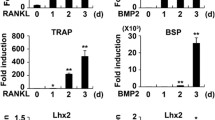
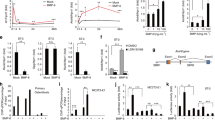
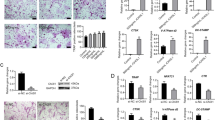
Abstract
Cleidocranial dysplasia is an autosomal dominant skeletal disorder resulting from RUNX2 mutations. The influence of RUNX2 mutations on osteoclastogenesis and bone resorption have not been reported. To investigate the role of RUNX2 in osteoclast, RUNX2 expression in macrophages (RAW 264.7 cells) was detected. Stable RAW 264.7 cell lines expressing wild-type RUNX2 or mutated RUNX2 (c.514delT, p.172 fs) were established, and their functions in osteoclasts were investigated. Wild-type RUNX2 promoted osteoclast differentiation, formation of F-actin ring, and bone resorption, while mutant RUNX2 attenuated the positive differentiation effect. Wild-type RUNX2 increased the expression and activity of mTORC2. Subsequently, mTORC2 specifically promoted phosphorylation of AKT at the serine 473 residue. Activated AKT improved the nuclear translocation of NFATc1 and increased the expression of downstream genes, including CTSK. Inhibition of AKT phosphorylation abrogated the osteoclast formation of wild-type macrophages, whereas constitutively activated AKT rescued the osteoclast formation of mutant macrophages. The present study suggested that RUNX2 promotes osteoclastogenesis and bone resorption through the AKT/NFATc1/CTSK axis. Mutant RUNX2 lost the function of regulating osteoclast differentiation and bone remodeling, resulting in the defective formation of the tooth eruption pathway and impaction of permanent teeth in cleidocranial dysplasia. This study, for the first time, verifies the effect of RUNX2 on osteoclast differentiation and bone resorption and provides new insight for the explanation of cleidocranial dysplasia.








Similar content being viewed by others
References
Ott CE, Leschik G, Trotier F, Brueton L, Brunner HG, Brussel W, Guillen-Navarro E, Haase C, Kohlhase J, Kotzot D, Lane A, Lee-Kirsch MA, Morlot S, Simon ME, Steichen-Gersdorf E, Tegay DH, Peters H, Mundlos S, Klopocki E (2010) Deletions of the RUNX2 gene are present in about 10% of individuals with cleidocranial dysplasia. Hum Mutat 31(8):E1587–1593. https://doi.org/10.1002/humu.21298
Cooper SC, Flaitz CM, Johnston DA, Lee B, Hecht JT (2001) A natural history of cleidocranial dysplasia. Am J Med Genet 104(1):1–6
Zhang CY, Zheng SG, Wang YX, Zhu JX, Zhu X, Zhao YM, Ge LH (2009) Novel RUNX2 mutations in Chinese individuals with cleidocranial dysplasia. J Dental Res 88(9):861–866. https://doi.org/10.1177/0022034509342083
Jaruga A, Hordyjewska E, Kandzierski G, Tylzanowski P (2016) Cleidocranial dysplasia and RUNX2-clinical phenotype-genotype correlation. Clin Genet 90(5):393–402. https://doi.org/10.1111/cge.12812
Wise GE (2009) Cellular and molecular basis of tooth eruption. Orthod Cranio Res 12(2):67–73. https://doi.org/10.1111/j.1601-6343.2009.01439.x
Wise GE, He H, Gutierrez DL, Ring S, Yao S (2011) Requirement of alveolar bone formation for eruption of rat molars. Eur J Oral Sci 119(5):333–338. https://doi.org/10.1111/j.1600-0722.2011.00854.x
Wise GE, Yao S (2006) Regional differences of expression of bone morphogenetic protein-2 and RANKL in the rat dental follicle. Eur J Oral Sci 114(6):512–516. https://doi.org/10.1111/j.1600-0722.2006.00406.x
Castaneda B, Simon Y, Jacques J, Hess E, Choi YW, Blin-Wakkach C, Mueller C, Berdal A, Lezot F (2011) Bone resorption control of tooth eruption and root morphogenesis: involvement of the receptor activator of NF-kappaB (RANK). J Cell Physiol 226(1):74–85. https://doi.org/10.1002/jcp.22305
Wise GE, Yao S, Henk WG (2007) Bone formation as a potential motive force of tooth eruption in the rat molar. Clin Anat 20(6):632–639. https://doi.org/10.1002/ca.20495
Takarada T, Hinoi E, Nakazato R, Ochi H, Xu C, Tsuchikane A, Takeda S, Karsenty G, Abe T, Kiyonari H, Yoneda Y (2013) An analysis of skeletal development in osteoblast-specific and chondrocyte-specific runt-related transcription factor-2 (Runx2) knockout mice. J Bone Miner Res 28(10):2064–2069. https://doi.org/10.1002/jbmr.1945
Chen P, Wei D, Xie B, Ni J, Xuan D, Zhang J (2014) Effect and possible mechanism of network between microRNAs and RUNX2 gene on human dental follicle cells. J Cell Biochem 115(2):340–348. https://doi.org/10.1002/jcb.24668
Yan WJ, Zhang CY, Yang X, Liu ZN, Wang XZ, Sun XY, Wang YX, Zheng SG (2015) Abnormal differentiation of dental pulp cells in cleidocranial dysplasia. J Dental Res 94(4):577–583. https://doi.org/10.1177/0022034514566655
Udagawa N, Takahashi N, Akatsu T, Tanaka H, Sasaki T, Nishihara T, Koga T, Martin TJ, Suda T (1990) Origin of osteoclasts: mature monocytes and macrophages are capable of differentiating into osteoclasts under a suitable microenvironment prepared by bone marrow-derived stromal cells. Proc Natl Acad Sci USA 87(18):7260–7264. https://doi.org/10.1073/pnas.87.18.7260
Lacey DL, Timms E, Tan HL, Kelley MJ, Dunstan CR, Burgess T, Elliott R, Colombero A, Elliott G, Scully S, Hsu H, Sullivan J, Hawkins N, Davy E, Capparelli C, Eli A, Qian YX, Kaufman S, Sarosi I, Shalhoub V, Senaldi G, Guo J, Delaney J, Boyle WJ (1998) Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93(2):165–176
Yasuda H, Shima N, Nakagawa N, Yamaguchi K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A, Tsuda E, Morinaga T, Higashio K, Udagawa N, Takahashi N, Suda T (1998) Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc Natl Acad Sci USA 95(7):3597–3602. https://doi.org/10.1073/pnas.95.7.3597
Suda T, Takahashi N, Udagawa N, Jimi E, Gillespie MT, Martin TJ (1999) Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr Rev 20(3):345–357. https://doi.org/10.1210/edrv.20.3.0367
Enomoto H, Shiojiri S, Hoshi K, Furuichi T, Fukuyama R, Yoshida CA, Kanatani N, Nakamura R, Mizuno A, Zanma A, Yano K, Yasuda H, Higashio K, Takada K, Komori T (2003) Induction of osteoclast differentiation by Runx2 through receptor activator of nuclear factor-kappa B ligand (RANKL) and osteoprotegerin regulation and partial rescue of osteoclastogenesis in Runx2-/- mice by RANKL transgene. J Biol Chem 278(26):23971–23977. https://doi.org/10.1074/jbc.M302457200
Martin A, Xiong J, Koromila T, Ji JS, Chang S, Song YS, Miller JL, Han CY, Kostenuik P, Krum SA, Chimge NO, Gabet Y, Frenkel B (2015) Estrogens antagonize RUNX2-mediated osteoblast-driven osteoclastogenesis through regulating RANKL membrane association. Bone 75:96–104. https://doi.org/10.1016/j.bone.2015.02.007
Byon CH, Sun Y, Chen J, Yuan K, Mao X, Heath JM, Anderson PG, Tintut Y, Demer LL, Wang D, Chen Y (2011) Runx2-upregulated receptor activator of nuclear factor kappaB ligand in calcifying smooth muscle cells promotes migration and osteoclastic differentiation of macrophages. Arterioscler Thromb Vasc Biol 31(6):1387–1396. https://doi.org/10.1161/atvbaha.110.222547
Zhang C, Zheng S, Wang Y, Zhao Y, Zhu J, Ge L (2010) Mutational analysis of RUNX2 gene in Chinese patients with cleidocranial dysplasia. Mutagenesis 25(6):589–594. https://doi.org/10.1093/mutage/geq044
Liu Y, Sun X, Zhang X, Wang X, Zhang C, Zheng S (2019) RUNX2 mutation impairs osteogenic differentiation of dental follicle cells. Arch Oral Biol 97:156–164. https://doi.org/10.1016/j.archoralbio.2018.10.029
Dincsoy Bir F, Dinckan N, Guven Y, Bas F, Altunoglu U, Kuvvetli SS, Poyrazoglu S, Toksoy G, Kayserili H, Uyguner ZO (2017) Cleidocranial dysplasia: clinical, endocrinologic and molecular findings in 15 patients from 11 families. Eur J Med Genet 60(3):163–168. https://doi.org/10.1016/j.ejmg.2016.12.007
Mundlos S (1999) Cleidocranial dysplasia: clinical and molecular genetics. J Med Genet 36(3):177–182
Bergwitz C, Prochnau A, Mayr B, Kramer FJ, Rittierodt M, Berten HL, Hausamen JE, Brabant G (2001) Identification of novel CBFA1/RUNX2 mutations causing cleidocranial dysplasia. J Inherit Metab Dis 24(6):648–656. https://doi.org/10.1023/a:1012758925617
Yoshida T, Kanegane H, Osato M, Yanagida M, Miyawaki T, Ito Y, Shigesada K (2002) Functional analysis of RUNX2 mutations in Japanese patients with cleidocranial dysplasia demonstrates novel genotype-phenotype correlations. Am J Hum Genet 71(4):724–738. https://doi.org/10.1086/342717
Song I, Kim JH, Kim K, Jin HM, Youn BU, Kim N (2009) Regulatory mechanism of NFATc1 in RANKL-induced osteoclast activation. FEBS Lett 583(14):2435–2440. https://doi.org/10.1016/j.febslet.2009.06.047
Asagiri M, Takayanagi H (2007) The molecular understanding of osteoclast differentiation. Bone 40(2):251–264. https://doi.org/10.1016/j.bone.2006.09.023
Lu SY, Li M, Lin YL (2014) Mitf regulates osteoclastogenesis by modulating NFATc1 activity. Exp Cell Res 328(1):32–43. https://doi.org/10.1016/j.yexcr.2014.08.018
Matsumoto M, Kogawa M, Wada S, Takayanagi H, Tsujimoto M, Katayama S, Hisatake K, Nogi Y (2004) Essential role of p38 mitogen-activated protein kinase in cathepsin K gene expression during osteoclastogenesis through association of NFATc1 and PU.1. J Biol Chem 279 (44):45969–45979. https://doi.org/10.1074/jbc.M408795200
Matsuo K, Galson DL, Zhao C, Peng L, Laplace C, Wang KZ, Bachler MA, Amano H, Aburatani H, Ishikawa H, Wagner EF (2004) Nuclear factor of activated T-cells (NFAT) rescues osteoclastogenesis in precursors lacking c-Fos. J Biol Chem 279(25):26475–26480. https://doi.org/10.1074/jbc.M313973200
Moon JB, Kim JH, Kim K, Youn BU, Ko A, Lee SY, Kim N (2012) Akt induces osteoclast differentiation through regulating the GSK3beta/NFATc1 signaling cascade. J Immunol 188(1):163–169. https://doi.org/10.4049/jimmunol.1101254
Bhaskar PT, Hay N (2007) The two TORCs and Akt. Dev Cell 12(4):487–502. https://doi.org/10.1016/j.devcel.2007.03.020
Wu M, Chen W, Lu Y, Zhu G, Hao L, Li YP (2017) Galpha13 negatively controls osteoclastogenesis through inhibition of the Akt-GSK3beta-NFATc1 signalling pathway. Nat Commun 8:13700. https://doi.org/10.1038/ncomms13700
Jang HD, Shin JH, Park DR, Hong JH, Yoon K, Ko R, Ko CY, Kim HS, Jeong D, Kim N, Lee SY (2011) Inactivation of glycogen synthase kinase-3beta is required for osteoclast differentiation. J Biol Chem 286(45):39043–39050. https://doi.org/10.1074/jbc.M111.256768
Copp J, Manning G, Hunter T (2009) TORC-specific phosphorylation of mammalian target of rapamycin (mTOR): phospho-Ser2481 is a marker for intact mTOR signaling complex 2. Cancer Res 69(5):1821–1827. https://doi.org/10.1158/0008-5472.can-08-3014
Masri J, Bernath A, Martin J, Jo OD, Vartanian R, Funk A, Gera J (2007) mTORC2 activity is elevated in gliomas and promotes growth and cell motility via overexpression of rictor. Cancer Res 67(24):11712–11720. https://doi.org/10.1158/0008-5472.can-07-2223
Huang S, Yang ZJ, Yu C, Sinicrope FA (2011) Inhibition of mTOR kinase by AZD8055 can antagonize chemotherapy-induced cell death through autophagy induction and down-regulation of p62/sequestosome 1. J Biol Chem 286(46):40002–40012. https://doi.org/10.1074/jbc.M111.297432
Tandon M, Chen Z, Pratap J (2014) Runx2 activates PI3K/Akt signaling via mTORC2 regulation in invasive breast cancer cells. Breast Cancer Res 16(1):R16. https://doi.org/10.1186/bcr3611
Lotinun S, Kiviranta R, Matsubara T, Alzate JA, Neff L, Luth A, Koskivirta I, Kleuser B, Vacher J, Vuorio E, Horne WC, Baron R (2013) Osteoclast-specific cathepsin K deletion stimulates S1P-dependent bone formation. J Clin Investig 123(2):666–681. https://doi.org/10.1172/jci64840
Chen W, Yang S, Abe Y, Li M, Wang Y, Shao J, Li E, Li YP (2007) Novel pycnodysostosis mouse model uncovers cathepsin K function as a potential regulator of osteoclast apoptosis and senescence. Hum Mol Genet 16(4):410–423. https://doi.org/10.1093/hmg/ddl474
Saftig P, Hunziker E, Wehmeyer O, Jones S, Boyde A, Rommerskirch W, Moritz JD, Schu P, von Figura K (1998) Impaired osteoclastic bone resorption leads to osteopetrosis in cathepsin-K-deficient mice. Proc Natl Acad Sci USA 95(23):13453–13458. https://doi.org/10.1073/pnas.95.23.13453
Panwar P, Soe K, Guido RV, Bueno RV, Delaisse JM, Bromme D (2016) A novel approach to inhibit bone resorption: exosite inhibitors against cathepsin K. Br J Pharmacol 173(2):396–410. https://doi.org/10.1111/bph.13383
Prates TP, Taira TM, Holanda MC, Bignardi LA, Salvador SL, Zamboni DS, Cunha FQ, Fukada SY (2014) NOD2 contributes to Porphyromonas gingivalis-induced bone resorption. J Dental Res 93(11):1155–1162. https://doi.org/10.1177/0022034514551770
Sharmin F, McDermott C, Lieberman J, Sanjay A, Khan Y (2017) Dual growth factor delivery from biofunctionalized allografts: Sequential VEGF and BMP-2 release to stimulate allograft remodeling. J Orthop Res 35(5):1086–1095. https://doi.org/10.1002/jor.23287
Faienza MF, Ventura A, Piacente L, Ciccarelli M, Gigante M, Gesualdo L, Colucci S, Cavallo L, Grano M, Brunetti G (2014) Osteoclastogenic potential of peripheral blood mononuclear cells in cleidocranial dysplasia. Int J Med Sci 11(4):356–364. https://doi.org/10.7150/ijms.7793
Yoda S, Suda N, Kitahara Y, Komori T, Ohyama K (2004) Delayed tooth eruption and suppressed osteoclast number in the eruption pathway of heterozygous Runx2/Cbfa1 knockout mice. Arch Oral Biol 49(6):435–442. https://doi.org/10.1016/j.archoralbio.2004.01.010
Collin-Osdoby P, Osdoby P (2012) RANKL-mediated osteoclast formation from murine RAW 264.7 cells. Methods Mol Biol 816:187–202. https://doi.org/10.1007/978-1-61779-415-5_13
Laha D, Deb M, Das H (2019) KLF2 (kruppel-like factor 2 [lung]) regulates osteoclastogenesis by modulating autophagy. Autophagy 15(12):2063–2075. https://doi.org/10.1080/15548627.2019.1596491
Hao S, Meng J, Zhang Y, Liu J, Nie X, Wu F, Yang Y, Wang C, Gu N, Xu H (2017) Macrophage phenotypic mechanomodulation of enhancing bone regeneration by superparamagnetic scaffold upon magnetization. Biomaterials 140:16–25. https://doi.org/10.1016/j.biomaterials.2017.06.013
Yamamoto A, Miyazaki T, Kadono Y, Takayanagi H, Miura T, Nishina H, Katada T, Wakabayashi K, Oda H, Nakamura K, Tanaka S (2002) Possible involvement of IkappaB kinase 2 and MKK7 in osteoclastogenesis induced by receptor activator of nuclear factor kappaB ligand. J Bone Miner Res 17(4):612–621. https://doi.org/10.1359/jbmr.2002.17.4.612
Acknowledgments
This work was supported by the grants of National Natural Science Foundation of China (grant number 81771053, 81772873 and 81970920). The authors are grateful to all the participants in this study.
Author information
Authors and Affiliations
Contributions
Conceptualization: YX, YW and SZ; Methodology: YX, DL, JL and CZ; Data Curation: YX and YL; Writing—Original Draft Preparation: YX; Writing—Review & Editing: YW and SZ.
Corresponding authors
Ethics declarations
Conflict of interest
Yuejiao Xin, Yang Liu, Dandan Liu, Jie Li, Chenying Zhang, Yixiang Wang, and Shuguo Zheng declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This study was approved by the Ethical Committee of Peking University School and the Hospital of Stomatology (Approval No. PKUSSIRB-2012004) and conducted strictly in accordance with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All participants signed an informed consent prior to study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xin, Y., Liu, Y., Liu, D. et al. New Function of RUNX2 in Regulating Osteoclast Differentiation via the AKT/NFATc1/CTSK Axis. Calcif Tissue Int 106, 553–566 (2020). https://doi.org/10.1007/s00223-020-00666-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-020-00666-7