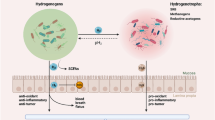
Abstract
Stable intracellular and intercellular osmolarity is vital for all physiological processes. Although it is the first organ that receives food, the osmolarity around the mouth epithelium has never been systematically investigated. We found that oral epithelial cells are a population of ignored cells routinely exposed to hypertonic environments mainly composed of saline, glucose, etc. in vivo after chewing food. By using cultured oral epithelial cells as an in vitro model, we found that the hypotonic environments caused by both high NaCl and high glucose induced cell death in a dose- and time-dependent manner. Transcriptomics revealed similar expression profiles after high NaCl and high glucose stimulation. Most of the common differentially expressed genes were enriched in “mitophagy” and “autophagy” according to KEGG pathway enrichment analysis. Hypertonic stimulation for 1 to 6 h resulted in autophagosome formation. The activation of autophagy protected cells from high osmolarity-induced cell death. The activation of Hsp70 by the pharmacological activator handelin significantly improved the cell survival rate after hypertonic stimulation. The protective role of Hsp70 activation was partially dependent on autophagy activation, indicating a crosstalk between Hsp70 and autophagy in hypertonic stress response. The extract of the handelin-containing herb Chrysanthemum indicum significantly protected oral epithelial cells from hypertonic-induced death, providing an inexpensive way to protect against hypertonic-induced oral epithelial damage. In conclusion, the present study emphasized the importance of changes in osmolarity in oral health for the first time. The identification of novel compounds or herbal plant extracts that can activate autophagy or HSPs may contribute to oral health and the food industry.





Similar content being viewed by others
References
Arenholt-Bindslev D, Jepsen A, MacCallum DK, Lillie JH (1987) The growth and structure of human oral keratinocytes in culture. J Invest Dermatol 88(3):314–319
Beck FX, Neuhofer W, Muller E (2000) Molecular chaperones in the kidney: distribution, putative roles, and regulation. Am J Physiol Ren Physiol 279(2):F203–F215. https://doi.org/10.1152/ajprenal.2000.279.2.F203
Bobkova NV, Evgen'ev M, Garbuz DG, Kulikov AM, Morozov A, Samokhin A, Velmeshev D, Medvinskaya N, Nesterova I, Pollock A, Nudler E (2015) Exogenous Hsp70 delays senescence and improves cognitive function in aging mice. Proc Natl Acad Sci U S A 112(52):16006–16011. https://doi.org/10.1073/pnas.1516131112
Brinkman JE, Sharma S (2019) Physiology, Body Fluids. StatPearls, Treasure Island
Burg MB, Ferraris JD, Dmitrieva NI (2007) Cellular response to hyperosmotic stresses. Physiol Rev 87(4):1441–1474. https://doi.org/10.1152/physrev.00056.2006
Das G, Shravage BV, Baehrecke EH (2012) Regulation and function of autophagy during cell survival and cell death. Cold Spring Harb Perspect Biol 4(6). https://doi.org/10.1101/cshperspect.a008813
Edkins AL, Price JT, Pockley AG, Blatch GL (2018) Heat shock proteins as modulators and therapeutic targets of chronic disease: an integrated perspective. Philos Trans R Soc Lond Ser B Biol Sci 373(1738). https://doi.org/10.1098/rstb.2016.0521
Evans CG, Chang L, Gestwicki JE (2010) Heat shock protein 70 (hsp70) as an emerging drug target. J Med Chem 53(12):4585–4602. https://doi.org/10.1021/jm100054f
Fernandes TR, Pontieri V, Moretti AI, Teixeira DO, Abatepaulo F, Soriano FG, Negri EM, Velasco IT, Souza HP (2007) Hypertonic saline solution increases the expression of heat shock protein 70 and improves lung inflammation early after reperfusion in a rodent model of controlled hemorrhage. Shock 27(2):172–178. https://doi.org/10.1097/01.shk.0000238062.46708.a5
Finan JD, Guilak F (2010) The effects of osmotic stress on the structure and function of the cell nucleus. J Cell Biochem 109(3):460–467. https://doi.org/10.1002/jcb.22437
Garcia-Perez A, Burg MB (1991) Renal medullary organic osmolytes. Physiol Rev 71(4):1081–1115. https://doi.org/10.1152/physrev.1991.71.4.1081
Hendry EB (1962) The osmotic pressure and chemical composition of human body fluids. Clin Chem 8:246–265
Jacob P, Hirt H, Bendahmane A (2017) The heat-shock protein/chaperone network and multiple stress resistance. Plant Biotechnol J 15(4):405–414. https://doi.org/10.1111/pbi.12659
Kaushik S, Cuervo AM (2018) The coming of age of chaperone-mediated autophagy. Nat Rev Mol Cell Biol 19(6):365–381. https://doi.org/10.1038/s41580-018-0001-6
Knepper MA (1982) Measurement of osmolality in kidney slices using vapor pressure osmometry. Kidney Int 21(4):653–655
Kuwabara M, Hisatome I, Roncal-Jimenez CA, Niwa K, Andres-Hernando A, Jensen T, Bjornstad P, Milagres T, Cicerchi C, Song Z, Garcia G, Sanchez-Lozada LG, Ohno M, Lanaspa MA, Johnson RJ (2017) Increased serum sodium and serum Osmolarity are independent risk factors for developing chronic kidney disease; 5 year cohort study. PLoS One 12(1):e0169137. https://doi.org/10.1371/journal.pone.0169137
Leitch V, Agre P, King LS (2001) Altered ubiquitination and stability of aquaporin-1 in hypertonic stress. Proc Natl Acad Sci U S A 98(5):2894–2898. https://doi.org/10.1073/pnas.041616498
Masin J, Fiser R, Linhartova I, Osicka R, Bumba L, Hewlett EL, Benz R, Sebo P (2013) Differences in purinergic amplification of osmotic cell lysis by the pore-forming RTX toxins Bordetella pertussis CyaA and Actinobacillus pleuropneumoniae ApxIA: the role of pore size. Infect Immun 81(12):4571–4582. https://doi.org/10.1128/IAI.00711-13
Massey AC, Kaushik S, Sovak G, Kiffin R, Cuervo AM (2006) Consequences of the selective blockage of chaperone-mediated autophagy. Proc Natl Acad Sci U S A 103(15):5805–5810. https://doi.org/10.1073/pnas.0507436103
Mejean C, Morzel M, Neyraud E, Issanchou S, Martin C, Bozonnet S, Urbano C, Schlich P, Hercberg S, Peneau S, Feron G (2015) Salivary composition is associated with liking and usual nutrient intake. PLoS One 10(9):e0137473. https://doi.org/10.1371/journal.pone.0137473
Nalpas NC, Magee DA, Conlon KM, Browne JA, Healy C, McLoughlin KE, Rue-Albrecht K, McGettigan PA, Killick KE, Gormley E, Gordon SV, MacHugh DE (2015) RNA sequencing provides exquisite insight into the manipulation of the alveolar macrophage by tubercle bacilli. Sci Rep 5:13629. https://doi.org/10.1038/srep13629
Nedelsky NB, Todd PK, Taylor JP (2008) Autophagy and the ubiquitin-proteasome system: collaborators in neuroprotection. Biochim Biophys Acta 1782(12):691–699. https://doi.org/10.1016/j.bbadis.2008.10.002
Nunes P, Ernandez T, Roth I, Qiao X, Strebel D, Bouley R, Charollais A, Ramadori P, Foti M, Meda P, Feraille E, Brown D, Hasler U (2013) Hypertonic stress promotes autophagy and microtubule-dependent autophagosomal clusters. Autophagy 9(4):550–567. https://doi.org/10.4161/auto.23662
Overduin J, Tylee TS, Frayo RS, Cummings DE (2014) Hyperosmolarity in the small intestine contributes to postprandial ghrelin suppression. Am J Physiol Gastrointest Liver Physiol 306(12):G1108–G1116. https://doi.org/10.1152/ajpgi.00072.2014
Pachori A, Kambalimath H, Maran S, Niranjan B, Bhambhani G, Malhotra G (2018) Evaluation of changes in salivary pH after intake of different eatables and beverages in children at different time intervals. Int J Clin Pediatr Dent 11(3):177–182. https://doi.org/10.5005/jp-journals-10005-1507
Park SH, Baek KH, Shin I, Shin I (2018) Subcellular Hsp70 inhibitors promote Cancer cell death via different mechanisms. Cell Chem Biol 25(10):1242–1254 e1248. https://doi.org/10.1016/j.chembiol.2018.06.010
Ritossa F (1962) A new puffing pattern induced by temperature shock and DNP in drosophila. Experientia 18(12):571–573. https://doi.org/10.1007/bf02172188
Rondon-Berrios H, Argyropoulos C, Ing TS, Raj DS, Malhotra D, Agaba EI, Rohrscheib M, Khitan ZJ, Murata GH, Shapiro JI, Tzamaloukas AH (2017) Hypertonicity: clinical entities, manifestations and treatment. World J Nephrol 6(1):1–13. https://doi.org/10.5527/wjn.v6.i1.1
Sala G, Marinig D, Arosio A, Ferrarese C (2016) Role of chaperone-mediated autophagy dysfunctions in the pathogenesis of Parkinson's disease. Front Mol Neurosci 9:157. https://doi.org/10.3389/fnmol.2016.00157
Sheikh-Hamad D, Garcia-Perez A, Ferraris JD, Peters EM, Burg MB (1994) Induction of gene expression by heat shock versus osmotic stress. Am J Phys 267(1 Pt 2):F28–F34. https://doi.org/10.1152/ajprenal.1994.267.1.F28
Shim EH, Kim JI, Bang ES, Heo JS, Lee JS, Kim EY, Lee JE, Park WY, Kim SH, Kim HS, Smithies O, Jang JJ, Jin DI, Seo JS (2002) Targeted disruption of hsp70.1 sensitizes to osmotic stress. EMBO Rep 3(9):857–861. https://doi.org/10.1093/embo-reports/kvf175
Stinson S (2012) Human biology : an evolutionary and biocultural perspective, 2nd edn. Wiley-Blackwell, Hoboken
Sun S, Jiang P, Su W, Xiang Y, Li J, Zeng L, Yang S (2016) Wild chrysanthemum extract prevents UVB radiation-induced acute cell death and photoaging. Cytotechnology 68(2):229–240. https://doi.org/10.1007/s10616-014-9773-5
Tutar L, Tutar Y (2010) Heat shock proteins; an overview. Curr Pharm Biotechnol 11(2):216–222
Vabulas RM, Raychaudhuri S, Hayer-Hartl M, Hartl FU (2010) Protein folding in the cytoplasm and the heat shock response. Cold Spring Harb Perspect Biol 2(12):a004390. https://doi.org/10.1101/cshperspect.a004390
Wang LC, Liao LX, Lv HN, Liu D, Dong W, Zhu J, Chen JF, Shi ML, Fu G, Song XM, Jiang Y, Zeng KW, Tu PF (2017) Highly selective activation of heat shock protein 70 by allosteric regulation provides an insight into efficient Neuroinflammation inhibition. EBioMedicine 23:160–172. https://doi.org/10.1016/j.ebiom.2017.08.011
Woo SK, Lee SD, Na KY, Park WK, Kwon HM (2002) TonEBP/NFAT5 stimulates transcription of HSP70 in response to hypertonicity. Mol Cell Biol 22(16):5753–5760. https://doi.org/10.1128/mcb.22.16.5753-5760.2002
Xiang Y, Yan C, Guo X, Zhou K, Li S, Gao Q, Wang X, Zhao F, Liu J, Lee WH, Zhang Y (2014) Host-derived, pore-forming toxin-like protein and trefoil factor complex protects the host against microbial infection. Proc Natl Acad Sci U S A 111(18):6702–6707. https://doi.org/10.1073/pnas.1321317111
Yang Z, Klionsky DJ (2009) An overview of the molecular mechanism of autophagy. Curr Top Microbiol Immunol 335:1–32. https://doi.org/10.1007/978-3-642-00302-8_1
Yeung KY, Medvedovic M, Bumgarner RE (2003) Clustering gene-expression data with repeated measurements. Genome Biol 4(5):R34. https://doi.org/10.1186/gb-2003-4-5-r34
Zhang Y (2015) Why do we study animal toxins? Dongwuxue Yanjiu 36(4):183–222
Funding
This work was financially supported by the National Natural Science Foundation of China to Yang Xiang (91649120) and Sujiao Sun (81560697), the Research Foundation of China Tobacco Company (110201601005577 (XX-05)), the Research Foundation of China Tobacco Yunnan Industrial Co., Ltd. (2018XY04) and Jiangxi Province (2018ACB21036, 20192 BC23004 and 20181BCD40001).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, J., Zhang, H., Sun, S. et al. Autophagy and Hsp70 activation alleviate oral epithelial cell death induced by food-derived hypertonicity. Cell Stress and Chaperones 25, 253–264 (2020). https://doi.org/10.1007/s12192-020-01068-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-020-01068-2