro findings exhibited decreased levels of GSH and GPx after mIRI. Interestingly, Mff knockout restored the content of GSH and GPx in mIRI-treated tubular cells (Fig. 2f, g). We next determined the redox balance by analyzing mitochondrial ROS (mito-ROS) and cytoplasmic ROS (cyto-ROS). As illustrated in Fig. 2 h and i , flow cytometry demonstrated that mito-ROS production was increased in mIRI-treated cells; the effect of which was reversed by Mff deletion. This result was further consolidated using immunofluorescence. As shown in Fig. 2j–l, either mito-ROS or cyto-ROS was agitated by mIRI in tubule cells. However, Mff knockout is followed by a repression of both mito-ROS and cyto-ROS, verifying the necessity of Mff in inducing oxidative stress.
Mff deletion improved mitochondrial quality control
Mitochondrial homeostasis plays a decisive role in the viability of tubule cells (Linkermann 2019). To further discern the role of upregulated Mff in tubular cell viability, we evaluated the function and morphology in mitochondria. Mitochondrial membrane potential, a recognized indicator of mitochondrial function, was dissipated by mIRI in vitro (Fig. 3a, b). However, Mff deletion significantly attenuated the mIRI-mediated mitochondrial depolarization. Given that SIRT1 and SIRT3 serve as highly conserved type III histone deacetylases pertinent to mitochondrial biogenesis (Ren et al. 2018), levels of these Sirtuins were evaluated. Our data revealed that mIRI significantly downregulated the levels of SIRT1/3, and the effect of which was obliterated by Mff knockout (Fig. 3c–e).
Mitochondrial quality control is modulated by Mff in ischemic AKI. a, b JC-1 probe was used to observe mitochondrial membrane potential. The red-to-green fluorescence intensity was evaluated as an index to quantify mitochondrial membrane potential. c–e Proteins were isolated after ischemic AKI and then western blotting was employed to observe levels of SIRT1 and SIRT3. f–h Immunofluorescence assay was used to evaluate alterations of mitochondrial fission. Average length of mitochondria was determined. Ratio of cells with fragmented mitochondria was also evaluated to quantify mitochondrial fission. Mean ± SEM, n = 6/group, #p < 0.05 vs. sham/control group; *p < 0.05 vs. IRI/mIRI group
With respect to mitochondrial structure, immunofluorescence assay demonstrated that cells subject to mIRI displayed shorter mitochondrial length. We then performed quantification analysis of mitochondrial cleavage by analyzing mitochondrial length in at least 100 cells. We also calculated the proportion of cells with fragmented mitochondria (Perry et al. 2018). As shown in Fig. 3f–h, mIRI treatment reduced mitochondrial length whereas Mff knockout elongated mitochondrial tubules. Along the same line, the percentage of cells with fragmented mitochondria was much higher in response to mIRI challenge, although such effect was mitigated in Mff-depleted cells. These results revealed a close tie between Mff deletion and the homeostatic mitochondrial quality control.
Decreased Pum2 augmented Mff translation and mitochondrial division
Although we previously reported that Mff phosphorylation and level were modulated by NR4A1 and DUSP1, respectively, in cardiac reperfusion injury (Jin et al. 2018; Zhou et al. 2018a), it remains elusive regarding the transcriptional regulation and mRNA stability of Mff. A recent study using aged mice revealed that Mff mRNA stability was negatively controlled by Pum2, a translation repressor binding to the mRNA encoding of Mff (D'Amico et al. 2019). In our current study, Western blot analysis confirmed a decline in the expression of Pum2 in tubular cells after IRI (Fig. 4a, b). Of note, Mff deletion had no influence on Pum2 expression (Fig. 4a, b), suggesting that Mff functions downstream of Pum2. To better understand the role of Pum2 in Mff-related mitochondrial cleavage and mitochondrial quality control, Pum2 was introduced using adenovirus. The adenovirus-mediated Pum2 overexpression (Pum2-OE) provoked a significant drop in the levels of Mff in tubular cells from WT mice under mIRI treatment, indicating that Pum2 acts as a repressor of Mff expression.
Pum2 negatively handles Mff in ischemic AKI. a, b Proteins were isolated after ischemic AKI and then western blotting was used to observe the levels of Pum2. c, d Pum2 adenovirus were delivered into WT cells to overexpress Pum2. Then, empty adenoviral vector was used as the control-OE (cont-OE) group. The expression of Mff was evaluated in response to Pum2-OE. e–g Immunofluorescence assay was used to observe the mitochondrial fission. Then, the average length of mitochondria and ratio of cells with fragmented mitochondria were determined to quantify mitochondrial fission. h, i Pum2 adenovirus and empty adenoviral vector were transfected into WT or Mff-KO cells in the presence or absence of mIRI. Then, mitochondrial potential was measured using JC-1 staining. The red-to-green fluorescence intensity was evaluated as an index to quantify mitochondrial membrane potential. j, k Pum2 adenovirus and empty adenoviral vector were transfected into WT or Mff-KO cells in the presence or absence of mIRI. ROS probe was used to observe levels of mitochondrial ROS (mito-ROS) using MitoSOX Red probe. Mean ± SEM, n = 6/group, #p < 0.05 vs. sham/control group; *p < 0.05 vs. IRI/ mIRI+cont-OE group
We then went on to examine the impact of Pum2-mediated Mff downregulation on mitochondrial mitosis. Immunofluorescence assay revealed that mIRI-mediated mitochondrial fragmentation was attenuated by Pum2-OE. Both average mitochondrial length and the fragmented mitochondria ratio were improved by Pum2-OE (Fig. 4e–g). To further confirm whether Pum2-offered mitochondrial protection is achieved through Mff repression, Pum2 overexpression was conducted in Mff-deleted cells prior to examination of mitochondrial potential and mito-ROS production. As shown in Fig. 4 h and i, decreased mitochondrial potential in mIRI-treated cells could be improved by Pum2-OE. However, in Mff-deleted cells under mIRI treatment, Pum2-OE had no additional protection on mitochondrial potential. In agreement with these findings, mito-ROS production was normalized by Pum2-OE in WT cells whereas little additional protective action was provided by Pum2-OE in Mff-deleted cells (Fig. 4j, k). These results describe that Pum2 regulates mitochondrial fission and mitochondrial homeostasis through reducing Mff expression.
Recovery of Pum2 sustained mitochondrial function and reduced AKI-mediated renal tubule damage
To discern whether Pum2 overexpression plays a protective role in attenuating renal tubule damage, we evaluated the phenotypic alterations of tubule cells with Pum2-OE. Initially, mitochondria-contained pro-apoptotic factors such as HtrA2/Omi were released from mitochondria, and the diffusion of HtrA2/Omi in the cytoplasm was noted in mIRI-treated cells (Fig. 5a–g). However, Pum2-OE prevented HtrA2/Omi effusion (Fig. 5a–g). Besides, we also found that the translocation of Drp1 and Bax from cytoplasm to mitochondria was induced by mIRI in WT cells; the effect of which was abrogated by Pum2-OE (Fig. 5a–g), suggesting that Pum2 may interrupt Drp1 and Bax migration onto mitochondria through inhibition of Mff transcription. In addition, cellular viability, as evaluated via CCK8, was inhibited by mIRI challenge and was reversed to near-normal levels once there is overexpression of Pum2 (Fig. 5h). In addition, mIRI-mediated pro-inflammatory response was also negatively controlled by Pum2-OE (Fig. 5i, j). These data suggest that Pum2 is capable of sustaining mitochondrial function and tubule cell viability.
Pum2 overexpression improves mitochondrial function and thus attenuates renal tubule damage. a–g Cytoplasmic and mitochondrial proteins were isolated in mIRI-treated tubular cells. Then, the expressions of HtrA2/Omi2, Drp1, and Bax in cytoplasm and mitochondria were determined using Western blotting. h CCK8 was employed to observe cell viability in response to Pum2 overexpression. i, j RNA was isolated from cells and then transcription of TGFα and IL-6 was analyzed after Pum2 overexpression. Mean ± SEM, n = 6/group, #p < 0.05 vs. sham/control group; *p < 0.05 vs. mIRI+cont-OE group
Discussion
Earlier evidence from our group has depicted a seemingly permissive role for Mff in the onset and development of myocardial IRI (Jin et al. 2018; Zhou et al. 2017). Mff deficiency impairs Drp1-mediated mitochondrial fission and thus maintains cardiac function by favoring mitochondrial function and cardiomyocyte viability. Recent studies have also revealed a rather important role for mitochondrial fragmentation in aggravating renal reperfusion injury (Perry et al. 2018). Although Drp1 is generally perceived as the primary mediator for mitochondrial fission, it does require a membrane receptor to dock safely and closely onto the outer mitochondrial membrane. However, there is little evidence illustrating the role of Drp1 receptors in the progression of acute tubular damage. Our current study validated the pathological contribution imposed by Mff in ischemic AKI. In an effort to dissect the relationship between Mff upregulation and pathological ischemic AKI, we found that genetic ablation of Mff partially retarded or reversed the pathological sequelae of ischemic AKI. Meanwhile, Mff deletion effectively rescued against AKI-induced oxidative stress and inflammation. In fact, the primary renoprotective mechanisms underlying Mff deletion are attributable to its role in mitochondrial fission regulation. As one of the Drp1 receptors, deletion of Mff was reported to directly compromise Drp1 localization onto mitochondria and therefore alleviate mitochondrial fission (Zhang et al. 2016). This notion is consistent with our current results that Mff downregulation, induced by Pum2 overexpression, blunt Drp1 translocation onto mitochondria, consolidating the obligatory role of Mff in Drp1-mediated fission. Despite these findings, post-transcriptional modification of Mff such as phosphorylation also plays a decisive role in promoting Drp1 recruitment and thus mitochondrial fission. For example, AMPK-mediated Mff phosphorylation at Ser172 enhances its affinity to Drp1, resulting in augmented mitochondrial fission in energy stress (Toyama et al. 2016). Moreover, succinate-induced Mff phosphorylation through ERK increases cardiomyocyte vulnerability to ischemic injury (Lu et al. 2018). However, the post-transcriptional modification mechanism underlying Mff in ischemic AKI has not been fully understood and future effort should focus on which amino acid residue(s) in Mff is prone to phosphorylation modification in tubular cells to facilitate Drp1 shuttle onto mitochondria.
Mitochondrial quality control requires a complex network encompassing mitochondrial dynamics, mitophagy, and mitochondrial biogenesis (Heusch 2018; Kalyanaraman et al. 2018). It ensures mitochondrial gene stability, ATP production, TCA-cycle metabolic substrate exchange, and cell oxidative status (Knupp et al. 2019; Xie et al. 2018a). Upon ischemic AKI challenge, several hostile damaging signals may build up and contribute to mitochondrial dysfunction (Mapuskar et al. 2019). In this study, our data support the dogma that Mff serves as an intricate checkpoint to functionally regulate mitochondrial quality through sustaining mitochondrial structure and function homeostasis at the sub-cellular levels. Beyond mitochondrial quality control, Mff-induced fission also governed renal oxidative stress and pro-inflammatory response. Mff reversed cellular antioxidative capacity in a manner dependent on GSH, GPx, and SIRT1/SIRT3. This effect might be driven through two independent mechanisms. First, inhibition of mitochondrial fission is reportedly to reduce the damage to mitochondrial DNA and the transcription of mitochondria-encoded electron transport chain, resulting into a decline in ROS production (Bao et al. 2019; Hasnat et al. 2019). Second, decreased mitochondrial fission improves the transcription of Nrf2 (Manczak et al. 2016; Wang et al. 2017), an important nuclear transcriptional signal to regulate a plethora of antioxidant enzymes (Chen et al. 2018; Ma 2013). Likely through promoting ROS generation and weakening antioxidative capacity, fragmented mitochondria directly provoke oxidative stress. With regard to inflammation, Drp1-related mitochondrial fission was shown to promote NLRP3 inflammasome formation (Zhou et al. 2016) and IL-1β production (Park et al. 2018), effects perceived to be secondary to oxidative stress triggered by excessive mitochondrial fission (Guo et al. 2018). The regulatory effects of Mff-induced mitochondrial fission on oxidative stress and inflammation may work in concert with disturbed mitochondrial quality control to sensitize kidney to ischemic AKI.
One intriguing finding from our current study is the decreased levels of Pum2 in ischemic AKI and protection against ischemic AKI-mediated Mff upregulation and renal tubular injury through overexpression of Pum2. Our recent studies illustrated that Mff expression and phosphorylation are modified by myocardial reperfusion injury through DUSP1 (Jin et al. 2018) and NR4A1 (Zhou et al. 2018a), respectively. These findings comprehensively described the post-transcriptional features of Mff. Here in this study, we employed an ischemic AKI model to evaluate the translation regulatory mechanisms for Mff and revealed a role for Pum2 as a transcriptional repressor for Mff (D'Amico et al. 2019). The regulatory mechanisms of Pum2 on Mff mRNA translation have been carefully examined elsewhere (Kanemaru et al. 2013). In addition to its classical role in regulating mRNA stabilization, Pum2 has been acknowledged as a pro-survival signal under various pathological conditions. The viability of cervical carcinoma (Duan et al. 2019), mesenchymal stem cells(Lee et al. 2019), breast cancer(Zhang et al. 2019a), and human dermal keratinocyte(Zhang et al. 2019b) is believed to be under the regulation of Pum2 independent of its action on mRNA translation. Pum2 works as a negative regulator of Jak and Runx2 pathways (Lee et al. 2019), the important signal transductors of cell survival and death (Aaronson and Horvath 2002). Second, Pum2 may control cell cycle through post-transcriptional repression of Cdkn1b (Lin et al. 2019a), a cell cycle inhibitor. Here in our hands, we mainly highlighted the pivotal role of Pum2 in regulating Mff translation and mitochondrial fission in ischemic AKI. Further study is warranted to discern the precise nature behind Pum2 downregulation in the setting of ischemic AKI challenge.
Conclusion
In summary, our current study has defined the role for Mff-driven mitochondrial mitosis in ischemic AKI and the negative regulation from Pum2 in the governance of Mff function and mitochondrial quality control, indicating the utility of Pum2-Mff in the therapeutics of ischemic AKI.
References
Aaronson DS, Horvath CM. A road map for those who don’t know JAK-STAT. Science. 2002;296:1653–5. https://doi.org/10.1126/science.1071545.
Bao D, Zhao J, Zhou X, Yang Q, Chen Y, Zhu J, et al. Mitochondrial fission-induced mtDNA stress promotes tumor-associated macrophage infiltration and HCC progression. Oncogene. 2019;38:5007–20. https://doi.org/10.1038/s41388-019-0772-z.
Bhargava P, Schnellmann RG. Mitochondrial energetics in the kidney. Nat Rev Nephrol. 2017;13:629–46. https://doi.org/10.1038/nrneph.2017.107.
Biernacki M, Ambrozewicz E, Gegotek A, Toczek M, Bielawska K, Skrzydlewska E. Redox system and phospholipid metabolism in the kidney of hypertensive rats after FAAH inhibitor URB597 administration. Redox Biol. 2018;15:41–50. https://doi.org/10.1016/j.redox.2017.11.022.
Chen T, Dai SH, Li X, Luo P, Zhu J, Wang YH, et al. Sirt1-Sirt3 axis regulates human blood-brain barrier permeability in response to ischemia. Redox Biol. 2018;14:229–36. https://doi.org/10.1016/j.redox.2017.09.016.
Curley D, Lavin Plaza B, Shah AM, Botnar RM. Molecular imaging of cardiac remodelling after myocardial infarction. Basic Res Cardiol. 2018;113:10. https://doi.org/10.1007/s00395-018-0668-z.
D'Amico D et al. (2019) The RNA-Binding Protein PUM2 Impairs Mitochondrial Dynamics and Mitophagy During Aging. Mol Cell 73:775-787 e710. https://doi.org/10.1016/j.molcel.2018.11.034.
DeLeon-Pennell KY, Mouton AJ, Ero OK, Ma Y, Padmanabhan Iyer R, Flynn ER, et al. LXR/RXR signaling and neutrophil phenotype following myocardial infarction classify sex differences in remodeling. Basic Res Cardiol. 2018;113:40. https://doi.org/10.1007/s00395-018-0699-5.
Duan W, Nian L, Qiao J, Liu NN. LncRNA TUG1 aggravates the progression of cervical cancer by binding PUM2. Eur Rev Med Pharmacol Sci. 2019;23:8211–8. https://doi.org/10.26355/eurrev_201910_19128.
Guo M, Wang X, Zhao Y, Yang Q, Ding H, Dong Q, et al. Ketogenic diet improves brain ischemic tolerance and inhibits NLRP3 inflammasome activation by preventing Drp1-mediated mitochondrial fission and endoplasmic reticulum stress. Front Mol Neurosci. 2018;11:86. https://doi.org/10.3389/fnmol.2018.00086.
Han W, Hu P, Wu F, Wang S, Hu Y, Li S, et al. FHL3 links cell growth and self-renewal by modulating SOX4 in glioma. Cell Death Differ. 2019;26:796–811. https://doi.org/10.1038/s41418-018-0152-1.
Hasnat M, Yuan Z, Naveed M, Khan A, Raza F, Xu D, et al. Drp1-associated mitochondrial dysfunction and mitochondrial autophagy: a novel mechanism in triptolide-induced hepatotoxicity. Cell Biol Toxicol. 2019;35:267–80. https://doi.org/10.1007/s10565-018-9447-8.
Heusch G. 25 years of remote ischemic conditioning: from laboratory curiosity to clinical outcome. Basic Res Cardiol. 2018;113:15. https://doi.org/10.1007/s00395-018-0673-2.
Huang S, Li Y, Yuan X, Zhao M, Wang J, Li Y, et al. The UbL-UBA Ubiquilin4 protein functions as a tumor suppressor in gastric cancer by p53-dependent and p53-independent regulation of p21. Cell Death Differ. 2019;26:516–30. https://doi.org/10.1038/s41418-018-0141-4.
Ji WK, Hatch AL, Merrill RA, Strack S, Higgs HN. Actin filaments target the oligomeric maturation of the dynamin GTPase Drp1 to mitochondrial fission sites. Elife. 2015;4:e11553. https://doi.org/10.7554/eLife.11553.
Jin Q, Li R, Hu N, Xin T, Zhu P, Hu S, et al. DUSP1 alleviates cardiac ischemia/reperfusion injury by suppressing the Mff-required mitochondrial fission and Bnip3-related mitophagy via the JNK pathways. Redox Biol. 2018;14:576–87. https://doi.org/10.1016/j.redox.2017.11.004.
Jung M, Dodsworth M, Thum T. Inflammatory cells and their non-coding RNAs as targets for treating myocardial infarction. Basic Res Cardiol. 2018;114:4. https://doi.org/10.1007/s00395-018-0712-z.
Kalyanaraman B, Cheng G, Hardy M, Ouari O, Lopez M, Joseph J, et al. A review of the basics of mitochondrial bioenergetics, metabolism, and related signaling pathways in cancer cells: therapeutic targeting of tumor mitochondria with lipophilic cationic compounds. Redox Biol. 2018;14:316–27. https://doi.org/10.1016/j.redox.2017.09.020.
Kanemaru K, Kubota J, Sekiya H, Hirose K, Okubo Y, Iino M. Calcium-dependent N-cadherin up-regulation mediates reactive astrogliosis and neuroprotection after brain injury. Proc Natl Acad Sci U S A. 2013;110:11612–7. https://doi.org/10.1073/pnas.1300378110.
Kim EH, Wong SW, Martinez J. Programmed necrosis and disease:we interrupt your regular programming to bring you necroinflammation. Cell Death Differ. 2019;26:25–40. https://doi.org/10.1038/s41418-018-0179-3.
Knupp J, Arvan P, Chang A. Increased mitochondrial respiration promotes survival from endoplasmic reticulum stress. Cell Death Differ. 2019;26:487–501. https://doi.org/10.1038/s41418-018-0133-4.
Kong MJ, Bak SH, Han KH, Kim JI, Park JW, Park KM. Fragmentation of kidney epithelial cell primary cilia occurs by cisplatin and these cilia fragments are excreted into the urine. Redox Biol. 2019;20:38–45. https://doi.org/10.1016/j.redox.2018.09.017.
Lee MH, Wu X, Zhu Y. RNA-binding protein PUM2 regulates mesenchymal stem cell fate via repression of JAK2 and RUNX2 mRNAs. J Cell Physiol. 2019. https://doi.org/10.1002/jcp.29281.
Li J, Cai SX, He Q, Zhang H, Friedberg D, Wang F, et al. Intravenous miR-144 reduces left ventricular remodeling after myocardial infarction. Basic Res Cardiol. 2018a;113:36. https://doi.org/10.1007/s00395-018-0694-x.
Li R, Xin T, Li D, Wang C, Zhu H, Zhou H. Therapeutic effect of Sirtuin 3 on ameliorating nonalcoholic fatty liver disease: the role of the ERK-CREB pathway and Bnip3-mediated mitophagy. Redox Biol. 2018b;18:229–43. https://doi.org/10.1016/j.redox.2018.07.011.
Lin K, et al. Mammalian Pum1 and Pum2 control body size via translational regulation of the cell cycle inhibitor Cdkn1b. Cell Rep. 2019a;26:2434–50 e2436. https://doi.org/10.1016/j.celrep.2019.01.111.
Lin Q, Li S, Jiang N, Shao X, Zhang M, Jin H, et al. PINK1-parkin pathway of mitophagy protects against contrast-induced acute kidney injury via decreasing mitochondrial ROS and NLRP3 inflammasome activation. Redox Biol. 2019b;26:101254. https://doi.org/10.1016/j.redox.2019.101254.
Linkermann A. Death and fire-the concept of necroinflammation. Cell Death Differ. 2019;26:1–3. https://doi.org/10.1038/s41418-018-0218-0.
Liu R, Chan DC. The mitochondrial fission receptor Mff selectively recruits oligomerized Drp1. Mol Biol Cell. 2015;26:4466–77. https://doi.org/10.1091/mbc.E15-08-0591.
Lu YT, Li LZ, Yang YL, Yin X, Liu Q, Zhang L, et al. Succinate induces aberrant mitochondrial fission in cardiomyocytes through GPR91 signaling. Cell Death Dis. 2018;9:672. https://doi.org/10.1038/s41419-018-0708-5.
Ma Q. Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol. 2013;53:401–26. https://doi.org/10.1146/annurev-pharmtox-011112-140320.
Manczak M, Kandimalla R, Fry D, Sesaki H, Reddy PH. Protective effects of reduced dynamin-related protein 1 against amyloid beta-induced mitochondrial dysfunction and synaptic damage in Alzheimer’s disease. Hum Mol Genet. 2016;25:5148–66. https://doi.org/10.1093/hmg/ddw330.
Mapuskar KA, Wen H, Holanda DG, Rastogi P, Steinbach E, Han R, et al. Persistent increase in mitochondrial superoxide mediates cisplatin-induced chronic kidney disease. Redox Biol. 2019;20:98–106. https://doi.org/10.1016/j.redox.2018.09.020.
Moore JB, et al. Epigenetically modified cardiac mesenchymal stromal cells limit myocardial fibrosis and promote functional recovery in a model of chronic ischemic cardiomyopathy. Basic Res Cardiol. 2018;114:3. https://doi.org/10.1007/s00395-018-0710-1.
Na HJ, Yeum CE, Kim HS, Lee J, Kim JY, Cho YS. TSPYL5-mediated inhibition of p53 promotes human endothelial cell function. Angiogenesis. 2019;22:281–93. https://doi.org/10.1007/s10456-018-9656-z.
Osellame LD, Singh AP, Stroud DA, Palmer CS, Stojanovski D, Ramachandran R, et al. Cooperative and independent roles of the Drp1 adaptors Mff, MiD49 and MiD51 in mitochondrial fission. J Cell Sci. 2016;129:2170–81. https://doi.org/10.1242/jcs.185165.
Otera H, Wang C, Cleland MM, Setoguchi K, Yokota S, Youle RJ, et al. Mff is an essential factor for mitochondrial recruitment of Drp1 during mitochondrial fission in mammalian cells. J Cell Biol. 2010;191:1141–58. https://doi.org/10.1083/jcb.201007152.
Park HS, Liu G, Liu Q, Zhou Y (2018) Swine influenza virus induces RIPK1/DRP1-mediated interleukin-1 beta production. Viruses 10. https://doi.org/10.3390/v10080419.
Perry HM, et al. Dynamin-related protein 1 deficiency promotes recovery from AKI. J Am Soc Nephrol. 2018;29:194–206. https://doi.org/10.1681/ASN.2017060659.
Qian X, Du Y, Jiang G, Lin F, Yao L. Survival motor neuron (SMN) protein insufficiency exacerbates renal ischemia/reperfusion injury. Front Physiol. 2019;10:559. https://doi.org/10.3389/fphys.2019.00559.
Ren W, et al. Melatonin alleviates weanling stress in mice: involvement of intestinal microbiota. J Pineal Res. 2018;64. https://doi.org/10.1111/jpi.12448.
Sajib S, Zahra FT, Lionakis MS, German NA, Mikelis CM. Mechanisms of angiogenesis in microbe-regulated inflammatory and neoplastic conditions. Angiogenesis. 2018;21:1–14. https://doi.org/10.1007/s10456-017-9583-4.
Sedlackova L, Korolchuk VI. Mitochondrial quality control as a key determinant of cell survival. Biochim Biophys Acta Mol Cell Res. 2019;1866:575–87. https://doi.org/10.1016/j.bbamcr.2018.12.012.
Soranno DE, Gil HW, Kirkbride-Romeo L, Altmann C, Montford JR, Yang H, et al. Matching human unilateral AKI, a reverse translational approach to investigate kidney recovery after ischemia. J Am Soc Nephrol. 2019;30:990–1005. https://doi.org/10.1681/ASN.2018080808.
Sun J, et al. Mitochondria in sepsis-induced AKI. J Am Soc Nephrol. 2019. https://doi.org/10.1681/ASN.2018111126.
Tahrir FG, Langford D, Amini S, Mohseni Ahooyi T, Khalili K. Mitochondrial quality control in cardiac cells: mechanisms and role in cardiac cell injury and disease. J Cell Physiol. 2019;234:8122–33. https://doi.org/10.1002/jcp.27597.
Tanaka T, Nangaku M. Regulatory roles of hypoxia-inducible, noncoding RNAs on mitochondrial dynamics during AKI. Kidney Int. 2019;95:252–3. https://doi.org/10.1016/j.kint.2018.12.002.
Toyama EQ, Herzig S, Courchet J, Lewis TL Jr, Losón OC, Hellberg K, Young NP, Chen H, Polleux F, Chan DC, Shaw RJ (2016) Metabolism. AMP-activated protein kinase mediates mitochondrial fission in response to energy stress Science 351:275–281. doi:https://doi.org/10.1126/science.aab4138.
Vial J, Royet A, Cassier P, Tortereau A, Dinvaut S, Maillet D, et al. The Ectodysplasin receptor EDAR acts as a tumor suppressor in melanoma by conditionally inducing cell death. Cell Death Differ. 2019;26:443–54. https://doi.org/10.1038/s41418-018-0128-1.
Wang S, Zhu X, Xiong L, Ren J. Ablation of Akt2 prevents paraquat-induced myocardial mitochondrial injury and contractile dysfunction: role of Nrf2. Toxicol Lett. 2017;269:1–14. https://doi.org/10.1016/j.toxlet.2017.01.009.
Xie X, Venit T, Drou N, Percipalle P (2018a) In mitochondria ?-actin regulates mtDNA transcription and is required for mitochondrial quality control. iScience 3:226-237. doi:https://doi.org/10.1016/j.isci.2018.04.021.
Xie Y, Jiang D, Xiao J, Fu C, Zhang Z, Ye Z, et al. Ischemic preconditioning attenuates ischemia/reperfusion-induced kidney injury by activating autophagy via the SGK1 signaling pathway. Cell Death Dis. 2018b;9:338. https://doi.org/10.1038/s41419-018-0358-7.
Zhang L, Chen Y, Li C, Liu J, Ren H, Li L, et al. RNA binding protein PUM2 promotes the stemness of breast cancer cells via competitively binding to neuropilin-1 (NRP-1) mRNA with miR-376a. Biomed Pharmacother. 2019a;114:108772. https://doi.org/10.1016/j.biopha.2019.108772.
Zhang X, Chen L, Xiao B, Liu H, Su Y. Circ_0075932 in adipocyte-derived exosomes induces inflammation and apoptosis in human dermal keratinocytes by directly binding with PUM2 and promoting PUM2-mediated activation of AuroraA/NF-kappaB pathway. Biochem Biophys Res Commun. 2019b;511:551–8. https://doi.org/10.1016/j.bbrc.2019.02.082.
Zhang Z, Liu L, Wu S, Xing D. Drp1, Mff, Fis1, and MiD51 are coordinated to mediate mitochondrial fission during UV irradiation-induced apoptosis. FASEB J. 2016;30:466–76. https://doi.org/10.1096/fj.15-274258.
Zhou H et al. (2017) Mff-dependent mitochondrial fission contributes to the pathogenesis of cardiac microvasculature ischemia/reperfusion injury via induction of mROS-mediated Cardiolipin oxidation and HK2/VDAC1 disassociation-involved mPTP opening. J Am Heart Assoc 6. doi:https://doi.org/10.1161/JAHA.116.005328.
Zhou H, Wang J, Zhu P, Zhu H, Toan S, Hu S, et al. NR4A1 aggravates the cardiac microvascular ischemia reperfusion injury through suppressing FUNDC1-mediated mitophagy and promoting Mff-required mitochondrial fission by CK2alpha. Basic Res Cardiol. 2018a;113:23. https://doi.org/10.1007/s00395-018-0682-1.
Zhou H, Zhu P, Wang J, Zhu H, Ren J, Chen Y. Pathogenesis of cardiac ischemia reperfusion injury is associated with CK2alpha-disturbed mitochondrial homeostasis via suppression of FUNDC1-related mitophagy. Cell Death Differ. 2018b;25:1080–93. https://doi.org/10.1038/s41418-018-0086-7.
Zhou Z, et al. PEDF inhibits the activation of NLRP3 Inflammasome in hypoxia cardiomyocytes through PEDF receptor/phospholipase A2. Int J Mol Sci. 2016;17. https://doi.org/10.3390/ijms17122064.
Zhu G, et al. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J Cell Physiol. 2019. https://doi.org/10.1002/jcp.28941.
Zhu H, Jin Q, Li Y, Ma Q, Wang J, Li D, et al. Melatonin protected cardiac microvascular endothelial cells against oxidative stress injury via suppression of IP3R-[Ca(2+)]c/VDAC-[Ca(2+)]m axis by activation of MAPK/ERK signaling pathway. Cell Stress Chaperones. 2018a;23:101–13. https://doi.org/10.1007/s12192-017-0827-4.
Zhu P, Hu S, Jin Q, Li D, Tian F, Toan S, et al. Ripk3 promotes ER stress-induced necroptosis in cardiac IR injury: a mechanism involving calcium overload/XO/ROS/mPTP pathway. Redox Biol. 2018b;16:157–68. https://doi.org/10.1016/j.redox.2018.02.019.
Funding
This work was supported in part by China Postdoctoral Science Foundation (2019TQ0128) and the NSFC (81900252, 81900254 and 91749128).
Author information
Authors and Affiliations
Contributions
HZ, JW, and JR were involved in the conception and design of the study, performance of experiments, data analysis and interpretation, and manuscript writing. RBL and JW were involved in development of the methodology. HZ and PJZ acquired the data. JW and ST analyzed and interpreted data. JR and HZ obtained financial support, supervised the study, and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 11192 kb)
Rights and permissions
About this article
Cite this article
Wang, J., Zhu, P., Toan, S. et al. Pum2-Mff axis fine-tunes mitochondrial quality control in acute ischemic kidney injury. Cell Biol Toxicol 36, 365–378 (2020). https://doi.org/10.1007/s10565-020-09513-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-020-09513-9