Abstract
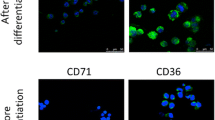
Macrophages are characterized by phenotypical and functional heterogeneity. In different microenvironments, macrophages can polarize into two types: classically activated macrophages (M1) or alternatively activated macrophages (M2). M1 macrophages are a well-known bacteriostatic macrophage, and conversely, M2 macrophages may play an important role in tumor growth and tissue remodeling. M1 macrophages have been reported to have high intracellular iron stores, while M2 macrophages contain lower intracellular iron. It has been well-described that disturbances of iron homeostasis are associated with altered immune function. Thus, it is important to investigate if chronic iron overload is capable of polarizing macrophages. Human monocytic leukemia THP-1 cells were maintained in culture medium that contained 100 μM ferrous sulfate heptahydrate (FeSO4) (I-THP-1) and differentiated into THP-1-derived macrophages (I-TDMs) by induction with phorbol 12-myristate 13-acetate (PMA). We characterized that I-TDMs not only enhanced the surface expression of CD163 and CD206 but also increased arginase and decreased iNOS protein expression. I-TDMs enhanced pSTAT6 expression and decreased pSTAT1 and NF-κB expressions. Furthermore, the gene expression profile of I-TDMs was comparable with M2 macrophages by performing human oligonucleotide DNA microarray analysis. Finally, functional assays demonstrated I-TDMs secreted higher levels of IL-10 but not M1 cytokines. Additionally, the conditional medium of I-TDMs had enhanced migration and increased invasion of A375 melanoma cells which was similar to the characteristics of tumor-associated macrophages. Taken together, we demonstrated that THP-1-derived macrophages polarized to a phenotype of M2-like characteristics when subjected to chronic iron overload.






Similar content being viewed by others
References
Wang XF, Wang HS, Zhang F, Guo Q, Wang H, Wang KF, Zhang G, Bu XZ, Cai SH, du J (2014) Nodal promotes the generation of M2-like macrophages and downregulates the expression of IL-12. Eur J Immunol 44(1):173–183
Tiemessen MM, Jagger AL, Evans HG, van Herwijnen MJ, John S, Taams LS (2007) CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/macrophages. Proc Natl Acad Sci U S A 104(49):19446–19451
Lawrence T, Natoli G (2011) Transcriptional regulation of macrophage polarization: enabling diversity with identity. Nat Rev Immunol 11(11):750–761
Bannon P, Wood S, Restivo T, Campbell L, Hardman MJ, Mace KA (2013) Diabetes induces stable intrinsic changes to myeloid cells that contribute to chronic inflammation during wound healing in mice. Dis Model Mech 6(6):1434–1447
Kittan NA, Allen RM, Dhaliwal A, Cavassani KA, Schaller M, Gallagher KA, Carson WF IV, Mukherjee S, Grembecka J, Cierpicki T, Jarai G, Westwick J, Kunkel SL, Hogaboam CM (2013) Cytokine induced phenotypic and epigenetic signatures are key to establishing specific macrophage phenotypes. PLoS One 8(10):e78045
Medzhitov R, Janeway C Jr (2000) Innate immunity. N Engl J Med 343(5):338–344
Recalcati S, Locati M, Marini A, Santambrogio P, Zaninotto F, De Pizzol M et al (2010) Differential regulation of iron homeostasis during human macrophage polarized activation. Eur J Immunol 40(3):824–835
Kao JK, Wang SC, Ho LW, Huang SW, Chang SH, Yang RC et al (2016) Chronic iron overload results in impaired bacterial killing of THP-1 derived macrophage through the inhibition of lysosomal acidification. PLoS One 11(5):e0156713
Trouplin V, Boucherit N, Gorvel L, Conti F, Mottola G, Ghigo E (2013) Bone marrow-derived macrophage production. J Vis Exp 81:e50966
Liu T, Zhang L, Joo D, Sun SC (2017) NF-kappaB signaling in inflammation. Signal Transduct Target Ther 2:17023
Darnell JE Jr, Kerr IM, Stark GR (1994) Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science. 264(5164):1415–1421
Munder M, Eichmann K, Moran JM, Centeno F, Soler G, Modolell M (1999) Th1/Th2-regulated expression of arginase isoforms in murine macrophages and dendritic cells. J Immunol 163(7):3771–3777
Munder M, Eichmann K, Modolell M (1998) Alternative metabolic states in murine macrophages reflected by the nitric oxide synthase/arginase balance: competitive regulation by CD4+ T cells correlates with Th1/Th2 phenotype. J Immunol 160(11):5347–5354
Ramsay G (1998) DNA chips: state-of-the art. Nat Biotechnol 16(1):40–44
Patel U, Rajasingh S, Samanta S, Cao T, Dawn B, Rajasingh J (2017) Macrophage polarization in response to epigenetic modifiers during infection and inflammation. Drug Discov Today 22(1):186–193
Benoit M, Desnues B, Mege JL (2008) Macrophage polarization in bacterial infections. J Immunol 181(6):3733–3739
Mantovani A, Sozzani S, Locati M, Allavena P, Sica A (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23(11):549–555
Condeelis J, Pollard JW (2006) Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 124(2):263–266
Gordon S, Taylor PR (2005) Monocyte and macrophage heterogeneity. Nat Rev Immunol 5(12):953–964
Davis MJ, Tsang TM, Qiu Y, Dayrit JK, Freij JB, Huffnagle GB et al (2013) Macrophage M1/M2 polarization dynamically adapts to changes in cytokine microenvironments in Cryptococcus neoformans infection. MBio 4(3):e00264–e00213
Spence S, Fitzsimons A, Boyd CR, Kessler J, Fitzgerald D, Elliott J, Gabhann JN, Smith S, Sica A, Hams E, Saunders SP, Jefferies CA, Fallon PG, McAuley D, Kissenpfennig A, Johnston JA (2013) Suppressors of cytokine signaling 2 and 3 diametrically control macrophage polarization. Immunity. 38(1):66–78
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Invest 122(3):787–795
Nathan CF, Murray HW, Wiebe ME, Rubin BY (1983) Identification of interferon-gamma as the lymphokine that activates human macrophage oxidative metabolism and antimicrobial activity. J Exp Med 158(3):670–689
Lisi L, Ciotti GM, Braun D, Kalinin S, Curro D, Dello Russo C et al (2017) Expression of iNOS, CD163 and ARG-1 taken as M1 and M2 markers of microglial polarization in human glioblastoma and the surrounding normal parenchyma. Neurosci Lett 645:106–112
Mantovani A (2009) Cancer: inflaming metastasis. Nature. 457(7225):36–37
Sica A, Larghi P, Mancino A, Rubino L, Porta C, Totaro MG, Rimoldi M, Biswas SK, Allavena P, Mantovani A (2008) Macrophage polarization in tumour progression. Semin Cancer Biol 18(5):349–355
Pollard JW (2009) Trophic macrophages in development and disease. Nat Rev Immunol 9(4):259–270
Wyckoff J, Wang W, Lin EY, Wang Y, Pixley F, Stanley ER, Graf T, Pollard JW, Segall J, Condeelis J (2004) A paracrine loop between tumor cells and macrophages is required for tumor cell migration in mammary tumors. Cancer Res 64(19):7022–7029
Kitamura T, Kometani K, Hashida H, Matsunaga A, Miyoshi H, Hosogi H, Aoki M, Oshima M, Hattori M, Takabayashi A, Minato N, Taketo MM (2007) SMAD4-deficient intestinal tumors recruit CCR1+ myeloid cells that promote invasion. Nat Genet 39(4):467–475
Bode JG, Ehlting C, Haussinger D (2012) The macrophage response towards LPS and its control through the p38(MAPK)-STAT3 axis. Cell Signal 24(6):1185–1194
Tan HY, Wang N, Li S, Hong M, Wang X, Feng Y (2016) The reactive oxygen species in macrophage polarization: reflecting its dual role in progression and treatment of human diseases. Oxidative Med Cell Longev 2016:2795090
Fang L, Hodge J, Saaoud F, Wang J, Iwanowycz S, Wang Y, Hui Y, Evans TD, Razani B, Fan D (2017) Transcriptional factor EB regulates macrophage polarization in the tumor microenvironment. Oncoimmunology. 6(5):e1312042
Cairo G, Recalcati S, Mantovani A, Locati M (2011) Iron trafficking and metabolism in macrophages: contribution to the polarized phenotype. Trends Immunol 32(6):241–247
Sohn YS, Ghoti H, Breuer W, Rachmilewitz E, Attar S, Weiss G, Cabantchik ZI (2012) The role of endocytic pathways in cellular uptake of plasma non-transferrin iron. Haematologica. 97(5):670–678
Agoro R, Taleb M, Quesniaux VFJ, Mura C (2018) Cell iron status influences macrophage polarization. PLoS One 13(5):e0196921
Mikita J, Dubourdieu-Cassagno N, Deloire MS, Vekris A, Biran M, Raffard G, Brochet B, Canron MH, Franconi JM, Boiziau C, Petry KG (2011) Altered M1/M2 activation patterns of monocytes in severe relapsing experimental rat model of multiple sclerosis. Amelioration of clinical status by M2 activated monocyte administration. Mult Scler 17(1):2–15
Khallou-Laschet J, Varthaman A, Fornasa G, Compain C, Gaston AT, Clement M et al (2010) Macrophage plasticity in experimental atherosclerosis. PLoS One 5(1):e8852
Labonte AC, Bachali P, Catalina M, Robl R, Geraci NS, Lipsky P et al (2018) Identification of perturbations in macrophage polarization in active systemic lupus erythematosus. J Immunol 200:45.5–45.5
Funes SC, Rios M, Escobar-Vera J, Kalergis AM (2018) Implications of macrophage polarization in autoimmunity. Immunology. 154(2):186–195
Fonseca-Nunes A, Jakszyn P, Agudo A (2014) Iron and cancer risk--a systematic review and meta-analysis of the epidemiological evidence. Cancer Epidemiol Biomark Prev 23(1):12–31
Manz DH, Blanchette NL, Paul BT, Torti FM, Torti SV (2016) Iron and cancer: recent insights. Ann N Y Acad Sci 1368(1):149–161
Chang VC, Cotterchio M, Khoo E (2019) Iron intake, body iron status, and risk of breast cancer: a systematic review and meta-analysis. BMC Cancer 19(1):543
Chung WS, Lin CL, Lin CL, Kao CH (2015) Thalassaemia and risk of cancer: a population-based cohort study. J Epidemiol Community Health 69(11):1066–1070
Acknowledgments
We are grateful to Chien-Sheng Hsu for his excellent technical assistance.
Funding
This work was supported by grants from the Changhua Christian Hospital Research Program (NCHU-CCH 10503), Taiwan, Republic of China.
Author information
Authors and Affiliations
Contributions
J.J.S., J.K.K., and S.C.W. participated in experimental design; performed research; collected, analyzed, and interpreted data; performed statistical analysis; and drafted and revised the manuscript; L.W.H., R.C.Y., and S.W.H. performed research, collected and analyzed data, and edited the manuscript; C.H.L., M.S.L., and R.C.Y. participated in the collection, analysis, and interpretation of data, and they edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kao, JK., Wang, SC., Ho, LW. et al. M2-like polarization of THP-1 monocyte-derived macrophages under chronic iron overload. Ann Hematol 99, 431–441 (2020). https://doi.org/10.1007/s00277-020-03916-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-03916-8