Abstract
Serine/arginine (SR) proteins play significant roles in pre-mRNA splicing in eukaryotes. To investigate how gene expression influences fungal development and pathogenicity in Fusarium graminearum, a causal agent of Fusarium head blight (FHB) of wheat and barley, our previous study identified a SR protein FgSrp1 in F. graminearum, and showed that it is important for conidiation, plant infection and pre-mRNA processing. In this study, we identified another SR protein FgSrp2 in F. graminearum, which is orthologous to Schizosaccharomyces pombe Srp2. Our data showed that, whereas yeast Srp2 is essential for growth, deletion of FgSRP2 resulted in only slight defects in vegetative growth and perithecia melanization. FgSrp2 localized to the nucleus and both its N- and C-terminal regions were important for the localization to the nucleus. FgSrp2 interacted with FgSrp1 to form a complex in vivo. Double deletion of FgSRP1 and FgSRP2 revealed that they had overlapping functions in vegetative growth and sexual reproduction. RNA-seq analysis revealed that, although deletion of FgSRP2 alone had minimal effects, deletion of both FgSRP1 and FgSRP2 caused significant changes in gene transcription and RNA splicing. Overall, our results indicated that FgSrp2 regulates vegetative growth, sexual reproduction and pre-mRNA processing by interacting with FgSrp1.






Similar content being viewed by others
References
Ariyachet C, Solis NV, Liu Y, Prasadarao NV, Filler SG, McBride AE (2013) SR-like RNA-binding protein Slr1 affects Candida albicans filamentation and virulence. Infect Immun 81:1267–1276. https://doi.org/10.1128/IAI.00864-12
Bai G, Shaner G (2004) Management and resistance in wheat and barley to fusarium head blight. Annu Rev Phytopathol 42:135–161. https://doi.org/10.1146/annurev.phyto.42.040803.140340
Black DL (2003) Mechanisms of alternative pre-messenger RNA splicing. Annu Rev Biochem 72:291–336. https://doi.org/10.1146/annurev.biochem.72.121801.161720
Bossie MA, DeHoratius C, Barcelo G, Silver P (1992) A mutant nuclear protein with similarity to RNA binding proteins interferes with nuclear import in yeast. Mol Biol Cell 3:875–893. https://doi.org/10.1091/mbc.3.8.875
Bruno KS, Tenjo F, Li L, Hamer JE, Xu JR (2004) Cellular localization and role of kinase activity of PMK1 in Magnaporthe grisea. Eukaryot Cell 3:1525–1532. https://doi.org/10.1128/ec.3.6.1525-1532.2004
Busch A, Hertel KJ (2012) Evolution of SR protein and hnRNP splicing regulatory factors. Wiley Interdiscip Rev RNA 3:1–12. https://doi.org/10.1002/wrna.100
Catlett NL, Lee B-N, Yoder OC, Turgeon BG (2003) Split-marker recombination for efficient targeted deletion of fungal genes. Fungal Genet Rep 50:9–11. https://doi.org/10.4148/1941-4765.1150
Chen D, Wang Y, Zhou X, Wang Y, Xu JR (2014) The Sch9 kinase regulates conidium size, stress responses, and pathogenesis in Fusarium graminearum. PLoS ONE 9:e105811. https://doi.org/10.1371/journal.pone.0105811
Chen L, Tong Q, Zhang C, Ding K (2019) The transcription factor FgCrz1A is essential for fungal development, virulence, deoxynivalenol biosynthesis and stress responses in Fusarium graminearum. Curr Genet 65:153–166. https://doi.org/10.1007/s00294-018-0853-5
Cuomo CA, Guldener U, Xu JR, Trail F, Turgeon BG, Di Pietro A, Walton JD, Ma LJ, Baker SE, Rep M, Adam G, Antoniw J, Baldwin T, Calvo S, Chang YL, Decaprio D, Gale LR, Gnerre S, Goswami RS, Hammond-Kosack K, Harris LJ, Hilburn K, Kennell JC, Kroken S, Magnuson JK, Mannhaupt G, Mauceli E, Mewes HW, Mitterbauer R, Muehlbauer G, Munsterkotter M, Nelson D, O'Donnell K, Ouellet T, Qi W, Quesneville H, Roncero MI, Seong KY, Tetko IV, Urban M, Waalwijk C, Ward TJ, Yao J, Birren BW, Kistler HC (2007) The Fusarium graminearum genome reveals a link between localized polymorphism and pathogen specialization. Science 317:1400–1402. https://doi.org/10.1126/science.1143708
Dean R, Van Kan JA, Pretorius ZA, Hammond-Kosack KE, Di Pietro A, Spanu PD, Rudd JJ, Dickman M, Kahmann R, Ellis J, Foster GD (2012) The top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol 13:414–430. https://doi.org/10.1111/j.1364-3703.2011.00783.x
Dimont E, Shi J, Kirchner R, Hide W (2015) edgeRun: an R package for sensitive, functionally relevant differential expression discovery using an unconditional exact test. Bioinformatics 31:2589–2590. https://doi.org/10.1093/bioinformatics/btv209
Erkelenz S, Mueller WF, Evans MS, Busch A, Schoneweis K, Hertel KJ, Schaal H (2013) Position-dependent splicing activation and repression by SR and hnRNP proteins rely on common mechanisms. RNA (New York, NY) 19:96–102. https://doi.org/10.1261/rna.037044.112
Gale LR, Ward TJ, Balmas V, Kistler HC (2007) Population subdivision of Fusarium graminearum sensu stricto in the upper Midwestern United States. Phytopathology 97:1434–1439. https://doi.org/10.1094/phyto-97-11-1434
Gao X, Jin Q, Jiang C, Li Y, Li C, Liu H, Kang Z, Xu JR (2016) FgPrp4 kinase is important for spliceosome B-complex activation and splicing efficiency in Fusarium graminearum. PLoS Genet 12:e1005973. https://doi.org/10.1371/journal.pgen.1005973
Goswami RS, Kistler HC (2004) Heading for disaster: Fusarium graminearum on cereal crops. Mol Plant Pathol 5:515–525. https://doi.org/10.1111/j.1364-3703.2004.00252.x
Gross T, Richert K, Mierke C, Lutzelberger M, Kaufer NF (1998) Identification and characterization of srp1, a gene of fission yeast encoding a RNA binding domain and a RS domain typical of SR splicing factors. Nucleic Acids Res 26:505–511
Guindon S, Delsuc F, Dufayard JF, Gascuel O (2009) Estimating maximum likelihood phylogenies with PhyML. Methods Mol Biol (Clifton, NJ) 537:113–137. https://doi.org/10.1007/978-1-59745-251-9_6
Hou Z, Xue C, Peng Y, Katan T, Kistler HC, Xu JR (2002) A mitogen-activated protein kinase gene (MGV1) in Fusarium graminearum is required for female fertility, heterokaryon formation, and plant infection. Mol Plant-Microbe Interact MPMI 15:1119–1127. https://doi.org/10.1094/mpmi.2002.15.11.1119
Huang Y, Steitz JA (2001) Splicing factors SRp20 and 9G8 promote the nucleocytoplasmic export of mRNA. Mol Cell 7:899–905
Huang Y, Steitz JA (2005) SRprises along a messenger's journey. Mol Cell 17:613–615. https://doi.org/10.1016/j.molcel.2005.02.020
Huang Y, Yario TA, Steitz JA (2004) A molecular link between SR protein dephosphorylation and mRNA export. Proc Natl Acad Sci USA 101:9666–9670. https://doi.org/10.1073/pnas.0403533101
Jeong S (2017) SR proteins: binders, regulators, and connectors of RNA. Mol Cells 40:1–9. https://doi.org/10.14348/molcells.2017.2319
Jia LJ, Tang WH (2015) The omics era of Fusarium graminearum: opportunities and challenges. New Phytol 207:1–3. https://doi.org/10.1111/nph.13457
Jiang C, Cao S, Wang Z, Xu H, Liang J, Liu H, Wang G, Ding M, Wang Q, Gong C, Feng C, Hao C, Xu JR (2019) An expanded subfamily of G-protein-coupled receptor genes in Fusarium graminearum required for wheat infection. Nat Microbiol 4:1582–1591. https://doi.org/10.1038/s41564-019-0468-8
Kazan K, Gardiner DM (2018) Transcriptomics of cereal-Fusarium graminearum interactions: what we have learned so far. Mol Plant Pathol 19:764–778. https://doi.org/10.1111/mpp.12561
Kim D, Langmead B, Salzberg SL (2015a) HISAT: a fast-spliced aligner with low memory requirements. Nat Methods 12:357–360. https://doi.org/10.1038/nmeth.3317
Kim HK, Jo SM, Kim GY, Kim DW, Kim YK, Yun SH (2015b) A large-scale functional analysis of putative target genes of mating-type loci provides insight into the regulation of sexual development of the cereal pathogen Fusarium graminearum. PLoS Genet 11:e1005486. https://doi.org/10.1371/journal.pgen.1005486
King R, Urban M, Hammond-Kosack MC, Hassani-Pak K, Hammond-Kosack KE (2015) The completed genome sequence of the pathogenic ascomycete fungus Fusarium graminearum. BMC Genom 16:544. https://doi.org/10.1186/s12864-015-1756-1
Kress TL, Krogan NJ, Guthrie C (2008) A single SR-like protein, Npl3, promotes pre-mRNA splicing in budding yeast. Mol Cell 32:727–734. https://doi.org/10.1016/j.molcel.2008.11.013
Li Y, Chen D, Luo S, Zhu Y, Jia X, Duan Y, Zhou M (2019) Intron-mediated regulation of beta-tubulin genes expression affects the sensitivity to carbendazim in Fusarium graminearum. Curr Genet 65:1057–1069. https://doi.org/10.1007/s00294-019-00960-4
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930. https://doi.org/10.1093/bioinformatics/btt656
Liu W, Zhou X, Li G, Li L, Kong L, Wang C, Zhang H, Xu JR (2011) Multiple plant surface signals are sensed by different mechanisms in the rice blast fungus for appressorium formation. PLoS Pathog 7:e1001261. https://doi.org/10.1371/journal.ppat.1001261
Liu H, Zhang S, Ma J, Dai Y, Li C, Lyu X, Wang C, Xu JR (2015) Two Cdc2 kinase genes with distinct functions in vegetative and infectious hyphae in Fusarium graminearum. PLoS Pathog 11:e1004913. https://doi.org/10.1371/journal.ppat.1004913
Liu H, Wang Q, He Y, Chen L, Hao C, Jiang C, Li Y, Dai Y, Kang Z, Xu JR (2016) Genome-wide A-to-I RNA editing in fungi independent of ADAR enzymes. Genome Res 26:499–509. https://doi.org/10.1101/gr.199877.115
Liu H, Li Y, Chen D, Qi Z, Wang Q, Wang J, Jiang C, Xu JR (2017) A-to-I RNA editing is developmentally regulated and generally adaptive for sexual reproduction in Neurospora crassa. Proc Natl Acad Sci USA 114:E7756–E7765. https://doi.org/10.1073/pnas.1702591114
Long JC, Caceres JF (2009) The SR protein family of splicing factors: master regulators of gene expression. Biochem J 417:15–27. https://doi.org/10.1042/BJ20081501
Lutzelberger M, Gross T, Kaufer NF (1999) Srp2, an SR protein family member of fission yeast: in vivo characterization of its modular domains. Nucleic Acids Res 27:2618–2626
Lv W, Wu J, Xu Z, Dai H, Ma Z, Wang Z (2019) The putative histone-like transcription factor FgHltf1 is required for vegetative growth, sexual reproduction, and virulence in Fusarium graminearum. Curr Genet 65:981–994. https://doi.org/10.1007/s00294-019-00953-3
Nguyen Ba AN, Pogoutse A, Provart N, Moses AM (2009) NLStradamus: a simple Hidden Markov Model for nuclear localization signal prediction. BMC Bioinform 10:202. https://doi.org/10.1186/1471-2105-10-202
Pandit S, Zhou Y, Shiue L, Coutinho-Mansfield G, Li H, Qiu J, Huang J, Yeo GW, Ares M Jr, Fu XD (2013) Genome-wide analysis reveals SR protein cooperation and competition in regulated splicing. Mol Cell 50:223–235. https://doi.org/10.1016/j.molcel.2013.03.001
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33:290–295. https://doi.org/10.1038/nbt.3122
Plass M, Agirre E, Reyes D, Camara F, Eyras E (2008) Co-evolution of the branch site and SR proteins in eukaryotes. Trends Genet 24:590–594. https://doi.org/10.1016/j.tig.2008.10.004
Seo JA, Kim JC, Lee DH, Lee YW (1996) Variation in 8-ketotrichothecenes and zearalenone production by Fusarium graminearum isolates from corn and barley in Korea. Mycopathologia 134:31–37. https://doi.org/10.1007/bf00437050
Shaw BD, Upadhyay S (2005) Aspergillus nidulans swoK encodes an RNA binding protein that is important for cell polarity. Fungal Genet Biol 42:862–872. https://doi.org/10.1016/j.fgb.2005.06.002
Son H, Park AR, Lim JY, Shin C, Lee YW (2017) Genome-wide exonic small interference RNA-mediated gene silencing regulates sexual reproduction in the homothallic fungus Fusarium graminearum. PLoS Genet 13:e1006595. https://doi.org/10.1371/journal.pgen.1006595
Tang Z, Kaufer NF, Lin RJ (2002) Interactions between two fission yeast serine/arginine-rich proteins and their modulation by phosphorylation. Biochem J 368:527–534. https://doi.org/10.1042/bj20021133
Tang Z, Tsurumi A, Alaei S, Wilson C, Chiu C, Oya J, Ngo B (2007) Dsk1p kinase phosphorylates SR proteins and regulates their cellular localization in fission yeast. Biochem J 405:21–30. https://doi.org/10.1042/BJ20061523
Wang C, Zhang S, Hou R, Zhao Z, Zheng Q, Xu Q, Zheng D, Wang G, Liu H, Gao X, Ma JW, Kistler HC, Kang Z, Xu JR (2011) Functional analysis of the kinome of the wheat scab fungus Fusarium graminearum. PLoS Pathog 7:e1002460. https://doi.org/10.1371/journal.ppat.1002460
Wang G, Sun P, Gong Z, Gu L, Lou Y, Fang W, Zhang L, Su L, Yang T, Wang B, Zhou J, Xu JR, Wang Z, Zheng W (2018) Srk1 kinase, a SR protein-specific kinase, is important for sexual reproduction, plant infection and pre-mRNA processing in Fusarium graminearum. Environ Microbiol 20:3261–3277. https://doi.org/10.1111/1462-2920.14299
Wu W, Zong J, Wei N, Cheng J, Zhou X, Cheng Y, Chen D, Guo Q, Zhang B, Feng Y (2018) CASH: a constructing comprehensive splice site method for detecting alternative splicing events. Brief Bioinform 19:905–917. https://doi.org/10.1093/bib/bbx034
Xu L, Wang M, Tang G, Ma Z, Shao W (2019) The endocytic cargo adaptor complex is required for cell-wall integrity via interacting with the sensor FgWsc2B in Fusarium graminearum. Curr Genet 65:1071–1080. https://doi.org/10.1007/s00294-019-00961-3
Yang P, Chen Y, Wu H, Fang W, Liang Q, Zheng Y, Olsson S, Zhang D, Zhou J, Wang Z, Zheng W (2018) The 5-oxoprolinase is required for conidiation, sexual reproduction, virulence and deoxynivalenol production of Fusarium graminearum. Curr Genet 64:285–301. https://doi.org/10.1007/s00294-017-0747-y
Zhang Y, Gao X, Sun M, Liu H, Xu JR (2017) The FgSRP1 SR-protein gene is important for plant infection and pre-mRNA processing in Fusarium graminearum. Environ Microbiol 19:4065–4079. https://doi.org/10.1111/1462-2920.13844
Zhang L, Wang L, Liang Y, Yu J (2019) FgPEX4 is involved in development, pathogenicity, and cell wall integrity in Fusarium graminearum. Curr Genet 65:747–758. https://doi.org/10.1007/s00294-018-0925-6
Zhao X, Xu JR (2007) A highly conserved MAPK-docking site in Mst7 is essential for Pmk1 activation in Magnaporthe grisea. Mol Microbiol 63:881–894. https://doi.org/10.1111/j.1365-2958.2006.05548.x
Zhao Z, Liu H, Luo Y, Zhou S, An L, Wang C, Jin Q, Zhou M, Xu JR (2014) Molecular evolution and functional divergence of tubulin superfamily in the fungal tree of life. Sci Rep 4:6746. https://doi.org/10.1038/srep06746
Zheng Q, Hou R, Juanyu Z, Ma J, Wu Z, Wang G, Wang C, Xu JR (2013) The MAT locus genes play different roles in sexual reproduction and pathogenesis in Fusarium graminearum. PLoS ONE 8:e66980. https://doi.org/10.1371/journal.pone.0066980
Zhou Z, Fu XD (2013) Regulation of splicing by SR proteins and SR protein-specific kinases. Chromosoma 122:191–207. https://doi.org/10.1007/s00412-013-0407-z
Zhou X, Heyer C, Choi YE, Mehrabi R, Xu JR (2010) The CID1 cyclin C-like gene is important for plant infection in Fusarium graminearum. Fungal Genet Biol 47:143–151. https://doi.org/10.1016/j.fgb.2009.11.001
Acknowledgements
We thank Dr. Larry Dunkle at Purdue University for language editing, and Drs. Cong Jiang, Qinhu Wang, and Guanghui Wang at Northwest A&F University for fruitful discussions. We also thank Xuli Gao, Shulin Cao and Chaoni Song for assistance with wheat head infection and DON production assays. This work was supported by the National Natural Science Foundation of China (No. 31671981), Project of Science and Technology New Star of Shaanxi Province (No. 2017KJXX-77), and National Undergraduate Training Program for Innovation and Entrepreneurship (No. 201810712104).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Editorial responsibility: M. Kupiec.
Electronic supplementary material
Below is the link to the electronic supplementary material.
294_2020_1054_MOESM1_ESM.pdf
Figure S1. Multiple alignments of FgSrp2 with its orthologs from other fungi. The two conserved RRM (RNA recognition motif) domains are marked with blue boxes. The one R (arginine)-rich region is marked with a line below the alignment. The NLS (nuclear localization signal) is marked with red boxes. IDs of proteins were described as follows: FgSrp2 (FGRAMPH1_01G27477 or FGSG_09282), Fusarium graminearum; NCU07069, Neurospora crassa; ANIA_04978, Aspergillus nidulans; Mycgr3T99129, Zymoseptoria tritici; SS1G_14437, Sclerotinia sclerotiorum; srp2 (SPAC16.02c), Schizosaccharomyces pombe; CaNpl3 (MEK_01413), Candida albicans; NPL3 (YDR432W), Saccharomyces cerevisiae; CC1G_14612, Coprinopsis cinerea (PDF 1054 kb)
294_2020_1054_MOESM2_ESM.pdf
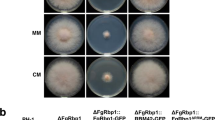
Figure S2. Phenotypes of wild type (PH-1), Fgsrp2 deletion mutant, and transformants of Fgsrp1 expressing the FgSRP2∆N-GFP or FgSRP2∆NLS-GFP alleles, and of wild type expressing the FgSRP2∆C-GFP allele in growth and sexual reproduction. Growth was assayed on three-day old PDA cultures. Sexual reproduction was assayed on carrot agar cultures. Perithecia, ascus and ascospore were examined after 8 days post-fertilization (PDF 528 kb)
294_2020_1054_MOESM3_ESM.tif
Figure S3. Expression levels of FgSRP1 and FgSRP2 genes in Fgsrp2 and Fgsrp1 deletion mutants, respectively. The relative expression of FgSRP1 and FgSRP2 genes was assayed by qRT-PCR with RNA isolated from 12-h germlings of the wild-type PH-1, and the Fgsrp1 and Fgsrp2 deletion mutants. The expression levels of FgSRP1 and FgSRP2 gene in PH-1 were arbitrarily set to 1. Mean and standard (TIF 404 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Dai, Y., Huang, Y. et al. The SR-protein FgSrp2 regulates vegetative growth, sexual reproduction and pre-mRNA processing by interacting with FgSrp1 in Fusarium graminearum. Curr Genet 66, 607–619 (2020). https://doi.org/10.1007/s00294-020-01054-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-020-01054-2